To evaluate the effects of an intensive postoperative physiotherapy program focused on respiratory exercises in patients undergoing lobectomy by open thoracotomy.
DesignQuasi-experimental study.
SettingTertiary referral academic hospital.
Participants208 patients undergoing lobectomy by open thoracotomy.
InterventionsControl group patients (n=102) received standard medical/nursing care, and experimental group patients (n=106) added to the standard clinical pathway a daily physiotherapy program focused on respiratory exercises until discharge.
OutcomesAnalyzed outcomes were the frequency of postoperative pulmonary complications (PPCs) more amenable to physiotherapy (pneumonia, atelectasis and respiratory insufficiency) and length of hospital stay (LOS).
ResultsBoth groups were comparable regarding preoperative and surgical characteristics. Incidence of PPCs was 20.6% in control and 6.6% in experimental group (P=.003). Median (IQR) LOS in control group was 14 (7) days (Huber M estimator 14.21) and 12 (6) days (Huber M estimator 12.81) in experimental. Logistic regression model identified the evaluated physiotherapy program (P=.017; EXP [B] 95% CI 0.081–0.780) and % FEV1 (P=.042; EXP [B] 95% CI 0.941–0.999) as protective factors for the development of PPCs in patients undergoing lobectomy.
ConclusionsImplementing a postoperative intensive physiotherapy program focused on respiratory exercises reduces the risk of PPCs and resultant LOS on patients undergoing lobectomy.
Evaluar los efectos de un programa intensivo de fisioterapia postoperatoria basado en ejercicios respiratorios dirigido a pacientes lobectomizados mediante toracotomía abierta.
DiseñoEstudio cuasiexperimental.
EmplazamientoHospital universitario terciario.
ParticipantesDoscientos ocho (208) pacientes lobectomizados mediante toracotomía abierta.
IntervenciónLos pacientes del grupo control (n=102) recibieron atención médica/de enfermería estándar y los pacientes del grupo experimental (n=106), además de la atención clínica estándar, se sometieron a un programa de fisioterapia diaria basada en ejercicios respiratorios hasta el momento del alta hospitalaria.
Variables de resultadoLas variables de resultado estudiadas incluyeron la frecuencia de complicaciones pulmonares postoperatorias (CPP) más susceptibles de tratamiento fisioterapéutico (neumonía, atelectasias e insuficiencia respiratoria) y la duración de la estancia hospitalaria (DEH).
ResultadosLas características preoperatorias y quirúrgicas de ambos grupos fueron comparables. La incidencia de CPP registrada fue de un 20,6% en el grupo control y un 6,6% en el grupo experimental (p=0,003). La mediana y el RIC de la DEH fue de 14 y 7días, respectivamente (estimador M de Huber 14,21) en el grupo control y de 12 y 6días (estimador M de Huber 12,81) en el grupo experimental. El modelo de regresión logística creado identificó al programa de fisioterapia evaluado (p=0,017; EXP (B) [IC 95% 0,081–0,780]) y al porcentaje del FEV1 (p=0,042; EXP (B) [IC 95% 0,941–0,999]) como factores protectores frente al desarrollo de CPP en los pacientes intervenidos de lobectomía.
ConclusionesLa implementación de un programa intensivo de fisioterapia postoperatoria basado en ejercicios respiratorios reduce el riesgo de desarrollar CPP y la DEH en pacientes lobectomizados.
Perioperative physiotherapy has regularly been used to prevent or reduce pulmonary morbidity after lung resection1,2 and has recently been recommended by the European Respiratory Society, the European Society of Thoracic Surgeons and the American College of Chest Physicians for providing functional benefits.3,4 In 2006, Varela and co-workers5 published a cross-sectional study showing that implementing an intensive perioperative respiratory physiotherapy program for patients undergoing lobectomy, in addition to incentive spirometry and standard nursing care, decreased the rate of postoperative atelectasis and produced costs savings associated with a shorter hospital stay. The experimental program started the day before surgery and was based on aerobic exercise up to discharge.
