Despite the fact that the recommendations of the main clinical guidelines have restricted the indications for inhaled corticosteroids in chronic obstructive pulmonary disease (COPD), currently more than 80% of patients are receiving this treatment in Spain, mostly with high doses. A detailed review of the literature does not justify the use of these high doses, a position that agrees with the recommendations of the FDA. A re-evaluation of their safety, the consistency of the data on their efficacy showing similar results with moderate doses and a better patient selection require the use of this treatment in COPD patients to be reconsidered.
A pesar de que las recomendaciones de las principales guías clínicas han restringido las indicaciones de los corticoides inhalados en la enfermedad pulmonar obstructiva crónica (EPOC), actualmente más del 80% de los pacientes están recibiendo este tratamiento en España, en su mayoría con dosis elevadas. Una revisión detallada de la literatura no justifica el uso de estas dosis elevadas, posicionamiento que está de acuerdo con las recomendaciones de la FDA. Una revaloración de su seguridad, la consistencia de los datos de eficacia, que muestran resultados similares con dosis moderadas, y una mejor selección de los pacientes exige un replanteamiento del uso de este tratamiento en el paciente con EPOC.
In the 1990s, most studies about inhaled corticosteroids (IC) presented negative results in patients with chronic obstructive pulmonary disease (COPD). Several clinical assays confirmed that IC did not modify lung function loss and had only a mild impact on symptoms.1–3 These findings upheld the concept that COPD is a corticosteroid-resistant disease, and the general opinion when recommending them for treatment was negative. By the year 2000, however, although their use was not included in the main clinical guidelines, 54% of patients with mild COPD and 63.4% of severe patients seen in pulmonology departments in Spain were receiving IC for their disease.4
The publication of the ISOLDE study in 2000 brought about several relevant changes5: (a) it proposed for the first time that we could improve the state of COPD patients with anti-inflammatory drugs, even if they did not present asthmatic characteristics; (b) important aspects of the treatment were evaluated other than improvement in FEV1 (exacerbations, quality of life, etc.); (c) IC could favorably influence the clinical course of patients, even if the accelerated loss in lung function was not modified. At that time, the inflammation seen in COPD was considered to be relatively resistant to IC, so high doses were therefore contemplated. The doses used in the ISOLDE study (500mcg of fluticasone every 12h) have continued to be used throughout the last decade as standard COPD treatment for patients in Europe.
Despite the restrictive recommendations of the main clinical guidelines published in 2001,6,7 by 2005 71% of patients were receiving IC,8 mostly with high doses of fluticasone (500mcg every 12h). In 2008, the percentage of patients treated with IC in Spain surpassed 80%.9 The use of high IC doses is largely based on studies that found positive results for exacerbations and quality of life. Furthermore, expectations have also been generated in the last decade regarding the modification in cardiovascular risk and a possible reduction in mortality. These expectations are based on observational studies with important methodological limitations that have not been later confirmed in controlled studies.10,11 These expectations were amplified by an attractive hypothesis that associated systemic and pulmonary inflammation, a hypothesis that also has not been confirmed.12,13
The objective of this review is to analyze the scientific evidence which is the basis for the current use of high doses of IC in COPD, and to establish whether this treatment should be re-evaluated in our therapeutic strategy. We will contemplate high doses with 500mcg of fluticasone every 12h, moderate doses with 250mcg of fluticasone or 320mcg of budesonide every 12h, and low doses that are below these values. This classification is valid for other IC when adjusted to equipotent doses.
