To evaluate whether the location and number of lung biopsies obtained by video-assisted thoracoscopy (VAT) influence the diagnosis of diffuse interstitial lung disease (ILD). To assess the applicability of an ambulatory surgery program (ASP).
MethodsProspective, multicenter study of VAT lung biopsies due to suspected ILD from January 2007 to December 2009, including 224 patients from 13 Spanish centers (mean age 57.1 years; 52.6% females). Data were prospectively collected in every institution and sent to the coordination center for analysis.
ResultsThe most affected areas in high-resolution chest CT were the lower lobes (55%). Bronchoscopy was performed in 84% and transbronchial biopsy in 49.1%. In 179 cases (79.9%), more than one biopsy was performed, with a diagnostic agreement of 97.2%. A definitive histopathologic diagnosis was obtained in 195 patients (87%). Idiopathic pulmonary fibrosis was the most frequent diagnosis (26%). There were no statistically significant factors that could predict a greater diagnostic yield (neither anatomical location nor number of biopsies). Seventy patients (31.3%) were included in an ASP. After discharge, there were complications in 12 patients (5.4%), similar between patients admitted postoperatively (9/154: 5.8%) and those included in an ASP (3/70: 4.3%).
ConclusionsAnatomical location and number of lung biopsy specimens did not seem to influence the diagnosis. The patients included in an ASP had a complication rate comparable to that of the hospitalized, so this procedure can be included in a surgical outpatient program. Lung biopsy obtained by VAT is a powerful and safe tool for diagnosis of suspected ILD, resulting in a definitive diagnosis for the majority of patients with a low morbidity rate.
Analizar si la localización y el número de biopsias pulmonares obtenidas por videotoracoscopia (BP-VTC) influyen en el diagnóstico de las enfermedades pulmonares intersticiales difusas (EPID) y evaluar la aplicabilidad de un programa de cirugía ambulatoria (CA).
Pacientes y métodoEstudio prospectivo y multicéntrico de BP-VTC practicadas en pacientes con sospecha de EPID desde enero de 2007 a diciembre de 2009, incluyendo 224 pacientes de 13 centros españoles. Los datos fueron recogidos de manera prospectiva en cada centro y enviados al centro coordinador para su análisis.
ResultadosLas zonas más afectadas en la TACAR torácica fueron los lóbulos inferiores (55%). Se llevó a cabo broncoscopia en el 84%, y biopsia transbronquial en el 49,1%. En 179 casos (79,9%) se realizó más de una biopsia (concordancia diagnóstica: 97,2%). Se obtuvo un diagnóstico histopatológico definitivo en 195 pacientes (87%). No se hallaron factores estadísticamente significativos que pudieran predecir un mayor rendimiento diagnóstico (ni la localización anatómica de la biopsia ni el número de ellas). Setenta pacientes (31,3%) fueron incluidos en un programa de CA. Los índices de morbilidad tras el alta fueron comparables entre ingresados (9/154: 5.8%) y ambulatorios (3/70: 4.3%).
ConclusionesLa localización anatómica y el número de BP no parecen influir en el diagnóstico de EPID. Los pacientes ambulatorios tuvieron un índice de complicaciones comparable a los casos que ingresaron, por lo cual este procedimiento podría ser incluido en un programa de CA. La BP-CVT es una herramienta fiable y segura para el diagnóstico de los pacientes con sospecha de EPID.
The term diffuse interstitial lung disease (ILD) refers to a heterogeneous group of disorders with several common characteristics in which the deep pulmonary tissue suffers from inflammation and scarring.1 Diffuse ILD is divided into two large groups: idiopathic and those with an identifiable cause, the latter representing only 30% of the total.2
The decision to perform surgical lung biopsy (SLB) in these patients is based on the probability that the histologic examination of the tissue obtained could provide specific information about the cause of the parenchyma disease.3
The current diffuse ILD clinical guidelines advocate SLB as the optimal method for reaching a definitive diagnosis for the process. However, they also insist on the need to individualize and adequately weigh the risks and benefits of the surgical procedure.1,4–6
Therefore, in order to maximize the benefit of SLB, it is crucial to define the optimal number and location for the biopsy samples to improve the diagnostic performance of the surgery. Within this process of improving the risk/benefit ratio of surgery in diffuse ILD, the change from conventional thoracotomy to video-assisted thoracoscopic surgery (VATS) has favored SLB because, with the new technique, post-operative morbidity has been significantly improved and hospital stays have been consequently shortened.7 Thus, several groups have included VATS lung biopsy (VATS-LB) in outpatient surgery programs, although this policy still has not become generalized. The present study was done under the auspices of the Diffuse Interstitial Lung Disease Integrated Research Program of the Spanish Society of Pulmonology and Thoracic Surgery (PII-EPID of SEPAR).
