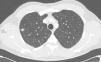
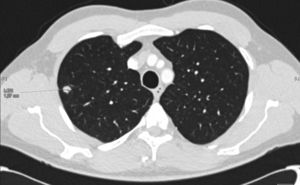
The presence of a solitary pulmonary nodule (SPN) in transplanted patients always raises the possibility of a malignant lesion, but the causes of SPN are very diverse, and infection is another common etiology.1Cryptococcus neoformans lung infection has a low incidence and rarely presents with isolated lung involvement.2
We report the case of a 46-year-old man with terminal chronic kidney disease caused by reflux nephropathy, renal transplant from a cadaver donor 10 years previously, receiving immunosuppressive treatment with prednisone, tacrolimus, and sodium mycophenolate. He attended a routine follow-up visit, where he was seen to be totally asymptomatic, with no suspicious respiratory or infectious clinical symptoms. No significant findings were made on physical examination. Chest X-ray revealed an ovoid lesion in the posterior segment of the right upper lobe (RUL), 12mm in diameter, not observed in the study performed 1 year previously. The lesion was explored in greater depth with a chest CT, which showed a lung nodule suggestive of neoplasm in the RUL (Fig. 1), consistent with stage IA (T1aN0MX). The PET scan showed pathological uptake of this lesion, so malignancy was suspected. The nodule was resected surgically via an atypical segmentectomy with diagnostic and therapeutic intent.
The histological study reported necrotizing granulomatous inflammation with the presence of small rounded structures, heterogeneous in size and with frequent fragmentation, located predominantly within the cytoplasm with signs indicating budding, grayish or eosinophilic on hematoxylin–eosin staining. Stains performed for fungal structures were positive for periodic acid-Schiff (PAS), Grocott's methenamine-silver, mucicarmin, and Alcian Blue, and negative for Ziehl–Neelsen staining, so morphologically the microorganisms were indicative of cryptococcus.
Microbiological testing was completed with serologies that were negative for both C. neoformans and human immunodeficiency virus antibodies. Brain CT scan showed no pathological findings, so lumbar puncture was not performed and additional antifungal treatment was not started. The patient subsequently made good clinical progress and currently remains totally asymptomatic.
Fungal infections are the third cause of infection in solid organ transplantation.3 Specifically, the prevalence of C. neoformans infection in kidney transplantation is low, affecting 0.5%–2.8% of transplanted patients, but it is the third leading cause of fungal infection after Candida spp. and Aspergillus spp.3
C. neoformans is an encapsulated yeast present in soil contaminated with bird and bat droppings.4 Infection is acquired by inhalation of the microorganism,2,4 and the main form of host defense is innate and T-cell-mediated immunity,5 probably explaining the predisposition to this disease among individuals receiving immunosuppressive treatment.6 The most frequent form of infection is reactivation of latent microorganisms acquired in a previous infection.
The annual incidence of cryptococcosis has been calculated at more than 2:1000 renal transplant patients. Some authors believe that the incidence is increasing because of the increased survival of these patients,7 while others, in contrast, report that the incidence remains unchanged.8 The mean time between transplantation and the onset of cryptococcosis is 25 to 33 months, so presentation in our case is unusually late (120 months). Mortality is as high as 20%7 and appears to be greater if the infection appears in the first year after transplantation,8 when mortality is between 20% and 27%.9
Clinical manifestations of cryptococcal infection in the transplanted population are often non-specific and depend on the organ involved. Central nervous system (CNS) involvement, such as subacute meningoencephalitis, is the most common form of clinical presentation.10 The second most affected organ is the lung.2 The clinical manifestations of this form cover a broad spectrum, from acute processes such as pneumonia or pleuritis to radiological findings in asymptomatic patients, especially single nodule or multiple nodules observed on a routine chest X-ray.11 The importance of this form of presentation lies in the fact that the lung is the gateway to disseminated infection, so an intensive diagnostic and therapeutic approach should be taken even in asymptomatic infection. After clinical reassessment, in the absence of neurologic symptoms, negative brain imaging, lack of uptake in PET testing at other levels, and negative serologies against cryptococcus, disseminated disease and CNS involvement could be excluded in our patient.
Microbiological diagnosis of pulmonary cryptococcosis can be confirmed by culturing the fungus in a sputum sample or by detecting cryptococcal antigen in serum.12 Serological detection usually means that the infection is disseminated, which always requires the disease to be ruled out at other levels, especially CNS.13
Another way to diagnose this infection, as in the case presented, is by histological study of a biopsy or surgical specimen, demonstrating the presence of encapsulated yeasts with the usual fungal stains such as PAS and Grocott, and especially mucin, mucicarmin and Alcian blue stains, since the Cryptococcus genus is the only frequently pathogenic fungus that produces mucinous capsular material.14
Fluconazole may be used in mild forms of cryptococcal lung infection. In severe cases with diffuse pulmonary infiltrates or in the context of disseminated infection, treatment is based on amphotericin B combined with flucytosine.15 Surgical treatment, as described, may be indicated in case of diagnostic uncertainty or therapeutic failure. Finally, because infection after transplantation is exceptional and occurs late, antifungal prophylaxis is not indicated.
In conclusion, cryptococcal infection should be considered in the differential diagnosis of SPN in immunocompromised patients, such as the transplanted population, and the diagnostic and therapeutic approach should be proactive as the respiratory tract is considered the gateway to disseminated infection.
The authors wish to thank Dr. Esparza, Dr. Báez, and Dr. Pérez-Arellano for their invaluable help.
Please cite this article as: Fernández Granados S, Fernández Tagarro E, Ramírez Puga A, Guerra Rodríguez R, García Cantón C. Nódulo pulmonar solitario como forma de presentación de la criptococosis en el trasplantado renal. Arch Bronconeumol. 2020;56:396–398.