Data Sheets or the Best Available Evidence?The doses given on pharmaceutical product data sheets are not always the best doses for patients. The recommendations found on data sheets are based on studies specifically done to register a drug, which regulating agencies later take as a reference in order to establish the indication in its reference area. The history of a drug, however, does not end with the registration. Usually, either during or after the registration period, information is still compiled and it may recommend reconsidering certain indications. Several administrative aspects and the lack of coordination in previous years between the two main regulating agencies (EMEA and FDA) have made it difficult to incorporate this new information along with recommendations on the data sheet. This work method, which is not very coordinated between the regulating agencies, sometimes generates situations that are not always easy for clinicians to understand.14
In the United States, the approved dose of salmeterol-fluticasone for the treatment of COPD is 50/250mcg every 12h, based initially on 2 6-month studies (SFCA 3006 and SFCA 3007) that were basically done in patients with chronic bronchitis.15 Out of the patients included, 54% had positive bronchodilator tests (>12% and 200ml). In these studies, the 50/500mcg doses did not provide additional advantages, so the FDA did not recommend their use in COPD patients. Later studies with 250 and 500mcg of fluticasone (SCO 30003, SFCB 3024, SCO 40043 and SCO 100250)10,15–17 extended the indication to patients with chronic bronchitis and emphysema, as both groups were included in these studies. Once again, the results were conclusive, so that the 50/500mcg dose did not provide any additional benefit over 50/250mcg, either in pulmonary function or in exacerbations. The current situation is that in the United States the use of 500mcg of fluticasone is not authorized for the treatment of COPD, based on comparative studies with 250mcg.
In 2009, the FDA also approved budesonide-formoterol 160/4.5mcg every 12h for treating obstruction, both in patients with a predominance of chronic bronchitis as well as emphysema. This approval was based on the results of two pivotal studies with 6 months18 and 12 months19 of follow-up. In both, the effectiveness of budesonide-formoterol was evaluated in the maintenance treatment of airflow obstruction, including a total of 3668 COPD patients. Mean pre-bronchodilator FEV1 at the start of the study was 34% predicted. The patients who received budesonide-formoterol 320/9mcg twice a day had better results than the other treatment groups in the two main variables (pre-dose and post-dose FEV1), therefore it is the dose approved by the FDA. Nevertheless, a global view of lung function, exacerbations and quality of life showed no relevant advantages over 160/9mcg.
In Europe, the authorized and recommended dose of fluticasone for the treatment of COPD is 500mcg every 12h because the registration studies were done with this dose. No comparative studies were done, however, to evaluate the advantages and disadvantages over more moderate doses, such as 250mcg. Also in Europe, the EMEA has authorized formoterol associated with 320mcg of budesonide for the treatment of COPD. This dose, in the moderate range, is equivalent to approximately half of 500mcg of fluticasone. Unlike the position of the FDA, which only accepts moderate doses both for fluticasone as well as for budesonide, the EMEA accepts, with evident clinical incongruity, moderate doses for budesonide and high doses for fluticasone, ignoring the studies with 250mcg of fluticasone evaluated by the FDA. Compared with the transparent policy of the FDA, the EMEA hardly provides any information about the criteria on which these decisions are based; therefore, it is not always possible to correctly evaluate them, especially in a first phase in which not all the data are published. Fortunately, this is not the case of IC, and clinicians have sufficient information to decide whether it is correct for the decision of this agency to maintain high doses of fluticasone as a standard treatment in COPD patients.15,20
Who to Treat With Inhaled Corticosteroids?The most widespread message is that IC, associated with long-acting β2-adrenergic agonists (LABA), has beneficial effects on respiratory symptoms, quality of life and exacerbations, especially in patients with moderate and severe COPD. Their current position in patients with a certain FEV1 value assumes that all the patients with COPD are similar if adjusted for FEV1 values. Fortunately this is a concept that has been overcome. Even in the ISOLDE study, when the patients were selected by the FEV1 value, a significant percentage with values above 50% also had a history of frequent exacerbations, a finding recently confirmed in the ECLIPSE study.21 Currently, a history of frequent exacerbations is the main criteria for establishing the indication for IC in COPD. Do these patients not deserve a treatment that improves their symptoms or reduces their exacerbations simply because their FEV1 is above 50%? It is evident that FEV1 helps us have a general vision of a population, but it is not a valid criterion for establishing this treatment in clinical practice in a particular patient.
