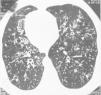
Kartagener's syndrome (KS) is characterized by bronchiectasis, paranasal sinusitis and situs inversus totalis (SIT). Association of diffuse bronchiolitis (DB) with KS has been documented from Japan only. Fourteen patients with SIT were seen in one unit over 7 years. All patients underwent a similar work up which included high resolution computed-tomography (HRCT) of thorax and CT-paranasal sinuses. Semen analysis was done in 2/3 adult males. Eleven patients (6 males and 5 females) had KS while 3 adult females had SIT alone. HRCT-thorax detected bronchiectasis in 10/11 patients with KS. HRCT-thorax confirmed DB in 6/11 patients with KS. One adult male had total sperm count of 2.5million/ml without sperm motility while the other had no sperms.
This series documents the largest number of patients with SIT/KS from India and highlights for the first time, the association of DB with KS from India.
El síndrome de Kartagener (SK) se caracteriza por bronquiectasias, sinusitis paranasal y situs inversus total (SIT). La asociación de la bronquiolitis difusa (BD) con el SK se ha documentado únicamente en Japón. En una unidad se identificaron 14 pacientes con SIT durante un periodo de 7 años. Todos los pacientes fueron objeto de un estudio diagnóstico similar que incluyó una tomografía computarizada (TC) de alta resolución (TCAR) del tórax y TC de senos paranasales. Se realizó un análisis de semen en 2/3 de los varones adultos. Once pacientes (6 varones y 5 mujeres) tenían un SK, mientras que en 3 mujeres adultas había tan solo un SIT. La TCAR de tórax detectó bronquiectasias en 10/11 pacientes con SK. La TCAR de tórax confirmó la BD en 6/11 pacientes con SK. Un varón adulto presentó un recuento espermático total de 2,5millones/ml, sin motilidad espermática, mientras que en los otros no hubo espermatozoides.
Esta serie documenta el mayor número de pacientes con SIT/SK descrito en la India y resalta por primera vez la asociación de BD con SK en este país.
Situs inversus total (SIT) entails a mirror-image reversal of all the asymmetrical structures of the body and it is estimated to occur in 1/8000–1/25000 newborns.1 Most patients with SIT lead a completely normal life but, in approximately 20%–25% of patients, associated primary ciliary dyskinesia (PCD) may also be observed.1 PCD, also known as “immotile ciliary syndrome”, is a genetic disease caused by defects of the structure and function of the cilia that lead to abnormal mucociliary clearance which leads to a clinical disease of the sinus and pulmonary regions. Kartagener's syndrome2 (KS), which is currently classified as a subgroup of PCD, is characterized by the triad of bronchiectasis, paranasal sinusitis and SIT.
We present a review of 14 patients with SIT, 11 of whom had KS. In 6 of these patients, there were signs of diffuse bronchiolitis (DB) on thoracic CT. This manifestation associated with KS was first reported in Japan,3 and it still has not been widely accepted. The presence of oligo/azoospermia was observed in 2 of our patients with KS. As far as we know, this is the largest patient series of SIT cases in India.
Case DescriptionsOver the course of a 7-year period, between April 2003 and March 2010, approximately 11361 new patients (1944 of them from the pediatric age group) with respiratory symptoms were remitted to a unit at our institute. 14 of them (6 males and 8 females; 6 adults and 8 pediatric patients) were diagnosed with SIT. Out of these 14 patients, 11 presented SIT with KS, while 3 only presented SIT (all adult females). The diagnosis of KS was based on the demonstration of the classic triad of the syndrome, consisting of bronchiectasis, paranasal sinusitis and SIT. All patients underwent a similar diagnostic study that included sputum culture for aerobic microorganisms, including Mycobacterium tuberculosis, electrocardiogram, complete spirometry, abdominal ultrasound, computed tomography (CT) with contrast as well as thoracic high resolution CT (HRCT) and CT of paranasal sinuses (PNS). By means of CT, the severity of the bronchiectasis4 and sinusitis5 was evaluated with previously described methods. A semen analysis was done in 2/3 of the adult males who gave their consent.
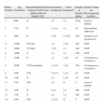
The clinical characteristics of these 14 patients are indicated, by order of presentation, in Table 1.
Demographic Characteristics and Clinical Profile.
