The association between GOLD categorizations and future exacerbations has not been fully investigated. This study elucidates whether the GOLD 2017 classification is associated with different future exacerbation risk in patients with chronic obstructive pulmonary disease (COPD) compared with the previous GOLD categorization. Another objective was to investigate the impacts of the symptoms and FEV1 on the predicted future exacerbation independently of previous exacerbation history.
MethodsWe analyzed patients from three prospective COPD cohorts (SNUH, KOCOSS, and KOLD) and evaluated the risk of moderate to severe exacerbation among different models, including GOLD grade (FEV1), GOLD 2011, and GOLD 2017.
ResultsIn total, 611 COPD patients were included (36 from SNUH, 257 from KOCOSS, and 318 from KOLD). GOLD 2017 classification, excluding FEV1% for categorization criteria, showed no differences in future exacerbation risk compared with GOLD grade and GOLD 2011 based on c-statistics. Among those with no frequent exacerbation history and FEV1 ≥50%, the group with more symptoms was significantly associated with future exacerbations than the group with less symptoms. A lower FEV1 (FEV1 <50%) was not associated with a higher future exacerbation risk than a higher FEV1 (FEV1 ≥50%), regardless of prior exacerbation history and symptom group.
ConclusionThe GOLD 2017 classification was not different from GOLD grade and GOLD 2011 regarding the association with future exacerbation risk, and there were no significant differences in exacerbation risk according to FEV1%. This suggests that FEV1 might not be an important factor in future exacerbation risk. These results partly support the GOLD 2017 assessment tool.
La asociación entre la categorización GOLD y la aparición de futuras exacerbaciones no se ha investigado a fondo. Este estudio analiza si la clasificación GOLD 2017 se asocia a un riesgo de padecer exacerbaciones futuras en pacientes con enfermedad pulmonar obstructiva crónica (EPOC) diferente al asociado a categorizaciones GOLD previas. Otro de los objetivos fue investigar el impacto de los síntomas y del volumen espiratorio forzado en el primer segundo (FEV1) en la exacerbación futura, independientemente de la historia previa de exacerbaciones.
MétodosSe analizaron prospectivamente 3 cohortes de pacientes con EPOC (SNUH, KOCOSS y KOLD) y se evaluó el riesgo de exacerbación moderada y grave entre los diferentes modelos, incluyendo el grado GOLD (FEV1), GOLD 2011 y GOLD 2017.
ResultadosSe incluyeron un total de 611 pacientes con EPOC (36 de SNUH, 257 de KOCOSS y 318 de KOLD). La clasificación GOLD 2017 (excluyendo el porcentaje de FEV1 para el criterio de categorización) no mostró diferencias en el riesgo de exacerbación futura en comparación con el grado GOLD y GOLD 2011 según el estadístico C. Entre los pacientes sin historia previa de exacerbaciones y FEV1≥50%, aquellos con mayor número de síntomas presentaron una asociación significativamente mayor con la aparición de exacerbaciones futuras que el grupo con menor número de síntomas. Valores más bajos de FEV1 (FEV1<50%) no se asociaron con un mayor riesgo de exacerbación futura que valores más elevados de FEV1 (FEV1≥50%), independientemente de la historia previa de exacerbación y de los síntomas.
ConclusiónLa clasificación GOLD 2017 no resultó diferente al grado GOLD y a la clasificación GOLD 2011 respecto a la asociación con el riesgo de exacerbación futura. Tampoco se determinaron diferencias significativas en el riesgo de exacerbación de acuerdo con el porcentaje de FEV1. Esto sugiere que FEV1 podría no ser un factor importante para el riesgo de exacerbación futura. Estos resultados apoyan en parte el uso de la herramienta de evaluación GOLD 2017.
Chronic obstructive pulmonary disease (COPD) is characterized by persistent respiratory symptoms and airflow limitation. Patients with COPD experience exacerbations and disease progression that lead to increased mortality risk. The Global Initiative for Chronic Obstructive Lung Disease 2011 (GOLD 2011) guidelines introduced a multidimensional assessment tool to guide therapy by classifying COPD patients into four groups: A (less symptoms and low risk), B (more symptoms and low risk), C (less symptoms and high risk), and D (more symptoms and high risk).1
The factors used in COPD assessment include the symptoms, which are evaluated by the modified Medical Research Council (mMRC) grade,2 COPD Assessment Test (CAT), or St. George Respiratory Questionnaire (SGRQ) score3,4; forced expiratory volume in 1 second (FEV1),3,5,6 and previous exacerbation history.3,7 It is well known that each of these factors is associated with future risks, including exacerbation and mortality, although the ABCD categorization was not intended to predict outcomes. Among these factors, prior history of exacerbation is known to be the strongest predictor of future exacerbation.3 However, it has not been investigated whether symptoms or FEV1 aside from prior exacerbation history are more likely to be associated with future exacerbation risks. Furthermore, although the GOLD 2017 update excluded FEV1 for risk group assessment,8 it has not been fully investigated whether GOLD 2017 has a weaker or stronger relationship with future exacerbation than prior GOLD grade and GOLD 2011.
This study investigates whether the GOLD 2017 classification excluding FEV1 as a criterion is associated with different future exacerbation risk from the GOLD grade and GOLD 2011 categorizations. The study also compares the symptoms and FEV1 without the previous exacerbation history in terms of their associations with future exacerbations.
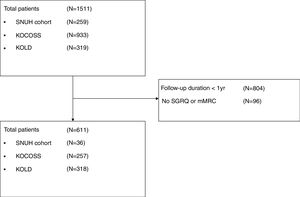
MethodsStudy populationPatients were recruited from three different prospective COPD cohort studies in South Korea: the Seoul National University Hospital (SNUH) cohort study (Airway registry, NCT02527486), the Korean COPD Subgroup Study (KOCOSS) (NCT02800499), and the Korean Obstructive Lung Disease Cohort (KOLD) (not registered because it was launched before 2005). The inclusion criteria were (1) age ≥40 years, (2) post-bronchodilator FEV1/forced vital capacity (FVC) <0.7, and (3) current or ex-smokers with a smoking history ≥10 pack-years. Patients with a history of asthma or who were not followed for at least one year were excluded. Informed consent was obtained from participants upon enrollment for each cohort. This study was approved by the Institutional Review Board of Seoul National University Hospital (IRB no. 1607-010-774).