Based on the data obtained prospectively in 13 centers distributed throughout Spain, our objective is to evaluate whether the location and the number of lung biopsies obtained by VATS influence the diagnosis of diffuse ILD. As a secondary objective, we studied the viability of an ambulatory surgery program (ASP) in these patients.
Patients and methodsThe data were collected prospectively from 1 January 2007 until 31 December 2009. A total of 13 Spanish Thoracic Surgery Departments participated in the study. Included in the study protocol were patients with clinical and radiological suspicion of diffuse ILD who were candidates for VATS-LB. Excluded from the protocol were those patients with a solitary lung nodule or with other focal pulmonary processes, patients with mechanical ventilation and those with PaO2<60mmHg. Open lung biopsies were also excluded. All the cases had been previously studied with high-resolution computed tomography (HRCT). Likewise, in most of the centers bronchoscopy and lung function studies were carried out as part of the diffuse ILD pre-operative protocol. The lung parenchyma areas to be biopsied were chosen depending on the pre-operative findings of the HRCT. The areas for biopsy chosen had an area of parenchyma that was macroscopically pathological in appearance next to an area with a normal macroscopic appearance.
The data were compiled on standardized data collection forms that included the following variables: age, sex, most affected areas on HRCT, anesthesia risk according to the ASA (American Society of Anesthesiologists); results of the bronchoscopy, bronchoalveolar lavage (BAL), bronchial aspirate (BA), transbronchial biopsy, lung function tests and arterial gas studies; VATS characteristics, including number of entry ports, biopsy location, endo-staples, whether the patient was included in an ambulatory surgery program, time until pleural drain was withdrawn, time until discharge, intra- and extra-hospital morbidity, perioperative mortality and histologic diagnosis of the samples. The “diagnostic efficacy” and “no diagnostic efficacy” groups were created according to the histologic diagnosis obtained with the samples of the surgical lung biopsy (specific diagnosis–specific entity/non-specific). Keeping in mind the criterion that the area for biopsy should include an area of parenchyma that is macroscopically pathologic in appearance together with an area that is macroscopically normal in appearance, each of the 13 participating departments opted for one or more biopsies according to their standard practice (two systematic biopsies vs the possibility of one single biopsy including healthy and diseases areas within the same surgical piece). The pathologists of each center analyzed the multiple biopsies together. The criteria for ambulatory surgery were determined based on the experience and standard practice of each participating department. The data collection sheets were sent to the coordinator for analysis. The pertinent statistical studies were carried out with the SAS program for Windows, version 8. The continuous variables were expressed as means and the categorical variables as percentages. The comparisons were done with the Wilcoxon and chi-squared statistical tests, depending on the case. In all the cases, P<.05 was considered statistically significant.
ResultsA total of 224 patients were included in the study. Mean age was 57.1 (range 25–77); there were 118 women (52.6%) and 106 men (47.4%). All the patients underwent pre-operative thoracic HRCT (100%).
The most affected areas on thoracic HRCT were the lower lobes (55%), upper lobes (30%) and diffuse affectation in 15%. The locations for the biopsies were the following: left upper lobe (33.3%), left lower lobe (28.8%), right lower lobe (13.3%), middle lobe (13.3%) and right upper lobe (11.1%).
The anesthesia risk according to the ASA was: I in 20% of cases, II in 51% and III in 29%.
Bronchoscopy was performed in 188 patients (84%), BA in 126 (56.2%), BAL in 147 (65.6%) and transbronchial biopsy in 110 (49.1%).
The results of the lung function tests were the following: mean FVC: 75%; FEV1: 76.1%; FEV1/FVC: 86%; TLC: 74%; RV: 81%; DLCO: 55.3% and KCO: 77.8%. Mean values from the arterial blood gas study were: PaO2: 73.5mmHg and PaCO2: 39mmHg.