To date, no single-blind randomized clinical trial evaluating the benefits of respiratory physiotherapy or an added specific intervention, focusing only on the immediate perioperative period, has shown significant differences between groups in the frequency of postoperative pulmonary complications (PPCs) or length of stay (LOS).6–10 There is a need to identify exactly which interventions are most beneficial in the immediate perioperative period in terms of reducing PPCs and in which patients, particularly now that guidelines are recommending surgery in increasingly higher risk patients.3 This is of clinical and economic importance as PPCs are directly associated with higher mortality and costs.11
Hence, the aim of this study was to evaluate the effects of a postoperative physiotherapy program based on respiratory exercises in reducing PPCs and LOS in patients undergoing lobectomy.
MethodsDesignQuasi-experimental study.
ParticipantsThis was a comparative study of the medical records of a total of 208 patients undergoing lobectomy in a tertiary referral teaching hospital (Complejo Hospitalario de Navarra). Control group patients underwent the procedure between 2006 and 2007 and experimental group patients between 2009 and 2010, after the hospital introduced a daily postoperative physiotherapy program until discharge. We did not include data from patients who underwent the procedure in 2008, as the program was not systematically established during that period. Both series were operated on by the same team, following similar operative and resectability selection criteria and anesthetic management. The approach in all patients was open thoracotomy, some with posterior muscle-sparing or a small axillary thoracotomy, at the surgeon's choice. This study was approved by the local institutional review board.
Control GroupPatients in the control group received a volumetric incentive spirometer device in the preoperative room and were instructed how to perform deep inspiratory maneuvers at a moderate flow, maintaining an inspiratory apnea for 3–5s. Patients were asked to start practicing the lung expansion exercises soon after the intervention in the post-anesthesia recovery unit, and to continue every hour until discharge. Once in the thoracic ward, nurses were responsible for encouraging patients to adopt an early upright position and ambulation from the first postoperative day.
Experimental GroupThe experimental group received standard care as above, plus daily targeted individual respiratory physiotherapy sessions until discharge. Treatment design included two main objectives: patients should (1) achieve proficiency in pulmonary re-expansion techniques and (2) become autonomous in airway clearance maneuvers. Physiotherapy sessions lasted around 20min and exercise techniques most commonly included were maximum sustained inspirations, debit controlled inspiratory exercises (EDIC), autogenic drainage, effective cough training, and progressive shoulder and thoracic cage weight-free mobility exercises. Patients were encouraged to practice the learned exercises independently during waking hours at least 3 more times a day. The importance of the patient's active role in their recovery during the immediate postoperative period (early ambulation, pulmonary hygiene, lung expansion exercises, etc.) was strongly emphasized during treatment sessions.
Variables and OutcomesThe comparability of both series was assessed by the following independent variables: sex, age, predicted preoperative forced expiratory volume in 1s (%FEV1), body mass index (BMI), smoking habit at the time of operation (current smoker or not), Charlson comorbidity index,12 diagnosis, and patient-controlled epidural analgesia (PCEA).
The primary outcome was the incidence of postoperative pulmonary complications (PPCs), considered the presence of at least a diagnosis of pneumonia, atelectasis or acute respiratory failure in medical records. Both pneumonia and atelectasis were documented medically when clinical signs [temperature >38°C, production of purulent (yellow/green) sputum differing from preoperative status, no breath sounds on auscultation, etc.] were accompanied by chest X-ray confirmation. Acute respiratory failure was recorded when clinical signs were accompanied by arterial blood gases showing PaO2<60mmHg. It should also be noted that the postoperative radiological protocol during the study period consisted of three chest X-rays: the first early in the recovery room, the second on the second postoperative day, and the last after tube removal. No other radiography was indicated unless dictated by clinical change.
The secondary outcome was length of stay (LOS) calculated as the difference between hospital admission and discharge dates.
Statistical AnalysisContinuous variables are expressed as mean (SD) when normally distributed and as median with interquartile range (IQR) when not. Categorical variables are expressed as frequency counts and percentages. Univariate analyses were performed using appropriate statistical tests according to the type and distribution of the data: t test or Mann–Whitney U-test for continuous variables and Chi-squared test for categorical variables. The Huber M-estimator was calculated for continuous outcomes. A P value of <.05 was considered significant. Variables that reached a P value of <.20 on bivariate analysis were considered eligible for entry in the logistic regression analysis. In addition, we have specified Hosmer–Lemershow goodness-of-fit, omnibus and Nagelkerke's R2 values for each model. Statistical analysis was performed using SPSS v.21 software.