Data collectionBaseline characteristics were collected at the baseline visit, including demographic factors, smoking status, symptoms scores (including mMRC dyspnea grade, CAT, and SGRQ score), comorbidities, exacerbation history in the past year, and pulmonary function tests (including post-bronchodilator spirometry). All patients underwent an evaluation of symptoms, exacerbation since the last visit, treatment history, and pulmonary function test (PFTs), including post-bronchodilator spirometry annually. Exacerbation history was assessed periodically by the patient's self-report according to cohort protocols (KOLD, SNUH cohort every 3 months; KOCOSS every 6 months). The medication possession ratios (MPRs) of the therapeutic drugs were calculated as the days during which each drug category was prescribed divided by the total number of days of follow-up.9 The included the inhaled corticosteroids, long-acting beta agonists (ICS/LABA), and long-acting muscarinic antagonists (LAMA).
Group classificationAccording to the GOLD guidelines, patients with COPD were classified into four subgroups (A–D) using symptom criteria based on the mMRC grade or CAT score. The risk criteria were based on FEV1 or exacerbation history. In addition, the SGRQ score was used as a surrogate for the CAT score with a cut-off score of ≥25.8 Acute exacerbation of COPD was defined as a worsening of symptoms beyond day-to-day variation with medication change. Exacerbation events that required outpatient clinic visits, systemic corticosteroid, or antibiotics but not admission were defined as moderate exacerbation. Acute exacerbation requiring an emergency room visit or hospitalization was defined as severe exacerbation.
We compared the risk of acute exacerbation among models made by various combinations of symptom (mMRC and SGRQ) and risk criteria (FEV1 and the history of prior exacerbation during the past year). The three models were based on the GOLD grade (grade 1: FEV1 ≥80%; grade 2: FEV1 50–79%; grade 3: FEV1 30–49%; grade 4: FEV1 <30%), GOLD 2011 guidelines,1 and GOLD 2017 guidelines.8 In addition, we categorized the participants into eight groups according to their previous exacerbation history, FEV1, and symptoms (supplement table 1).