Lung biopsy by VATS was carried out with 3 entry portals in 215 patients (96%) and through 2 portals in 9 patients (4%). The mean number of endo-staples used was 3.4 (range 1–6). There was no hospital mortality on this series of patients.
Seventy patients (31.3%) belonging to two of the centers were included in an ASP program and discharged the same day of the surgery. In these cases, the withdrawal of the thoracic drain was done immediately in the operating room in 18 cases (25.7%), in less than 1h in the recovery unit in 30 cases (42.8%) and between 1 and 6h after surgery in 22 cases (31.4%).
One hundred and fifty-four patients (68.7%) were hospitalized after the VATS-LB. The removal of the chest drain was done within <48h in 89.7% and the patients were discharged within <24h in 61.5%. In these hospitalized patients, morbidity was reported in 14 cases (9%): dyspnea was increased in 6, pain in 2, persistent air leak in 2, pneumothorax in 2 and high debit due to the thoracic drain in 2.
There were complications after the hospital discharge in 12 patients (5.4%), which was similar between hospitalized patients (9/154, 5.8%) and patients in the ambulatory surgery program (3/70, 4.3%).
In the ASP group, (3/70) these complications were pneumothorax (2) and pain (1). In the group of hospitalized patients (9/154): pain (69, pneumothorax (2) and hemothorax (1).
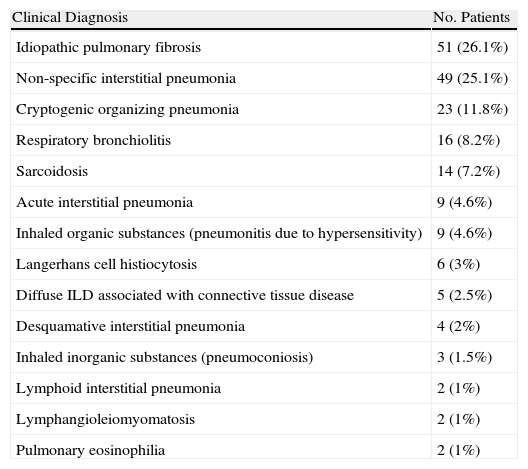
The most frequent clinical diagnosis was idiopathic lung fibrosis (26.1%) (Table 1).
Results of the Patients With Specific Diagnosis.
| Clinical Diagnosis | No. Patients |
| Idiopathic pulmonary fibrosis | 51 (26.1%) |
| Non-specific interstitial pneumonia | 49 (25.1%) |
| Cryptogenic organizing pneumonia | 23 (11.8%) |
| Respiratory bronchiolitis | 16 (8.2%) |
| Sarcoidosis | 14 (7.2%) |
| Acute interstitial pneumonia | 9 (4.6%) |
| Inhaled organic substances (pneumonitis due to hypersensitivity) | 9 (4.6%) |
| Langerhans cell histiocytosis | 6 (3%) |
| Diffuse ILD associated with connective tissue disease | 5 (2.5%) |
| Desquamative interstitial pneumonia | 4 (2%) |
| Inhaled inorganic substances (pneumoconiosis) | 3 (1.5%) |
| Lymphoid interstitial pneumonia | 2 (1%) |
| Lymphangioleiomyomatosis | 2 (1%) |
| Pulmonary eosinophilia | 2 (1%) |
As has been commented, the areas of lung parenchyma to be biopsied were chosen depending on the pre-op HRCT findings. It was necessary for the biopsy area to include an area of parenchyma that was macroscopically pathologic in appearance together with an area that was macroscopically normal. Following said criteria, in 179 cases (79.9%) we performed 2 biopsies and in 45 patients we were able to include both areas (pathologic and healthy) in one biopsy sample. In the patients with two biopsies, the rate of diagnostic agreement (same histologic diagnosis between samples) was 97.2% (174 cases), and in 2.8% (5 cases) it was different. In these 5 cases, the different diagnoses were: sarcoidosis/non-specific interstitial pneumonia in 4 and cryptogenic organizing pneumonia/idiopathic pulmonary fibrosis in 1.
In total, 195 of the 224 patients (87%) had a specific diagnosis (specific entity) while in 13% the diagnosis was not specific (anatomopathologic description of the tissue remitted).