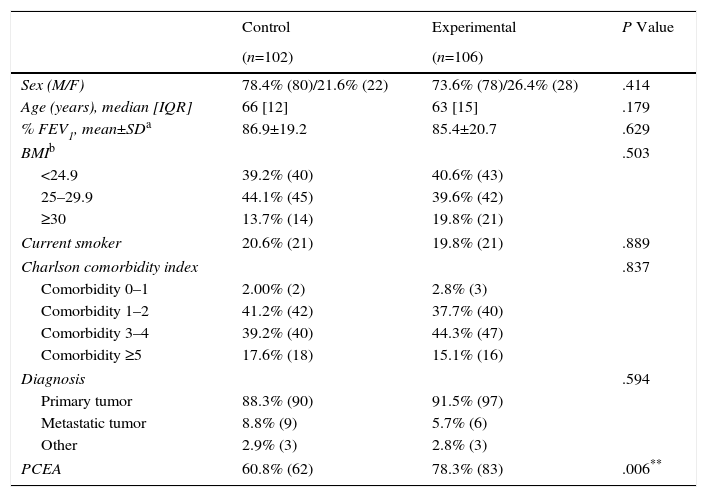
ResultsParticipantsIn the study period, a total of 208 patients underwent lobectomy by open thoracotomy (posterolateral or axillary). All information was collected directly from computerized medical records. Table 1 shows the baseline characteristics of participants. Patients in both series were comparable in sex, age, %FEV1, BMI, smoking habit at the time of the intervention, Charlson comorbidity index, and diagnosis. Significant differences were found in the postoperative analgesic method (P=.006).
Baseline and Demographic Characteristics of All Participants.
| Control | Experimental | P Value | |
|---|---|---|---|
| (n=102) | (n=106) | ||
| Sex (M/F) | 78.4% (80)/21.6% (22) | 73.6% (78)/26.4% (28) | .414 |
| Age (years), median [IQR] | 66 [12] | 63 [15] | .179 |
| % FEV1, mean±SDa | 86.9±19.2 | 85.4±20.7 | .629 |
| BMIb | .503 | ||
| <24.9 | 39.2% (40) | 40.6% (43) | |
| 25–29.9 | 44.1% (45) | 39.6% (42) | |
| ≥30 | 13.7% (14) | 19.8% (21) | |
| Current smoker | 20.6% (21) | 19.8% (21) | .889 |
| Charlson comorbidity index | .837 | ||
| Comorbidity 0–1 | 2.00% (2) | 2.8% (3) | |
| Comorbidity 1–2 | 41.2% (42) | 37.7% (40) | |
| Comorbidity 3–4 | 39.2% (40) | 44.3% (47) | |
| Comorbidity ≥5 | 17.6% (18) | 15.1% (16) | |
| Diagnosis | .594 | ||
| Primary tumor | 88.3% (90) | 91.5% (97) | |
| Metastatic tumor | 8.8% (9) | 5.7% (6) | |
| Other | 2.9% (3) | 2.8% (3) | |
| PCEA | 60.8% (62) | 78.3% (83) | .006** |
Data shown as % (n).
M, male; F, female; FEV1, forced expiratory volume in 1s; BMI, body mass index; PCEA, patient-controlled epidural analgesia.
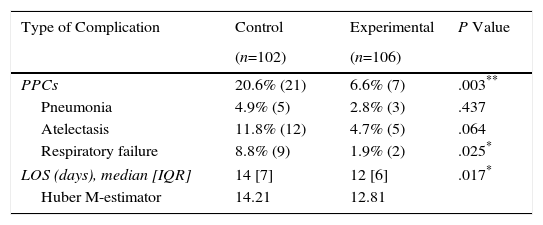
Table 2 presents the frequency and type of the recorded postoperative pulmonary complications and LOS. PPC incidence (which was recognized by the presence of at least a diagnosis of pneumonia, atelectasis or acute respiratory failure in medical records) was significantly reduced in the experimental group (20.6% in the control group vs 6.6% in the experimental group; P=.003). Respiratory failure, atelectasis and pneumonia incidence also decreased in the experimental group (incidence of respiratory failure: 8.8% in controls and 1.9% in the experimental group; incidence of atelectasis: 11.7% in controls and 4.7% in the experimental group; incidence of pneumonia: 4.9% in controls and 2.8% in the experimental group), but only respiratory failure reached statistical significance (P=.025). Median LOS was also significantly lower in the experimental group: 14 days (IQR 7) in the control group, and 12 days (IQR 6) in the experimental group (P=.017). In any case, LOS in the experimental group did not exceed 12.81 days according to Huber M-estimator and 14.21 days in the control group.