Statistical analysisThe main outcomes were whether at least one moderate or severe acute exacerbation occurred during the first year and the time to the first moderate or severe acute exacerbation. Multivariable logistic regression models and Cox proportional hazard regression models were used to evaluate the one-year exacerbation risk and time to first exacerbation risk, respectively. The effect sizes for risk were expressed as odds ratios (ORs) with confidence intervals (CIs) for logistic models and as hazard ratios (HRs) with 95% CIs for Cox models. The models were adjusted by covariates including age, sex, body mass index (BMI), smoking status, medication use, and comorbidities.
The Akaike information criterion (AIC) and Bayesian information criterion (BIC) were used to evaluate the model fit. A lower AIC or BIC indicates a better model fit. c-Statistics (the area under the receiver operating characteristic curve (AUROC) for logistic models and Harrell's C for the Cox regression model) were used for discriminant ability. AUCs were compared between groups using the Stata command roccomp, and Harrell's C was compared using Somer's D.
Kaplan–Meier curves were drawn to reveal the time to first exacerbation between groups. The models were calibrated using the Hosmer–Lemeshow chi-squared test for logistics models and a graphical comparison of the observed and predicted survival probabilities for the Cox hazard regression models. All statistical analyses were performed using Stata 14.2 (StataCorp, College Station, TX, USA). A P-value <0.05 was considered to indicate statistical significance.
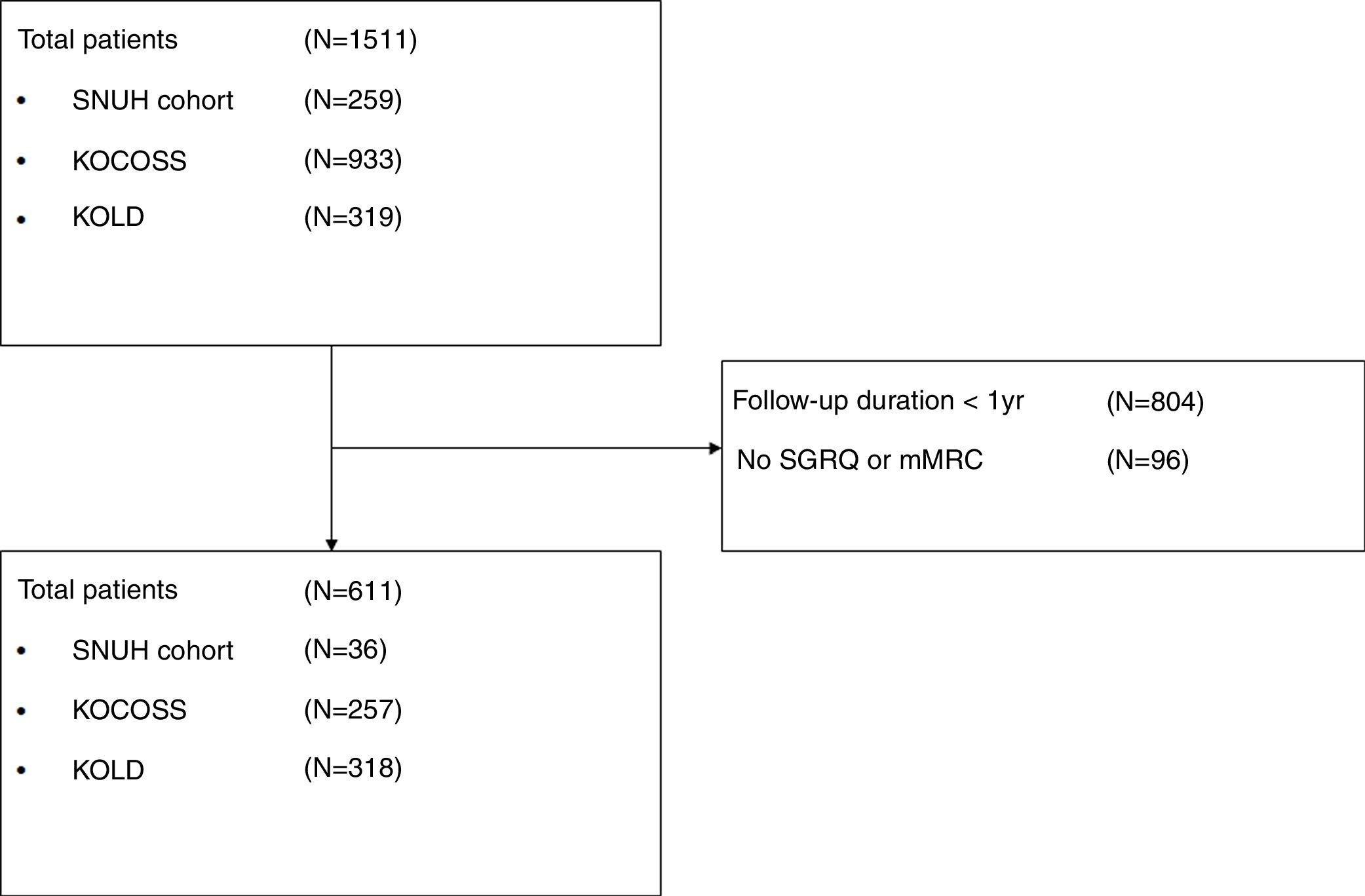
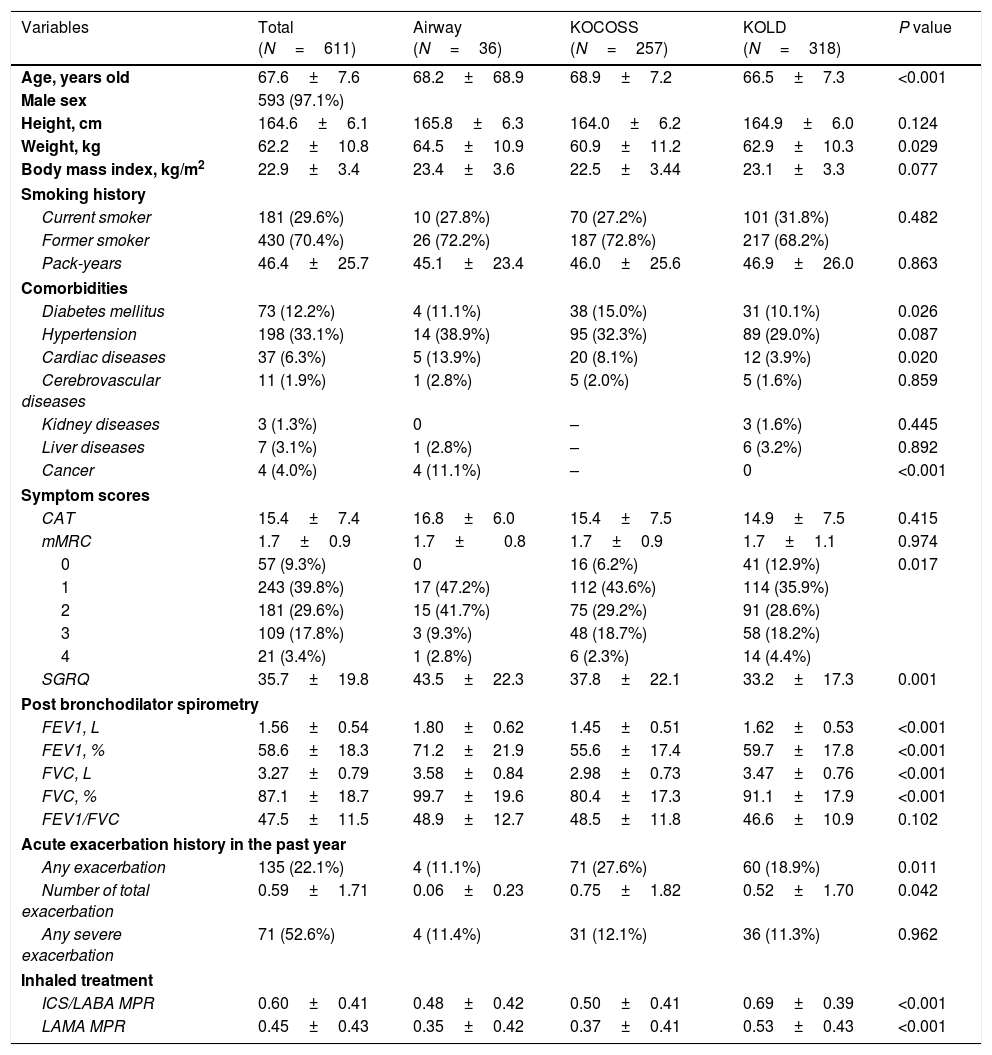
ResultsAmong 1511 patients who were enrolled from the three cohorts, 611 patients were included in the study (36 from SNUH, 257 from KOCOSS, and 318 from KOLD), as shown in Fig. 1. The baseline characteristics of patients are compared among the three cohorts in Table 1. The mean age was 67.6 years, and 593 patients (97.1%) were male. The average CAT and SGRQ scores were 15.4±7.4 and 35.7±19.8, respectively, and 311 (50.9%) patients had dyspnea with an mMRC grade ≥2. Of these, 22.1% had at least one exacerbation in the past year, and about half had experienced at least one severe exacerbation.