COPD is a heterogeneous disorder with different clinical profiles that are frequently associated with different pathogenic substrates.22,23 From a theoretical standpoint, patients who can most benefit from treatment with IC are those who present some type of common characteristic with asthma,24,25 meaning that they present a relevant reversibility (not only a bronchodilator test with an increase in FEV1 of 12% or 200ml), eosinophilia in sputum or high concentrations of nitric oxide in exhaled air.26 Studies with roflumilast have shown us that, when there is a favorable effect of a drug in a certain population, if we add patients in whom the drug is not effective, it is also possible to observe an overall beneficial effect. The result will be somewhat poorer, but in many cases it is possible to reach statistical significance by increasing the size of the sample. The result, although less striking, can extend the indication to larger populations, with a very relevant commercial benefit. Given that the effect is diluted by mixing different patients, the argument of resistance comes into play and the usual response is to increase the dose. If we analyze the sum of studies 111 and 112 with roflumilast, it is possible to clearly see this effect.27,28
These studies also demonstrate that the solution is not to increase the dose but to identify the population that could most benefit from the treatment, although this, from a commercial standpoint, would mean a partial loss of the market, and we would not be witness to so-called “blockbusters” as in previous years. In these cases, effectiveness does not improve by increasing the dose but by identifying the patient who will benefit the most from the treatment. The result is that IC will be administered to those patients who could really benefit from the treatment and it would not be necessary to give inappropriate doses in order to overcome a theoretical resistance, which may just be the statistical effect of mixing populations, in some of whom the risk/benefit balance may not be favorable.29
Recent studies indicate that the approach with roflumilast may also be valid for IC. Therefore, although this review assesses the use of high doses of IC (500mcg of fluticasone twice a day) versus moderate doses (250mcg of fluticasone or 320mcg of budesonide twice a day, a better evaluation of the patients may possibly support the use of even lower doses in certain populations, with no loss in efficacy.19,30 On the other hand, the data that are currently available indicate that if we refer to resistance in basic terms, this resistance occurs in any stage of the disease,31 and the selection by FEV1 values therefore does not seem justified.
What Is the Best Dose in Terms of Efficacy?Identifying the best possible dose in patients susceptible to improving with IC should be a priority line of research in COPD as the safety profile is largely related with the dose used. What data do we currently have available in order to optimize this treatment in our patients?
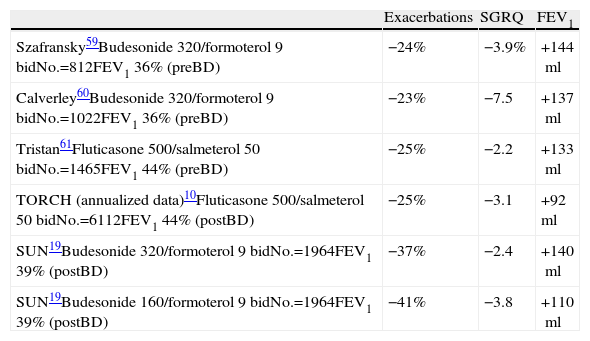
Sin et al.32 analyzed the impact of several doses of IC on mortality due to COPD with the data obtained upon discharge after hospitalization. These authors observed that the patients with moderate or high doses presented a reduction of 25% in the risk for all-cause mortality, and this risk was less than when lower doses were prescribed. Apparently, this study supports the use of high doses; it does, however, present important limitations, as it did not differentiate between moderate and high doses and databases were used with no spirometric confirmation of the diagnosis. Moreover, the favorable effect on mortality described in observational studies such as that by Sin have not been confirmed in controlled studies. In fact, in the TORCH study, doses of 500mcg of fluticasone every 12h were associated with an increase (not significant) in mortality of 6%. The data of this study advise against the use of IC as a monotherapy. Fortunately, there is also sufficient information about the efficacy of IC associated with bronchodilators. Consistently, doses of 500mcg of fluticasone every 12h have not provided any type of advantage in terms of effectiveness (lung function, quality of life and exacerbations) versus lower doses, both in monotherapy compared with placebo (Table 1) as well as compared with LABA (Table 2).