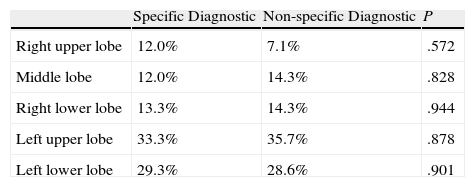
In order to evaluate whether the location of the VATS-LB influenced the diagnosis of diffuse ILD, we compared the diagnostic performance of the different areas biopsied according to whether a specific or non-specific diagnosis was reached. We did not find statistically significant differences (Table 2).
Location of the Pulmonary Biopsies According to Diagnostic Efficacy (Specific/Non-specific Diagnosis).
| Specific Diagnostic | Non-specific Diagnostic | P | |
| Right upper lobe | 12.0% | 7.1% | .572 |
| Middle lobe | 12.0% | 14.3% | .828 |
| Right lower lobe | 13.3% | 14.3% | .944 |
| Left upper lobe | 33.3% | 35.7% | .878 |
| Left lower lobe | 29.3% | 28.6% | .901 |
As for the number of biopsies, no differences were found here either: the specific diagnosis rates were similar between patients with more than one biopsy (155/179, 86.6%), and those with one single biopsy (40/45, 88.9%), P>.05.
To analyze the applicability of an ambulatory surgery program, we compared the ambulatory complication rate in patients who were hospitalized post-op (9/154, 5.8%) and patients included in ASP (discharged the same day as surgery: 3/70, 4.3%). No differences were found (P>.05).
With the intention of identifying any other independent factor that could influence the diagnostic performance of VATS-LB, we carried out a logistic regression analysis of the different variables collected in the study. This analysis found no significant factors.
DiscussionDiffuse interstitial lung disease encompasses a group of pathologies that have a great variability.8 Important progress has been made in its diagnosis thanks to radiological methods such as HRCT9 or minimally invasive methods like bronchoalveolar lavage.10 However, in those cases in which there is no clear environmental component or it is not associated with a systemic disease with lung affectation, it is still necessary to obtain pulmonary parenchyma samples by means of SLB in order to diagnose the process.11
Currently, the role of SLB in diffuse ILD is controversial. Despite the benefits obtained with the appearance of video-assisted thorascopic surgery for lung biopsy, many pulmonologists are reluctant to order surgery for their patients without guarantees that the results of the surgical biopsy will provide a specific diagnosis and therefore a significant change in the management of the patient.12 It is for this reason that the diagnostic efficacy of SLB should be as high as possible with a minimal morbidity.
In the present study with 13 participating Spanish centers, a specific diagnosis was reached in 87% of cases, with a morbidity of <10%.
The specific diagnosis rates published in the literature vary between 34% and 98%, depending on the series.13 The high effectiveness of VATS-LB in the present study is especially interesting, keeping in mind that most of the patients included in the protocol had undergone different diagnostic tests without reaching a definitive diagnosis (including transbronchial biopsy in 49.1% of cases). In this context, our data suggest that when transbronchial biopsy is not able to reach a diagnosis, VATS-LB may be a valid option.14
With regards to morbidity, minor complications appeared during hospitalization in 9% and at the ambulatory level in 5.4%. No peri-operative mortality was reported. As happens with the diagnostic performance, the rates of morbidity and mortality of SLB present great variability.15 This disagreement between studies reflects the different types of patients that are included in the diffuse ILD surgical series. In the present study, the patients included had to be able to tolerate selective ventilation for VTC and those patients with mechanical ventilation or with a PaO2<60mmHg were excluded; this selection could be the reason for our low morbidity.
The most common diagnosis was idiopathic pulmonary fibrosis (IPF, 26.1%). The epidemiological studies about diffuse ILD cite IPF as the most common cause,16 with an estimated prevalence of 20/100 000 men and 13/100 000 women.17
The question regarding which is the best lung area for carrying out biopsy is quite controversial. Gaensler and Carrington11 postulated in 1980 that for an optimal performance of SLB, the lingula and the middle lobe should be avoided. Later, Wetstein18 and Miller et al.19 demonstrated that there were no reasons to avoid the lingula and the middle lobe. In fact, Morell et al.20 systematically performed SLB in their series in either the right middle lobe or the left lingula.