| Patient Number | Age (Years)/Sex | Married/Infertility/Semen Analysis (Total Count [millions/ml] and Motility [%]) | Respiratory Symptomsa | Nasal Symptomsb | Quantity of Sputum, ml/day | Sputum Culture for Microorganisms Aerobios |
| 1 | 6/M | N | 1,2 | 1,2 | None | Lack of sputum |
| 2 | 17/F | N | 1,2,3,4 | 1,2 | 20 | Haemophilus influenzae |
| 3 | 16/F | N | 1,2,6 | 1,2,5,6 | 100 | Streptococcus pneumoniae |
| 4 | 22/M | N/2,5y0 | 1,2 | 1,5,6 | 100 | NOP |
| 5 | 28/M | Y/Y/0y0 | 1,2,6 | 1,2 | 100 | NOP |
| 6 | 16/M | N | 1,2 | 1,2 | 100 | NOP |
| 7 | 10/M | N | 1,2 | 1,2,6 | 10 | Pseudomonas aeruginosa |
| 8 | 50/M | Y/Y/Unrealized | 1,2,3,6 | 1,5,6 | 100 | Pseudomonas aeruginosa |
| 9 | 54/F | Y/N | 1,2,3,4 | 1,2,4 | 10 | NOP |
| 10 | 13/F | N | 1,2 | 1,2,6 | 10 | NOP |
| 11 | 77/F | Y/N | 1,2 | N | 5 | Lack of sputum |
| 12 | 15/F | N | 1,2,3 | 1,2 | 100 | NOP |
| 13 | 17/F | N | 1,2 | 1,2 | 100 | NOP |
| 14 | 35/F | Y/N | 1,2,3,4 | N | 5 | NOP |
M, males; F, females, N, no; Y, yes; NOP, no pathologic organism.
Chest HRCT was able to detect bronchiectasis in 10/11 patients with KS. It was observed in a minimum of 2 lobes and in a maximum of 5, but in the majority of the patients there were 4 affected lobes. The left lower lobe was the most affected, with signs in 8/11 patients. This was followed in frequency by the right lower and middle lobes and lingula (7/11 patients each). Bronchiectasis was observed in the upper lobes in 3/11 patients. The overall severity score of the bronchiectasis4 ranged between 0 and 13 out of a total maximum score of 18, with a mean of 5.81±2.75 (Tables 1 and 2). In 6 patients with KS, small diffuse centrilobular nodules were observed (2mm in diameter) with an appearance of “tree in bud” pattern predominantly in the middle lobes, lingula and lower lobes that would suggest diffuse bronchiolitis. In 3/6, we also observed air trapping, tram-tracking and bronchiolectasis. In 4/6, there were grouped thick-walled cystic spaces (bronchi), mainly in the middle lobes and lingula that were suggestive of atelectasis or consolidation (Table 2). In the 3 patients without KS, chest HRCT showed normal lung parenchyma and the CT of the PNS was also within the limits of normality. Table 2 details the radiological manifestations of the patients with KS.
Radiological Tests.
| Structural characteristics on thoracic HRCT | Patients with SK (n=11) |
| Bronchiectasis (any lobe) | 10 |
| Overall severity score of bronchiectasis18 | 5.81±2.75 |
| Affected lobe | |
| Upper left lobe | 3 (patients 1, 3, 4) |
| Middle left lobe | 7 (patients 2, 5, 6, 8, 10, 12, 13) |
| Lower left lobe | 8 (patients 2, 4, 5, 6, 8, 10, 12, 13) |
| Right upper lobe | 3 (patients 4, 5, 8) |
| Right lingula | 7 (patients 3, 5, 6, 8, 10, 12, 13) |
| Lower right lobe | 7 (patients 1, 4, 5, 6, 8, 12, 13) |
| Number of lobes affected | |
| None | 1 (patient 7) |
| One | 0 |
| Two | 3 (patients 1, 2, 3) |
| Three | 1 (patient 10) |
| Four | 4 (patients 4, 6, 12, 13) |
| Five | 1 (patients 5, 8) |
| Six | 0 |
| Thoracic HRCT: other signs | |
| Small centrilobular nodules with Tree-in-bud pattern | 6 (patients 2, 5, 6, 7, 8, 10) |
| Hyperinflation (air trapping) | 3 (patients 5, 8, 10) |
| Consolidation/Colapse of the middle lobe and/or lingula | 4 (patients 2, 6, 7, 10) |
| Pectus carinatum | 1 (patient 6) |
| TC de PNS structural characteristics | Patients con SK (n=7/11) |
| Sinusitis (any) | 7 |
| Overall severity score19 | 15.37±1.99 |
| Hypoplasia/agenesis of the frontal sinus | 6 (patients 1, 6, 7, 8, 12, 13) |
CT, computed tomography; HRCT, high resolution computed tomography; PNS, paranasal sinuses.
Two of the 3 adult males consented to a semen analysis. The 2 patients were married but did not have any children. In one patient, the sperm count was 2.5million/ml with no sperm motility, and in the other no spermatozoids were observed (Table 1). In this latter case, the level of fructose in semen was higher than 100mg/dl.
DiscussionSince the moment when it was described for the first time by Kartagener in 4 patients,2 the syndrome that bears his name has fascinated clinicians for the greater part of the last century. Researchers have discovered many associated anomalies, such as diffuse panbronchiolitis (DPB) in KS, which was described for the first time in Japan nearly 2 decades ago.3 Since then, there have only been another 2 reports of this anomaly,6,7 both also originating in Japan. This association has not been described in any other part of the world.