Results of the Main Clinical Assays With IC Associated With LABA: Effectiveness in Exacerbations, Quality of Life and Pulmonary Function Compared With Placebo.
| Exacerbations | SGRQ | FEV1 | |
| Szafransky59Budesonide 320/formoterol 9 bidNo.=812FEV1 36% (preBD) | −24% | −3.9% | +144ml |
| Calverley60Budesonide 320/formoterol 9 bidNo.=1022FEV1 36% (preBD) | −23% | −7.5 | +137ml |
| Tristan61Fluticasone 500/salmeterol 50 bidNo.=1465FEV1 44% (preBD) | −25% | −2.2 | +133ml |
| TORCH (annualized data)10Fluticasone 500/salmeterol 50 bidNo.=6112FEV1 44% (postBD) | −25% | −3.1 | +92ml |
| SUN19Budesonide 320/formoterol 9 bidNo.=1964FEV1 39% (postBD) | −37% | −2.4 | +140ml |
| SUN19Budesonide 160/formoterol 9 bidNo.=1964FEV1 39% (postBD) | −41% | −3.8 | +110ml |
Bid: every 12h; postBD: post-bronchodilator; preBD: pre-bronchodilator; SGRQ: St. George's Respiratory questionnaire.
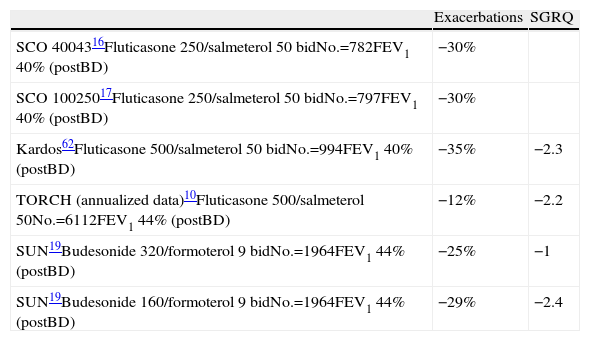
Results of the Main Clinical Assays With IC Associated With LABA: Effectiveness in Exacerbations and Quality of Life Compared With LABA.
| Exacerbations | SGRQ | |
| SCO 4004316Fluticasone 250/salmeterol 50 bidNo.=782FEV1 40% (postBD) | −30% | |
| SCO 10025017Fluticasone 250/salmeterol 50 bidNo.=797FEV1 40% (postBD) | −30% | |
| Kardos62Fluticasone 500/salmeterol 50 bidNo.=994FEV1 40% (postBD) | −35% | −2.3 |
| TORCH (annualized data)10Fluticasone 500/salmeterol 50No.=6112FEV1 44% (postBD) | −12% | −2.2 |
| SUN19Budesonide 320/formoterol 9 bidNo.=1964FEV1 44% (postBD) | −25% | −1 |
| SUN19Budesonide 160/formoterol 9 bidNo.=1964FEV1 44% (postBD) | −29% | −2.4 |
Bid: every 12h; postBD: post-bronchodilator; preBD: pre-bronchodilator; SGRQ: St. George's Respiratory Questionnaire.
Another argument that has positioned IC in COPD patients is their possible additional benefit on cardiovascular comorbidities.33,34 In a recent systematic review done by Loke et al.11 on 23 high-quality clinical assays, they conclude that IC do not favorably influence the reduction of myocardial infarction (RR=0.95; P=.68) or in death due to cardiovascular causes (RR=1.02; P=.89). As has happened with global all-cause mortality, these studies, with greater scientific rigor, have not confirmed the favorable results obtained from observational studies or post hoc analyses, which were so influential in positioning the use of IC in clinical practice. Currently, we do not only have data with a greater level of evidence about the absence of a favorable effect on cardiovascular risk or systemic inflammation, but also the concept itself of systemic inflammation in COPD should be reevaluated, given that only a small percentage of patients present consistently high inflammatory markers and that some of the markers that have mostly been associated with cardiovascular disease (C-reactive protein) can be related, not with pulmonary inflammation, but instead with collateral factors that are observed in some COPD patients (obesity, level of physical activity, etc.), which are unlikely to improve with high doses of IC.35,36
Is Safety Important When Using Inhaled Corticosteroids?IC can produce several local effects. A recent meta-analysis has confirmed that all the local side effects increase with higher doses.37 In addition, it is well-known that high doses of IC have systemic effects due to their absorption by the lungs.38 One of the main strategies for avoiding these adverse effects in the long-term is to use the lowest dose that achieves the best possible control of the patient. Is that what we are doing in COPD?