Our results, based on 224 patients, are similar to those published by Qureshi et al.21; there are no significant differences in the diagnostic performance between the different lung biopsy areas as long as the SLB had been directed by the thoracic HRCT results and included areas of healthy and pathologic parenchyma.
We completely coincide with Walker et al.22 in the concept that HRCT is the ideal pre-operative study for choosing the optimal location for SLB in diffuse ILD. The ground glass areas on chest CT are the ideal guide for directing biopsies. The criteria used in this paper were those suggested by the SEPAR guidelines1: the surgical sample(s) should include different areas, one with macroscopically pathologic appearance and another with macroscopically normal appearance.
Regarding the optimal number of biopsies, our results suggest that a single biopsy from an area with both pathological and normal parenchyma could be sufficient to obtain a diagnosis (179 cases of multiple biopsies with a diagnostic agreement of 97.2%). Chechani et al.23 obtained similar results with 100% agreement in their series of multiple biopsies. Nevertheless, it should be indicated that other authors have found lower agreement rates. Monaghan et al.24 analyzed 64 patients with multiple biopsies; they found agreement in the diagnosis of these multiple samples in 87.5% of patients. Flaherty et al.25 analyzed the biopsies of 168 patients, 109 of which with multiple biopsies, finding a diagnostic agreement in 74% of patients.
As a secondary objective, we analyzed the feasibility of an ambulatory surgery program for diffuse ILD patients undergoing VATS-LB. Out of all the alternatives to conventional hospitalization developed in recent decades, ASP has been the option that has grown the most.
The VATS-LB approach has become more and more accepted in the diagnosis of patients with diffuse ILD. To date, however, very little has been published about its inclusion in ambulatory programs.26
In our opinion, the key element that makes possible its inclusion is the real feasibility of early withdrawal of the thoracic drain, which has been shown to be a safe practice.27 Chang et al.28 published a series of 62 patients undergoing ambulatory VATS-LB. 72.5% of their patients were discharged within the first 8h post-op, 22.5% within the first 23h and only 5% required hospitalization. Satherley et al.29 compared 82 patients with thoracic drains and 93 without drains after VATS-LB. They found no significant differences either in the results or in the rate of complications. Molins et al.30 published their results in 32 patients who underwent VATS-LB included in an ASP. In this series, the majority of the patients were discharged within the first 4h of the intervention and only one patient required hospitalization due to an air leak observed before the removal of the chest drain.30
In the present study, 70 patients were included in an ASP, so the chest drain was removed immediately in the OR in 18 cases (25.7%), within an hour in the recovery unit in 30 (42.8%) and between 1 and 6h in 22 (31.4%). After hospital discharge, there were complications in 3 patients (3/70, 4.3%), similar to the complications in hospitalized patients. In our opinion, these results support the inclusion of this procedure in ambulatory surgery programs. This policy would be beneficial for the patient (as it would avoid hospitalization) and also for the hospital administration (an estimated in savings of hospital costs of 294.10 Euros per patient included in an ASP).30
The strengths of this study are its nation-wide, multi-center design (which minimizes the risk of selection bias) and the fact that it focuses only on those patients with diffuse ILD who are candidates for VATS-LB. We excluded those patients with acute respiratory distress or patients with lung nodules or masses and those with open SLB. The main limitations were that the histologic diagnosis was not standardized and depended on the pathologist at each of the 13 participating centers, and that the multiple biopsies were not analyzed blindly, but jointly instead.
In conclusion, the location and the number of lung biopsies do not seem to influence the diagnosis of diffuse ILD as long as they include a pathologic area and a normal area in the sample. The outpatients had a complication rate comparable to that of the hospitalized patients; therefore, this procedure could be included in an ambulatory surgery program. VATS-LB is a reliable and safe tool for the diagnosis of diffuse ILD. Its mortality rate is low and it provides a definitive diagnosis in most cases.
Conflict of InterestThe authors declare no conflicts of interest.
Please cite this article as: Fibla JJ, et al. Biopsia pulmonar por videotoracoscopia en el diagnóstico de la enfermedad pulmonar intersticial. Estudio prospectivo y multicéntrico en 224 pacientes. Arch Bronconeumol. 2012;48:81-5.
This project was carried out under the auspices of PII-SEPAR EPID (Diffuse Interstitial Lung Disease Integrated Research Project of the Spanish Society of Pulmonology and Thoracic Surgery).