Baseline characteristics of participants.
| Variables | Total (N=611) | Airway (N=36) | KOCOSS (N=257) | KOLD (N=318) | P value |
|---|---|---|---|---|---|
| Age, years old | 67.6±7.6 | 68.2±68.9 | 68.9±7.2 | 66.5±7.3 | <0.001 |
| Male sex | 593 (97.1%) | ||||
| Height, cm | 164.6±6.1 | 165.8±6.3 | 164.0±6.2 | 164.9±6.0 | 0.124 |
| Weight, kg | 62.2±10.8 | 64.5±10.9 | 60.9±11.2 | 62.9±10.3 | 0.029 |
| Body mass index, kg/m2 | 22.9±3.4 | 23.4±3.6 | 22.5±3.44 | 23.1±3.3 | 0.077 |
| Smoking history | |||||
| Current smoker | 181 (29.6%) | 10 (27.8%) | 70 (27.2%) | 101 (31.8%) | 0.482 |
| Former smoker | 430 (70.4%) | 26 (72.2%) | 187 (72.8%) | 217 (68.2%) | |
| Pack-years | 46.4±25.7 | 45.1±23.4 | 46.0±25.6 | 46.9±26.0 | 0.863 |
| Comorbidities | |||||
| Diabetes mellitus | 73 (12.2%) | 4 (11.1%) | 38 (15.0%) | 31 (10.1%) | 0.026 |
| Hypertension | 198 (33.1%) | 14 (38.9%) | 95 (32.3%) | 89 (29.0%) | 0.087 |
| Cardiac diseases | 37 (6.3%) | 5 (13.9%) | 20 (8.1%) | 12 (3.9%) | 0.020 |
| Cerebrovascular diseases | 11 (1.9%) | 1 (2.8%) | 5 (2.0%) | 5 (1.6%) | 0.859 |
| Kidney diseases | 3 (1.3%) | 0 | – | 3 (1.6%) | 0.445 |
| Liver diseases | 7 (3.1%) | 1 (2.8%) | – | 6 (3.2%) | 0.892 |
| Cancer | 4 (4.0%) | 4 (11.1%) | – | 0 | <0.001 |
| Symptom scores | |||||
| CAT | 15.4±7.4 | 16.8±6.0 | 15.4±7.5 | 14.9±7.5 | 0.415 |
| mMRC | 1.7±0.9 | 1.7±0.8 | 1.7±0.9 | 1.7±1.1 | 0.974 |
| 0 | 57 (9.3%) | 0 | 16 (6.2%) | 41 (12.9%) | 0.017 |
| 1 | 243 (39.8%) | 17 (47.2%) | 112 (43.6%) | 114 (35.9%) | |
| 2 | 181 (29.6%) | 15 (41.7%) | 75 (29.2%) | 91 (28.6%) | |
| 3 | 109 (17.8%) | 3 (9.3%) | 48 (18.7%) | 58 (18.2%) | |
| 4 | 21 (3.4%) | 1 (2.8%) | 6 (2.3%) | 14 (4.4%) | |
| SGRQ | 35.7±19.8 | 43.5±22.3 | 37.8±22.1 | 33.2±17.3 | 0.001 |
| Post bronchodilator spirometry | |||||
| FEV1, L | 1.56±0.54 | 1.80±0.62 | 1.45±0.51 | 1.62±0.53 | <0.001 |
| FEV1, % | 58.6±18.3 | 71.2±21.9 | 55.6±17.4 | 59.7±17.8 | <0.001 |
| FVC, L | 3.27±0.79 | 3.58±0.84 | 2.98±0.73 | 3.47±0.76 | <0.001 |
| FVC, % | 87.1±18.7 | 99.7±19.6 | 80.4±17.3 | 91.1±17.9 | <0.001 |
| FEV1/FVC | 47.5±11.5 | 48.9±12.7 | 48.5±11.8 | 46.6±10.9 | 0.102 |
| Acute exacerbation history in the past year | |||||
| Any exacerbation | 135 (22.1%) | 4 (11.1%) | 71 (27.6%) | 60 (18.9%) | 0.011 |
| Number of total exacerbation | 0.59±1.71 | 0.06±0.23 | 0.75±1.82 | 0.52±1.70 | 0.042 |
| Any severe exacerbation | 71 (52.6%) | 4 (11.4%) | 31 (12.1%) | 36 (11.3%) | 0.962 |
| Inhaled treatment | |||||
| ICS/LABA MPR | 0.60±0.41 | 0.48±0.42 | 0.50±0.41 | 0.69±0.39 | <0.001 |
| LAMA MPR | 0.45±0.43 | 0.35±0.42 | 0.37±0.41 | 0.53±0.43 | <0.001 |
CAT, COPD Assessment Test; mMRC, modified Medical Round Council; SGRQ, St. George Respiratory Questionnaire; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; ICS, inhaled corticosteroid; LABA, long-acting beta agonist; MPR, medication possession ratio; LAMA, long-acting muscarinic antagonist.