In Spain, the mean age of COPD patients seen in outpatient consultations or who are hospitalized is about 70.8,9,39 Senior patients are especially susceptible to the adverse effects of IC, although in order for their symptoms to be detected some may require prolonged periods of exposure to the drug, beyond the follow-up time of most clinical assays. The dose of 1000mcg of fluticasone per day has been associated with significant systemic effects, such as pneumonia, glaucoma, cataracts, diabetes, adrenal suppression and loss of bone density.40
Risk of FracturesIn a recent meta-analysis, the prolonged use of IC was associated with a significant increase in the risk of fractures, both in controlled studies as well as in observational studies. The risk increased 9% for each increase in dose equivalent to 500μg of beclomethasone.41 Although this dose-dependent risk of fractures is observed in older patients without COPD,42 in patients with COPD the risk is even greater as the disease itself is associated with osteoporosis, and smoking, reduced physical activity and nutritional aspects are additional risk factors.43,44 Correcting these risk factors and avoiding high doses of IC are the main measures for avoiding this complication.45
DiabetesThe use of IC is associated with an increased risk for developing diabetes and with the progression of the process when it is already established. This risk is more accentuated at higher doses. Although in most controlled assays in COPD there is no reported increased risk for diabetes associated with the use of IC, these studies lack statistical power to detect it. Recently, Suissa et al.46 carried out a population study of cohorts in order to evaluate the impact of the use of IC and their dose in the development of diabetes. It was also assessed whether their use conditions the start of insulin use in patients who had previously been diagnosed with type 2 diabetes. After adjusting for different co-variables, the use of IC was associated with a significant increase of 34% in the rate of diabetes (RR=1.34, 95%CI=1.29–1.39). There was also a confirmed clear relationship with the dose, and this effect was greater when doses equivalent to 1000mcg of fluticasone (RR=1.64; 95% CI=1.52–1.76) were used. The percentage was similar in the first prescription of insulin for the patients who were taking oral anti-diabetics (RR=1.34: 95%CI=1.17–1.53). As the authors comment, with an incidence of diabetes of 14.2/1000/year in their asthma-COPD cohort, the clinical assays published to date, including the TORCH study, lack statistical power to detect this effect.