Frequency and Type of the Morbidity and Length of Stay in Control and Experimental Groups.
| Type of Complication | Control | Experimental | P Value |
|---|---|---|---|
| (n=102) | (n=106) | ||
| PPCs | 20.6% (21) | 6.6% (7) | .003** |
| Pneumonia | 4.9% (5) | 2.8% (3) | .437 |
| Atelectasis | 11.8% (12) | 4.7% (5) | .064 |
| Respiratory failure | 8.8% (9) | 1.9% (2) | .025* |
| LOS (days), median [IQR] | 14 [7] | 12 [6] | .017* |
| Huber M-estimator | 14.21 | 12.81 | |
Data shown as % (n).
PPCs, postoperative pulmonary complications; LOS, length of hospital stay.
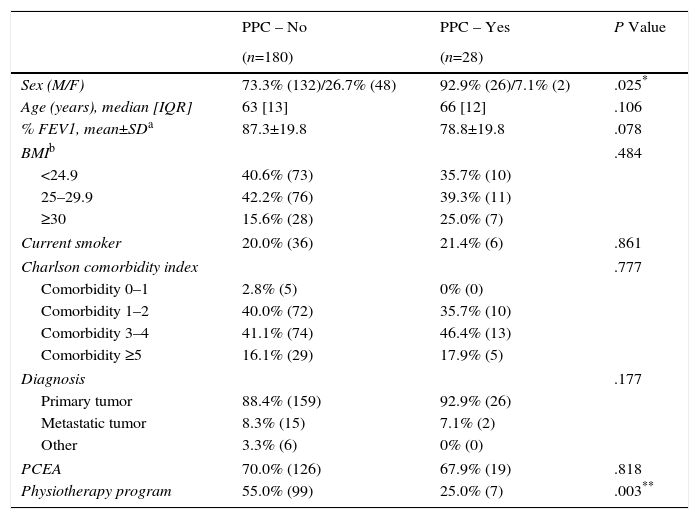
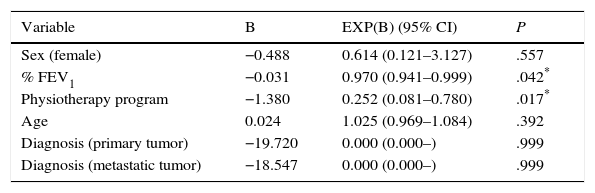
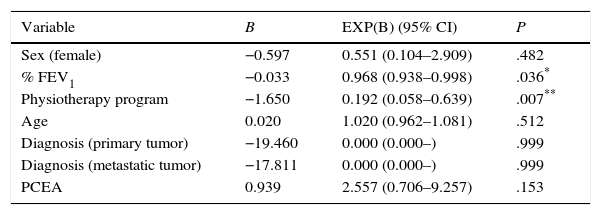
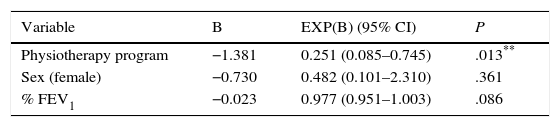
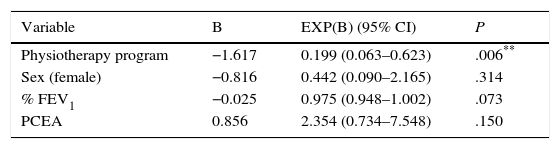
Variables that reached a P value of <.20 on bivariate analysis (Table 3) were considered eligible for entry in the logistic regression analysis. Results of the regression model are presented in Table 4. The risk of developing PPCs reached statistical significance for physiotherapy program (P=.017, 95% CI: 0.081–0.780) and for %FEV1: P=.042, 95% CI: 0.941–0.999 [sex: P=.557, 95% CI: 0.121–3.127; age: P=.392, 95% CI: 0.969–1.084; diagnosis (primary tumor): P=.999, 95% CI: 0.000–; diagnosis (metastatic tumor): P=.999, 95% CI: 0.000–]. Table 5 shows a regression model which also includes epidural catheter, because, while it showed no influence on bivariate analysis, its role in the control of postoperative pain is well recognized.13 No association was found between epidural analgesia and development of PPCs (P=.153, 95% CI: 0.706–9.257). Appendix 1a shows the logistic regression analysis results considering variables that showed a P value of <.05 on bivariate analysis (Table 3) and Appendix 1b shows the logistic regression analysis results considering variables that showed a P value of <.05 on bivariate analysis (Table 3), also including epidural catheter.