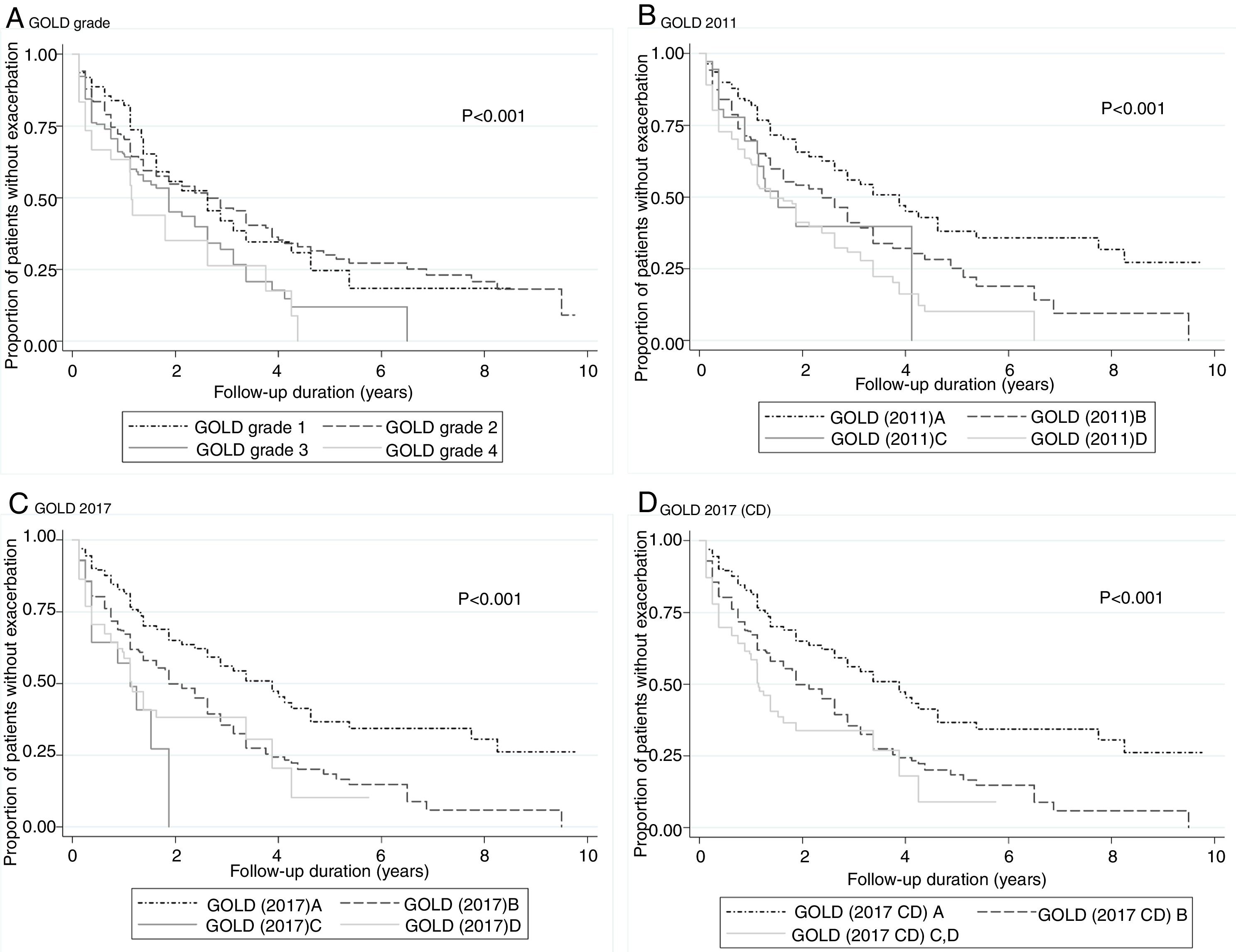
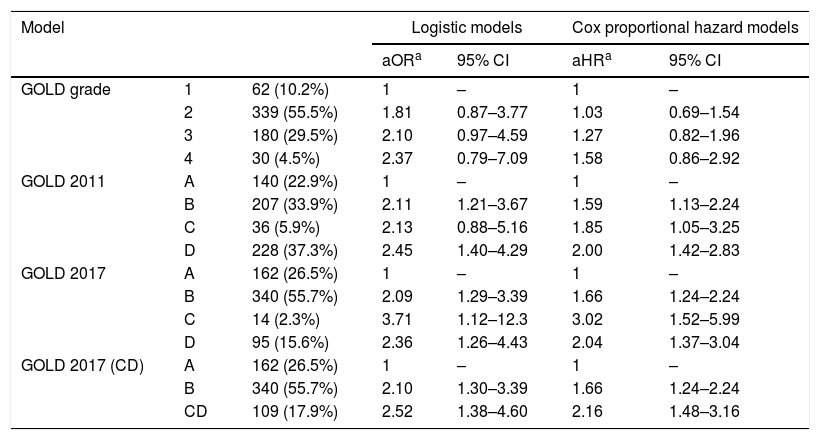
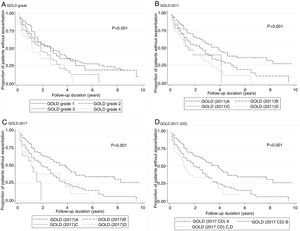
According to the GOLD 2017 classification, only 14 (2.3%) patients were categorized into group C. The proportion of patients classified into group D by GOLD 2017 was less than that by GOLD 2011 (15.6% vs. 37.3%; Table 2). The distribution of patients was significantly different in all comparisons between any two classification systems (P<0.001). The risks of moderate to severe exacerbation were not different among GOLD grades. Groups B–D had significantly higher exacerbation risk than group A according to GOLD 2011 and GOLD 2017 (except for group C by GOLD 2011 in the logistic model; adjusted odds ratio (aOR): 2.13; 95% CI: 0.88–5.16).
The risk of future exacerbation risk among groups according to different classification systems.