CataractsSenior patients treated with IC are especially susceptible. In this population, even small doses of IC have been associated with a greater risk for cataracts. One study done in Quebec47 confirmed this side effect with the use of IC, but what is most striking is that although this effect is already observed with low doses, it increases considerably with higher doses, and this risk increases by 19% for every 1000mcg/day of beclomethasone or for an equipotent dose of another IC. These same authors observed that when patients sent for cataract surgery were evaluated, this risk was 3 times higher in those who had been treated with high doses of IC for more than 2 years. This association between high doses of IC and a higher risk for cataracts has been consistently confirmed in other series, especially when analyzing older populations and when evaluating prolonged exposure time to the medication, which is the characteristic profile of COPD patients.38,48–50
GlaucomaAlso in the sphere of ophthalmology, the higher risk for glaucoma is detected especially in those who use high doses of IC.51 Nevertheless, in a large cohort of senior patients, it was not confirmed that the use of high doses of IC increased the risk for glaucoma. However, this subject is still controversial, as the same authors previously published results to the contrary.52,53
Adrenal SuppressionThis is possibly one of the best markers of the systemic effects of IC, although their short-term clinical transcendence is not very important. The presence of low levels of endogenous cortisol is usually not clinically relevant, as it is compensated with the exogenous corticosteroid. After the administration of 250 and 500mcg of fluticasone twice a day, the serum levels of cortisol were 10% and 21% lower, respectively, than in the placebo group, indicating increased systemic effects at high doses. If we had to equate the effect of IC to the observed effects of systemic corticosteroids, doses of fluticasone at 0.44–1.76mcg/day cause similar effects on serum cortisol as does 5–20mg/day of prednisone. The calculated relative equivalence between fluticasone and prednisone would be 8.5:1. In this way, we can extrapolate that approximately 1000mcg of fluticasone would equal 8.5mg of prednisone per day in terms of systemic activity.54
Pneumonia and Safety After 3 Years in the TORCH StudyThe TORCH study (SCO 30003) observed an increase in pneumonia in the two groups treated with fluticasone. Apart from the multiple discussions that this finding has generated because radiological confirmation was not required, the results generated a warning about the safety and need for using high doses of IC. Later, the combined analysis of the SCO 40043 and SCO 100250 studies (50/250mcg of salmeterol/fluticasone versus 50mcg of salmeterol) also confirmed a greater risk for pneumonia (8.1% vs 4.3%) than in the group treated only with salmeterol. These results are similar to those observed in the TORCH study if adjusted for exposure time. A recent meta-analysis, however, did not observe a greater incidence of pneumonia with budesonide. Thus, when it comes to safety aspects, the dose is important but there may also be effects related with the type of molecule.55
Some messages coming from the TORCH study have transmitted that the use of high doses of IC is extremely safe. This interpretation, however, may not be true. Although it was not statistically demonstrated that the greater incidence of pneumonia was associated with increased mortality, the study was not designed to answer that question and it lacks the statistical power in order to draw conclusions. Evidently, pneumonia in a COPD patient generates a negative impact in his/her evolution, regardless of the fact whether he/she has a better or poorer evolution with the concomitant use of IC.56,57 In a recent study done in Spain, COPD was an independent risk factor in patients with community-acquired pneumonia.58
Moderate-to-severe COPD patients present several risk factors that predispose them toward developing osteoporosis. In the TORCH study, the patients with less bone mineral density (BMD) withdrew from the study earlier than those with normal BMD. Thus, the information that is provided in this study is not very assessable. Although an ophthalmologic test was done (cataracts and glaucoma) and urine cortisol was measured in a subgroup of patients, the amount of information available is limited. Given the high incidence of cataracts at the start of the study, valid conclusions cannot be drawn about safety with regards to the appearance of cataracts (it must be mentioned that before being included in the assay many patients were already receiving high doses of IC). Other possible complications were either not analyzed or the information was inadequate in order to reach any conclusions.46
ConclusionSome authors postulate that the fact that in most European countries more than 70% of COPD patients are being treated with high doses of IC, this is the result of certain marketing campaigns despite there being more and more consistent evidence that the current recommended high doses in Europe are not the most adequate.22 Official agencies such as EMEA promote the use of high doses, despite the fact that this same agency positions moderate levels of budesonide on the same level. This incongruent situation and the lack of coordination with other agencies like the FDA do not help to reduce the level of confusion. Aside from this posture, the available evidence does not demonstrate any additional benefit with the use of high doses and it is possible to demonstrate a greater incidence of side effect that are not easily evaluable by the clinician. These adverse effects can be especially severe when used for prolonged periods in senior patients with associated diseases, which are characteristic features of COPD patients.
Conflict of InterestsDr. J.L. Izquierdo has received fees from Almirall, AstraZéneca, Bayer, Boehringer Ingelheim, Chiesi, Faes, Ferrer, GSK, Novartis, Nycomed and Pfizer for giving conferences and consulting.
Please cite this article: Izquierdo Alonso JL, Rodríguez Glez-Moro JM. Utilización excesiva de corticoides inhalados en la enfermedad pulmonar obstructiva crónica. Arch Bronconeumol. 2012;48:207–12.