Post-operative Pulmonary Complications: Bivariate Analysis.
| PPC – No | PPC – Yes | P Value | |
|---|---|---|---|
| (n=180) | (n=28) | ||
| Sex (M/F) | 73.3% (132)/26.7% (48) | 92.9% (26)/7.1% (2) | .025* |
| Age (years), median [IQR] | 63 [13] | 66 [12] | .106 |
| % FEV1, mean±SDa | 87.3±19.8 | 78.8±19.8 | .078 |
| BMIb | .484 | ||
| <24.9 | 40.6% (73) | 35.7% (10) | |
| 25–29.9 | 42.2% (76) | 39.3% (11) | |
| ≥30 | 15.6% (28) | 25.0% (7) | |
| Current smoker | 20.0% (36) | 21.4% (6) | .861 |
| Charlson comorbidity index | .777 | ||
| Comorbidity 0–1 | 2.8% (5) | 0% (0) | |
| Comorbidity 1–2 | 40.0% (72) | 35.7% (10) | |
| Comorbidity 3–4 | 41.1% (74) | 46.4% (13) | |
| Comorbidity ≥5 | 16.1% (29) | 17.9% (5) | |
| Diagnosis | .177 | ||
| Primary tumor | 88.4% (159) | 92.9% (26) | |
| Metastatic tumor | 8.3% (15) | 7.1% (2) | |
| Other | 3.3% (6) | 0% (0) | |
| PCEA | 70.0% (126) | 67.9% (19) | .818 |
| Physiotherapy program | 55.0% (99) | 25.0% (7) | .003** |
Data shown as % (n).
PPC, postoperative pulmonary complications; M, male; F, female; FEV1, forced expiratory volume in 1s; BMI, body mass index; PCEA, patient controlled epidural analgesia.
Logistic Regression Analysis of Variables Influencing PPCs.
| Variable | B | EXP(B) (95% CI) | P |
|---|---|---|---|
| Sex (female) | −0.488 | 0.614 (0.121–3.127) | .557 |
| % FEV1 | −0.031 | 0.970 (0.941–0.999) | .042* |
| Physiotherapy program | −1.380 | 0.252 (0.081–0.780) | .017* |
| Age | 0.024 | 1.025 (0.969–1.084) | .392 |
| Diagnosis (primary tumor) | −19.720 | 0.000 (0.000–) | .999 |
| Diagnosis (metastatic tumor) | −18.547 | 0.000 (0.000–) | .999 |
PPCs, postoperative pulmonary complications; FEV1, forced expiratory volume in 1s.
Logistic Regression Analysis of Variables Influencing PPCs, Including PCEA.
| Variable | B | EXP(B) (95% CI) | P |
|---|---|---|---|
| Sex (female) | −0.597 | 0.551 (0.104–2.909) | .482 |
| % FEV1 | −0.033 | 0.968 (0.938–0.998) | .036* |
| Physiotherapy program | −1.650 | 0.192 (0.058–0.639) | .007** |
| Age | 0.020 | 1.020 (0.962–1.081) | .512 |
| Diagnosis (primary tumor) | −19.460 | 0.000 (0.000–) | .999 |
| Diagnosis (metastatic tumor) | −17.811 | 0.000 (0.000–) | .999 |
| PCEA | 0.939 | 2.557 (0.706–9.257) | .153 |
PPCs, postoperative pulmonary complications; FEV1, forced expiratory volume in 1s; PCEA, patient-controlled epidural analgesia.
Our results indicate that the implementation of a postoperative intensive physiotherapy program based on respiratory exercises is effective in reducing the risk in patients undergoing lobectomy of PPCs (P=.017; 95% CI: 0.081–0.780) and LOS (P=.017; median [IQR] in control group 14 [7] and 12 [6] in experimental group; Huber M-estimator 14.21 in control group and 12.81 in experimental group). To the best of our knowledge, this is the first study showing the benefits derived from a physiotherapy program added to standard nursing care and incentive spirometry that it is not based on cardiopulmonary exercise.