| Model | Logistic models | Cox proportional hazard models | ||||
|---|---|---|---|---|---|---|
| aORa | 95% CI | aHRa | 95% CI | |||
| GOLD grade | 1 | 62 (10.2%) | 1 | – | 1 | – |
| 2 | 339 (55.5%) | 1.81 | 0.87–3.77 | 1.03 | 0.69–1.54 | |
| 3 | 180 (29.5%) | 2.10 | 0.97–4.59 | 1.27 | 0.82–1.96 | |
| 4 | 30 (4.5%) | 2.37 | 0.79–7.09 | 1.58 | 0.86–2.92 | |
| GOLD 2011 | A | 140 (22.9%) | 1 | – | 1 | – |
| B | 207 (33.9%) | 2.11 | 1.21–3.67 | 1.59 | 1.13–2.24 | |
| C | 36 (5.9%) | 2.13 | 0.88–5.16 | 1.85 | 1.05–3.25 | |
| D | 228 (37.3%) | 2.45 | 1.40–4.29 | 2.00 | 1.42–2.83 | |
| GOLD 2017 | A | 162 (26.5%) | 1 | – | 1 | – |
| B | 340 (55.7%) | 2.09 | 1.29–3.39 | 1.66 | 1.24–2.24 | |
| C | 14 (2.3%) | 3.71 | 1.12–12.3 | 3.02 | 1.52–5.99 | |
| D | 95 (15.6%) | 2.36 | 1.26–4.43 | 2.04 | 1.37–3.04 | |
| GOLD 2017 (CD) | A | 162 (26.5%) | 1 | – | 1 | – |
| B | 340 (55.7%) | 2.10 | 1.30–3.39 | 1.66 | 1.24–2.24 | |
| CD | 109 (17.9%) | 2.52 | 1.38–4.60 | 2.16 | 1.48–3.16 | |
OR (adjusted odds ratio) and aHR (adjusted hazard ratio): adjusted by age, sex, height, weight, smoking status, comorbidities including diabetes, hypertension, cardiac disease and cerebrovascular disease and medication possession ratio of respiratory medications (inhaled corticosteroid-long acting beta agonist and long-acting muscarinic antagonist).
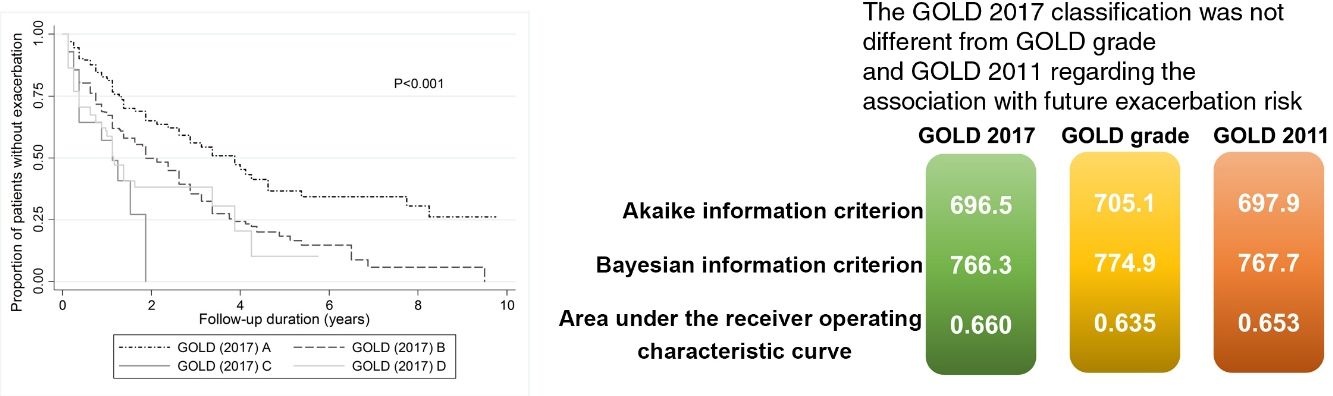
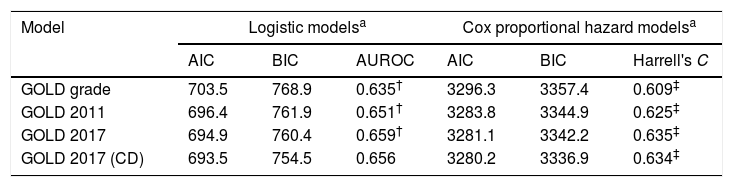
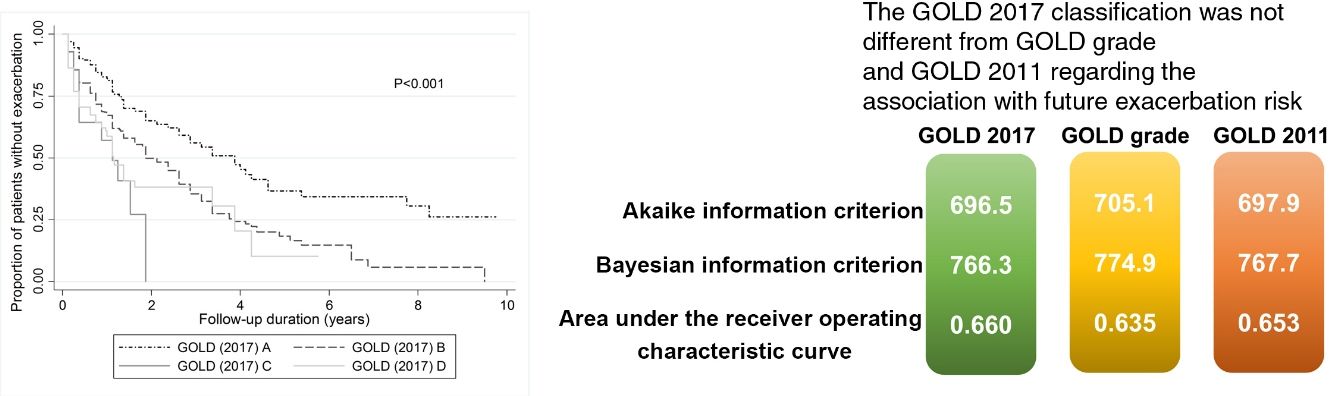
No model clearly showed better outcomes according to the order of groups A–D. According to the GOLD 2017 classification, group C had the highest exacerbation risk (aOR: 3.71; 95% CI: 1.12–12.3; adjusted hazard ratio (aHR): 3.02; 95% CI: 1.52–5.99). When groups C and D were combined into one category (group CD), the GOLD 2017 classification showed an increased risk of exacerbation in the order of A-B-CD (Table 2 and Fig. 2). The GOLD 2017 classification had lower AIC and BIC than the GOLD grade and GOLD 2011 classification. There were no statistically significant differences between GOLD 2017, GOLD 2011, and GOLD grade in c-statistics (AUROC: 0.635 vs. 0.651 vs. 0.659; P=0.492; Harrell's C, 0. 609 vs. 0.625 vs. 0.635 for GOLD grade, GOLD 2011 and GOLD 2017, respectively; P>0.05 for all comparisons; Table 3). The calibration of all models was fair (data not shown). We also made various other models by combining different symptom and risk criteria, which showed similar results (supplement tables 2 and 3).