DPB is a very uncommon form of bronchiolitis, characterized by sinus and bronchial inflammation. It has predominantly been observed in Japan, where it was described for the first time in 1969.8 Since then, there have been sporadic publications of cases documented in other Asian countries and also on other continents.9 On HRCT, DPB is characterized in early stages by numerous small nodules (<5mm) that are poorly defined and have centrilobular distribution. In stage 2, some nodules expand, forming small branching linear areas of attenuation (with a “tree in bud” appearance). In the most advanced phases, there is cystic dilation of the nodules that corresponds with bronchiolectasis. The peripheral areas are less dense, and the presence of an expiratory film would suggest air trapping.10 The fact that imaging tests play a key role in the diagnosis of DPB was vehemently emphasized by Hansell11 in the following affirmation: “The HRCT features of diffuse panbronchiolitis, in the appropriate clinical setting, are virtually pathognomonic.”
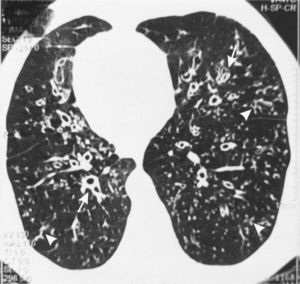
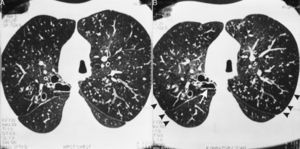
In 6 of our patients with KS, HRCT showed diffuse small centrilobular nodules with the “tree in bud” pattern, bronchiolectasis/bronchiectasis with tram-tracking indicative of diffuse thickening of the bronchial wall, which were “practically pathognomonic”11 for DPB (Fig. 1). Low attenuation areas were observed, which were accentuated on the images in the lung periphery obtained upon expiration, indicative of air trapping (Fig. 2A and B). These signs that suggest DPB in KS/PCD were initially described in 2 patients by Amitani et al.3 in 1990. They were later seen in 6 out of 8 patients with KS by Homma et al.6 in 1999, and in a patient with KS and a malignant disease7 who had been diagnosed with a DPB 11 years before the presentation.
Inspiratory (A) and expiratory (B) high-resolution computed tomography (HRCT) of the same patient as Fig. 1. Reduced attenuation of the pulmonary parenchyma in the periphery (arrowheads) is observed, which is accentuated on the expirator image (B) and suggests air trapping. Likewise, the aortic arch on the right side and cystic bronchiectasis in the posterior segment of the right upper lobe can be observed, which presents a reduction in size in the image obtained on expiration (arrow). There are also diffuse centrilobular nodules with “tree-in-bud” pattern and diffuse ectásicos bronchi with thickened walls.
Homma et al.6 highlighted the notable similarities between KS and DPB with regard to clinical, radiological and spirometric manifestations. Histologically, the inflammatory lesions present in KS were found in the membranous bronchioles, while in DPB they were located in the respiratory bronchioles. The authors proposed that the association of DB could be a “characteristic manifestation”.6 Although the cause of the DPB continues to be unknown, it has been proposed that in PCD the dyskinetic movements of the cilia could cause the appearance of DPB.3,12
Most patients with KS have a favorable prognosis and lead a rather normal life.13 It has been proposed that the ciliary dysfunction leads to a deterioration in ciliary clearance and to the formation of mucus plugs, initially in the small airways and later in the large airways, creating a propensity towards recurring infections that cause bronchiolitis. This leads to air trapping, increased airways resistance, reduced elasticity, hypoxemia, pulmonary hypertension and cor pulmonale. A notable obstruction of the airways was documented in 5/6 patients described by Homma et al.6 and in 4/6 of our patients. In both series, half of the 6 patients with BD presented signs of air trapping on thoracic CT. The appearance of BD in KS could increase morbidity and affect quality of life.
Male patients with KS invariably present infertility, while women present reduced fertility.14 Most infertile patients with KS have a normal spermatozoid count, but with a structural defect and a complete lack of motility.14 Oligospermia and azoospermia are very infrequently observed in KS. However, in our study the semen analysis of the 2 adult patients showed reduced sperm count and motility (oligoasthenozoospermia) in one case and azoospermia in the other. This latter patient presented normal fructose, which suggests the presence of a permeable vas deferens. The presence of azoospermia in KS has previously been reported.15–17 It seems that oligospermia or azoospermia, whose physiopatology has still yet to be defined, can be a variant associated with this syndrome. In addition, some case descriptions have been published of azoospermia obstructiva associated with SIT, which are often labeled as a very uncommon variant of Young syndrome.18,19
We could not determine any anomalies of the dynein arm in our patients, nor did we do any NO nasal inhalado tests. As far as we know, this study documents the largest number of SIT/KS cases in India, and it results for the first time the association of BD with KS in this country.
Conflict of InterestThe authors have no conflict of interest to declare.
Please cite this article as: Mittal V, Shah A. Situs inversus total: asociación de síndrome de Kartagener con bronquiolitis difusa y azoospermia. Arch Bronconeumol. 2012,48:179–82.