The importance of controlling factors that could affect the internal validity of a study, particularly incision type, is well established. In this regard, we emphasize that case selection criteria remained substantially unchanged during the study period; both series were operated on by the same team, following similar operative and resectability selection criteria and anesthetic management. The approach for all patients was open thoracotomy, some with posterior muscle-sparing or axillary thoracotomy, at the surgeon's choice, but we did not collect this information because we considered, as others have done before,5,14–16 that both surgical approaches are homogeneous. Furthermore, prior evidence is available showing that these different types of incision have similar postoperative pulmonary function outcomes, complications, and resulting pain.17–19 Given all this, we believe that all relevant clinical strategies have been included. Moreover, we are aware that surgical practices in recent years have changed and that video-assisted thoracoscopic surgery (VATS) lung resections are increasingly common, whereas all patients in the present study were operated on using open thoracotomy. Nevertheless, data published by the European Society of Thoracic Surgeons20 report open lobectomy as still the most widely used surgical procedure in lung resection, so the results of this study remain useful for current clinical practice and meaningful for a wide population.
We observed a significantly higher number of epidural catheters for postoperative analgesia in the experimental group (78.3%) compared with the control group (60.8%) (P=.006). Postoperative analgesia by epidural catheter consisted of bupivacaine, fentanyl and adrenalin, while the intravenous route consisted of morphine, metamizol and droperidol; both were administered using patient-controlled systems. Epidural analgesia has a recognized role in the control of postoperative pain,13 so it may be that patients experience less pain, thus influencing outcomes. In this study, however, analgesia by PCEA provided no significant benefit in the bivariate (P=.818) or multivariate analysis (P=.153, 95% CI: 0.706–9.257) of PPCs. These results are consistent with the latest systematic reviews conducted by Romero et al.21 and Joshi et al.22 in concluding that although epidural analgesia is more effective than intravenous in reducing post-thoracotomy pain, it has no proven efficacy in reducing the incidence of PPCs.
Moreover, if we compare our results with recent studies of physiotherapy interventions focusing only on the immediate perioperative period, our PPC frequency (6.6% in experimental group) was similar to that reported by Novoa et al.14 (4.7%) and Reeve et al.9 (4.8%) but considerably lower than that of Agostini et al.6 (12%), Arbane et al.23 (16%), and Ludwig et al.8 (19%). Variability in the reported PPC incidences between studies has usually been explained in part by the differences in the definitions included under the term “PCC”. In this study, PPCs included were limited to pulmonary complications more amenable to physiotherapy intervention,24–26 to wit, atelectasis, pneumonia and respiratory failure. Moreover, any medical report of atelectasis and pneumonia was always confirmed by radiological images and respiratory failure was confirmed by arterial blood gases.
Our lung function results indicate that preoperative %FEV1 played a role in the development of PPCs in the study population (P=.042; 95% CI: 0.941–0.999). These results are consistent with the recommendations published in the clinical guidelines of the European Respiratory Society and the European Society of Thoracic Surgeons,3 which suggest that the threshold for increased risk of postoperative mortality and morbidity in patients undergoing lung resection is a ppoFEV1 of 30%. The preoperative %FEV1 in our study was 86.9% (SD 19.2) in the control group and 85.4% (SD 20.7) in the experimental group, explaining how this variable was close to statistical significance in the bivariate analysis of PPCs (P=.078). Thus, the influence of %FEV1 on the risk of developing PPCs in our study should be interpreted with some caution.