Predicting ability for future exacerbation risk according to different classification systems.
| Model | Logistic modelsa | Cox proportional hazard modelsa | ||||
|---|---|---|---|---|---|---|
| AIC | BIC | AUROC | AIC | BIC | Harrell's C | |
| GOLD grade | 703.5 | 768.9 | 0.635† | 3296.3 | 3357.4 | 0.609‡ |
| GOLD 2011 | 696.4 | 761.9 | 0.651† | 3283.8 | 3344.9 | 0.625‡ |
| GOLD 2017 | 694.9 | 760.4 | 0.659† | 3281.1 | 3342.2 | 0.635‡ |
| GOLD 2017 (CD) | 693.5 | 754.5 | 0.656 | 3280.2 | 3336.9 | 0.634‡ |
AIC, Akaike information criteria; BIC, Bayesian information criteria; AUROC, area under receiver operating curve; mMRC, modified Medical Round Council; SGRQ, St. George Respiratory Questionnaire.
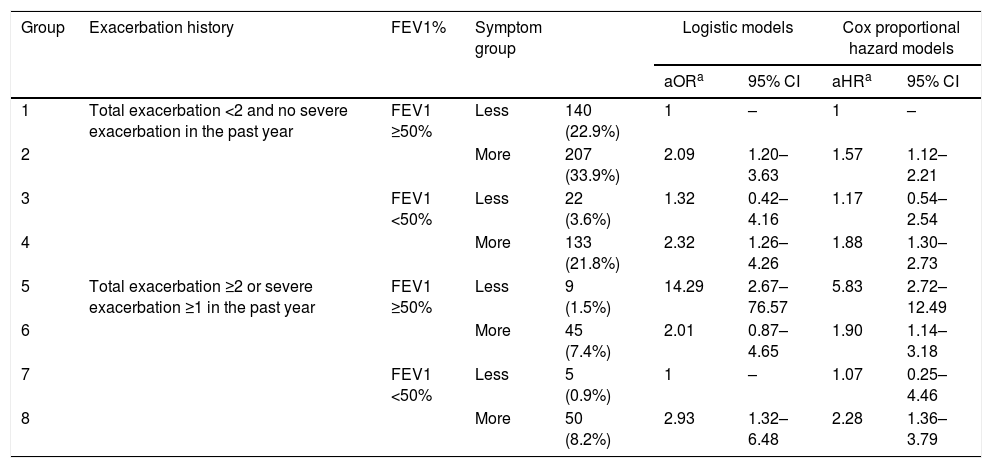
We also investigated the risk of exacerbation among 8 groups (Table 4) categorized by their previous exacerbation history, FEV1, and their symptoms. All models were adjusted by various covariates. Among those with a total exacerbation level <2 and no severe exacerbation in the past year (groups 1–4), the group with more symptoms had significantly higher future exacerbation risk than the group with less symptoms among those with FEV1 ≥50% (group 2 vs. group 1; P=0.009). We did not find a significantly higher exacerbation risk in the more-symptom group than in the less-symptom group in other comparisons (group 4 vs. group 3; group 6 vs. group 5; group 8 vs. group 7). A lower FEV1 (FEV1 <50%) was not associated with a higher future exacerbation risk than a higher FEV1 (FEV1 ≥50%), regardless of prior exacerbation history and symptom group (group 3 vs. group 1; group 4 vs. group 2; group 7 vs. group 5; group 8 vs. group 6). There were few participants in groups 3 (n=22), 5 (n=9), and 7 (n=5).
The risk of future exacerbation risk among groups categorized by previous exacerbation history, FEV1 and symptom.
| Group | Exacerbation history | FEV1% | Symptom group | Logistic models | Cox proportional hazard models | |||
|---|---|---|---|---|---|---|---|---|
| aORa | 95% CI | aHRa | 95% CI | |||||
| 1 | Total exacerbation <2 and no severe exacerbation in the past year | FEV1 ≥50% | Less | 140 (22.9%) | 1 | – | 1 | – |
| 2 | More | 207 (33.9%) | 2.09 | 1.20–3.63 | 1.57 | 1.12–2.21 | ||
| 3 | FEV1 <50% | Less | 22 (3.6%) | 1.32 | 0.42–4.16 | 1.17 | 0.54–2.54 | |
| 4 | More | 133 (21.8%) | 2.32 | 1.26–4.26 | 1.88 | 1.30–2.73 | ||
| 5 | Total exacerbation ≥2 or severe exacerbation ≥1 in the past year | FEV1 ≥50% | Less | 9 (1.5%) | 14.29 | 2.67–76.57 | 5.83 | 2.72–12.49 |
| 6 | More | 45 (7.4%) | 2.01 | 0.87–4.65 | 1.90 | 1.14–3.18 | ||
| 7 | FEV1 <50% | Less | 5 (0.9%) | 1 | – | 1.07 | 0.25–4.46 | |
| 8 | More | 50 (8.2%) | 2.93 | 1.32–6.48 | 2.28 | 1.36–3.79 | ||
OR (adjusted odds ratio) and aHR (adjusted hazard ratio): adjusted by age, sex, height, weight, smoking status, comorbidities including diabetes, hypertension, cardiac disease and cerebrovascular disease and medication possession ratio of respiratory medications (inhaled corticosteroid-long acting beta agonist and long-acting muscarinic antagonist).