We found that LOS (median [IQR] 14 [7] days; Huber M-estimator 14.21 in the control group and 12 [6] days; Huber M-estimator 12.81 in the experimental group) was significantly reduced by the implementation of the physiotherapy program (P=.017). Even though a decrease in LOS is demonstrated, hospitalization in our series was longer than that currently reported by other studies: Reeve et al.9 reported a median of 6 days, Agostini et al.6 5 days, and Arbane et al.23 7.7 days. The difference in LOS may be explained by diversity in medical care and discharge criteria. The criteria followed in our study required a drain of less than 200ml/day for chest tube removal, and no patient was discharged with chest tubes. It would be interesting to know how chest tube removal was managed in the above-mentioned studies, and if this was associated with LOS. While Agostini et al.6 and Reeve et al.9 included different operative procedures in their studies (lobectomies, pneumonectomies, wedge resections, sleeve resections, etc.) and the approach in all patients was open thoracotomy, the series of Arbane et al.23 included both lobectomies and pneumonectomies, approached by open thoracotomy or VATS. This heterogeneity in the study population could influence LOS, as minor lung resection procedures and VATS have better perioperative outcomes than other procedures or open thoracotomy, including duration of hospitalization.27 It would also be interesting to study the influence of LOS on post-hospitalization outcomes, such as emergency visits and/or hospital readmission frequency for any surgery-related cause. This topic was studied in 2004 by Varela et al., 28 who reported a readmission rate of 5% during the first 30 days after discharge in patients operated on by lobectomy; the authors found no influence of LOS on post-hospitalization outcomes (median LOS 7 days). We subsequently re-reviewed the medical records of our patients and found a decrease in readmission incidence for any surgery-related cause from 5.9% (6 patients) in the control group to 2.8% (3 patients) in the experimental group, though the reduction was not statistically significant (P=.268). In our opinion, cost-effectiveness studies that also take into account post-hospitalization outcomes would be necessary to assess the benefits of physiotherapy interventions from a broader perspective; this has important clinical bearing and is essential for taking evidence-based decisions.
We considered introducing propensity-matched methodology,29 but we finally decided not to, due to the similarity between the control and experimental groups in baseline and perioperative characteristics, and since this procedure would reduce the final sample size.
The limitations of our study are derived mainly from its retrospective design, which made it difficult to recover data. The fact that all perioperative practices remained unchanged through the study period could minimize design deficiencies. No diffusing capacity of the lung for carbon monoxide (DLCO) or exercise capacity variables were recorded, as these parameters were not determined at the time of data collection.
Finally, the significant clinical and economic impact of PPCs in patients undergoing lung resection (increased mortality and costs)11 generates a need to determine the role of postoperative physiotherapy in reducing such events. Future research should explore the optimum interventions for different sub-groups of patients, not only in the immediate perioperative period but also before and after thoracic surgery. This knowledge would help in the development of guidelines for clinicians and policymakers, and would also increase health-related quality of life in this patient population.
In conclusion, a postoperative intensive physiotherapy program focused on respiratory exercises has demonstrated efficacy in reducing the risk of PPCs and LOS in patients undergoing lobectomy.
Conflict of InterestThe authors declare no conflict of interest.
Ethical ApprovalThis study was approved by the local institutional review board.
The authors wish to thank Mª Jesús Condón Huerta, Head of the Physical Medicine and Rehabilitation Department of the Complejo Hospitalario de Navarra (Pamplona) for her support which made this study possible.
Logistic regression analysis of variables influencing PPCs.
| Variable | B | EXP(B) (95% CI) | P |
|---|---|---|---|
| Physiotherapy program | −1.381 | 0.251 (0.085–0.745) | .013** |
| Sex (female) | −0.730 | 0.482 (0.101–2.310) | .361 |
| % FEV1 | −0.023 | 0.977 (0.951–1.003) | .086 |
PPCs, postoperative pulmonary complications; FEV1, forced expiratory volume in 1s.
Logistic regression analysis of variables influencing PPCs, including PCEA.
| Variable | B | EXP(B) (95% CI) | P |
|---|---|---|---|
| Physiotherapy program | −1.617 | 0.199 (0.063–0.623) | .006** |
| Sex (female) | −0.816 | 0.442 (0.090–2.165) | .314 |
| % FEV1 | −0.025 | 0.975 (0.948–1.002) | .073 |
| PCEA | 0.856 | 2.354 (0.734–7.548) | .150 |
PPCs, postoperative pulmonary complications; FEV1, forced expiratory volume in 1s; PCEA, patient-controlled epidural analgesia.
Please cite this article as: Rodriguez-Larrad A, Vellosillo-Ortega JM, Ruiz-Muneta C, Abecia-Inchaurregui LC, Seco J. Los ejercicios respiratorios postoperatorios reducen el riesgo de complicaciones pulmonares en pacientes sometidos a lobectomía. Arch Bronconeumol. 2016;52:347–353.