**P<0.05 in Logistic models: group 1 vs. group 2, group 5 vs. group 6 and Cox models: group 1 vs. group 2, group 5 vs. group 6.
In our study, there were no differences in future exacerbation risks based on c-statistics between GOLD grade, GOLD 2011, and GOLD 2017 categorizations, although GOLD 2017 showed lower AIC and BIC. More symptoms were significantly associated with future exacerbation risks among those with no frequent exacerbation history and FEV1 ≥50%, but FEV1 <50% did not show an increased association with future exacerbations as compared to FEV1 ≥50%, regardless of prior exacerbation history and symptom categories. All statistical models were adjusted by various covariates, including demographic characteristics, smoking history, comorbidities, and treatment during the follow-up period.
The strength of this study is that we compared various classification systems, which were different in risk assessment: airflow limitation alone (GOLD stage), clinical presentation such as exacerbation history (GOLD 2017), and both of these (GOLD 2011). GOLD 2011 implemented a comprehensive assessment tool in order to guide therapy based on patient symptoms and risk of a future adverse event.1 However, the GOLD 2011 assessment tool applied heterogeneous grouping, which leads to mixed clinical outcomes within groups C and D.10,11
The COPDGene study showed heterogeneous exacerbation rates depending on whether the patients were categorized as group D according to lung function alone (0.89exacerbations/person-year), exacerbation history alone (1.34exacerbations/person-year), or both (1.86exacerbations/person-year). The patients categorized into high-risk groups based on FEV1 only (C1 or D1) showed a similar exacerbation risk to group B patients.12 Our study showed that he GOLD 2017 assessment tool without lung function shows no less association with future exacerbation risk compared with previous GOLD classifications. In addition, our study also suggested that categorization according to FEV1% is not associated with future exacerbation risk when considering prior exacerbation history and symptoms. The recently developed B-AE-D index includes exacerbation history but not lung function.13 Several published COPD guidelines also classify patients without considering lung function.14,15
It is well known that the history of previous exacerbation is the strongest predictor of the future exacerbation risk.3 However, it has not been clarified whether symptoms or FEV1 is more related to future exacerbation risk, aside from previous exacerbation risks. In our study, symptoms were significantly associated with future exacerbation, at least in patients with no previous frequent exacerbation history and FEV1 ≥50%. The more-symptom group showed a numerically higher risk of exacerbation in those with FEV1 <50%, but statistical significance was not detected, which could have been due to the small number of participants in some of the groups. However, FEV1 was not associated with exacerbation risk in all comparisons.
The influence on future exacerbation risk of the GOLD 2017 classification in which FEV1 was not considered in the categorization of patients was not less than that of the GOLD grade and GOLD 2011. This also suggests that FEV1 might not be an important predictor for future exacerbation risks. In addition, it might partly explain why GOLD group B paradoxically showed significantly poorer survival than group C in some cohort studies.16
In our study, the percentage of group C patients according to GOLD 2017 was only 2.3%, which was anticipated considering that one of the problems in the GOLD 2011 categorization is that only a small number of patients are classified into group C.5,17,18 The number of patients categorized into group C by the GOLD 2017 classification should be less than that by the GOLD 2011 classification because patients in group C according to GOLD 2011 with FEV1 <50% and <2 exacerbations in the past year were moved to group A. When groups C and D were combined into one category (group CD), GOLD 2017 showed an increased effect size of risks for exacerbation corresponding with the order of A-B-CD groups. Notably, there are already guidelines that use a classification similar to the A-B-CD system.19
This study has several limitations, including a short follow-up period of 1 year and a relatively small number of enrolled patients. Because these cohorts are still actively recruiting patients, many participants were followed up for less than 1 year at the date of our analysis. Excluding participants with a follow-up shorter than 1 year might lead to selection bias. However, international guidelines recommend that the study duration should be at least 1 year if the objective of the study is to investigate exacerbations.20 Thus, we included participants who were followed-up for at least 1 year. Further follow-up of these patients is needed to evaluate long-term outcomes, including exacerbations and mortality.
The generalizability of our study might be limited because it included only Korean patients from a predominantly male cohort. The possibility of including relatively mild patients (grade 4 represented only 4.5%) could be another limitation. Additionally, we did not consider other potential factors that might be related to exacerbations, such as COPD phenotypes (i.e., chronic bronchitis21), comorbid bronchiectasis,22,23 and exercise capacity24,25 because our study focused on the comparisons of items used in the GOLD classification. Instead, models were compared after adjusting for age, sex, body mass index, smoking status, medication use, and comorbidities.
Furthermore, most participants in our cohort were male (97.1%), which might affect the results because sex differences in the clinical course of COPD have been suggested.26 It should be stressed that patient characteristics lead to changes over time in the group to which patients belong, including symptoms, lung function, and exacerbation experience of those with COPD. However, we used only baseline characteristics. We applied SGRQ as symptom criteria, which is not a recommendation of the GOLD statement, as no participants underwent CAT or CCQ at baseline in one cohort that was initiated in 2005. However, GOLD states a symptom score equivalent to SGRQ ≥25 should be used as a threshold for considering regular treatment for symptoms.27 Finally, although GOLD assessment should also consider lung function severity, symptoms, and comorbidities in order to guide therapy, our analysis only focused on exacerbation. In addition, it should be considered that an association between the GOLD categorization and future outcomes might not only depend on the predictability of classifications but also on the adherence to the therapy according to the GOLD statement classification.
In conclusion, the GOLD 2017 classification in which FEV1 was not considered in the categorization of patients was not different from GOLD grade and GOLD 2011 in terms of the association with future exacerbation risk. FEV1 might be a less important factor for predicting exacerbation risk than prior exacerbation history and symptoms. These results partly support the GOLD 2017 assessment tool, although the size of group C was very small.
Conflict of interestsThe authors declare no conflict of interest.
This study was supported by a grant of the Korea Healthcare Technology R&D Project, Ministry for Health and Welfare, Republic of Korea (HI10C2020).