The use of pleural manometry (PM) during therapeutic thoracentesis is controversial.1,2 A small study of malignant pleural effusions (MPE) conducted 20 years ago suggested that pleural elastance (PEL), the ability of the lung to return to its natural position after the extraction of pleural fluid (PF),3 can predict the success of pleurodesis.4 This led to the recommendation that unexpandable lung (UEL)3 should be identified in order to guide management decisions.5 The objective of this study was to evaluate if the success of pleurodesis in MPE can be predicted by identifying various biochemical parameters in PF and determining PEL.
We performed a retrospective study of all MPEs (cytology or pleural biopsy positive for malignancy) managed with therapeutic thoracentesis with PM and subsequent pleurodesis between January 2014 and October 2016. Exclusion criteria were previous chemotherapy/radiation therapy, life expectancy <1 month, or loculated PE. Patients signed an informed consent form before therapeutic thoracentesis with PM was performed. Our study was approved by the hospital ethics committee (registry 2016/518).
Pleural pressure was measured with a digital manometer (Compass; Mirador Biomedical),6 using a previously described technique.7 Thoracentesis was completed when no more fluid could be extracted, pleural pressure reached −20cmH2O, or if chest pain developed.8PEL was calculated based on the formula: [opening pressure–closing pressure (cmH2O)]/volume of fluid extracted (in liters). A diagnosis of UEL was reached if incomplete pulmonary reexpansion was observed on the post-thoracocentesis chest X-ray.
Pleurodesis with a slow injection of suspension of 4g talc in 50cc 0.9% saline solution was administered after evacuating the PE via a chest tube (16F) and checking lung reexpansion on X-ray, irrespective of the amount of fluid drained on a daily basis.9 The tube was closed for 2–3h, and then connected to mild progressive aspiration. The chest tube was removed after 24h, regardless of the volume of fluid obtained, and without radiological monitoring.10 Pleurodesis was considered to have been successful if no reaccumulation or only partial accumulation of PE not requiring further thoracentesis occurred until the time of death.11 It was considered to have failed if the effusion recurred or new procedures were needed for the relief of symptoms. The decision to perform pleurodesis was not based on the PF analyses or PEL results.
Fasting PF and blood specimens were obtained simultaneously. Biochemical parameters determined in PF were those included in the routine protocol of our hospital. Data are listed as mean±standard deviation, or median and 25th–75th percentiles, depending on whether the distribution of the samples was normal or not. Pearson's Chi-squared test was used for the comparison between groups if the variables were qualitative, and the non-parametric Mann–Whitney test was used if they were quantitative. ROC curves and the area under the curve were calculated to assess the discrimination capacity of the markers in the prognosis of pleurodesis (success/failure).
In total, 148PMs were performed, of which 110 were cases of MPE. Pleurodesis was performed in 36 patients [20 women and 16 men (mean age: 65.2±12.9 years; range, 18–89)]. Seventy-four patients were excluded due to previous chemotherapy/radiation therapy (31), short life expectancy (28), loculated PE (8), and no signed informed consent (7). UEL was diagnosed in 8 patients (22.2%).
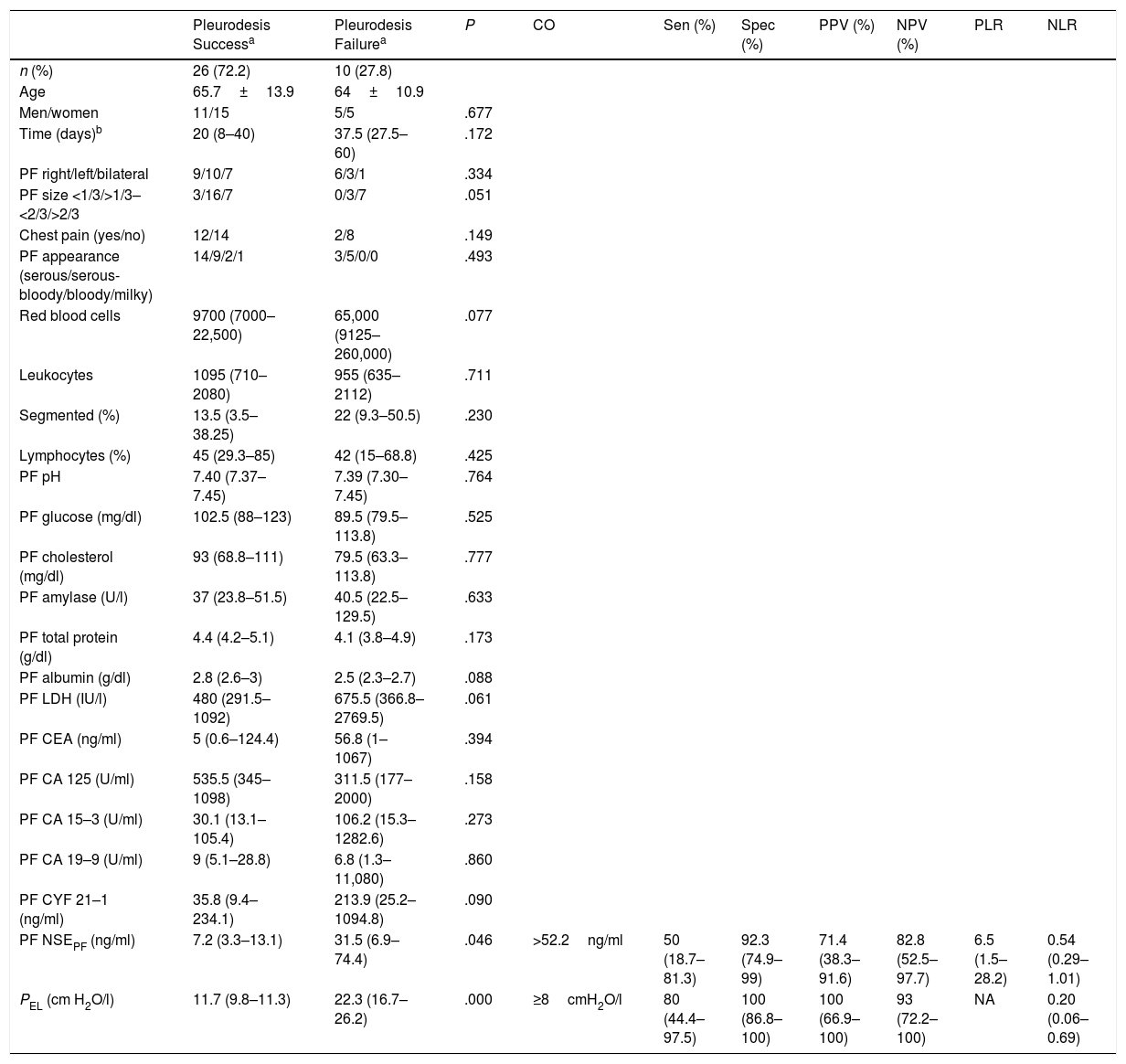
Pleurodesis was successful in 26 patients (72.2%), and failed in 10 (27.8%) (Table 1). Significant differences between the groups were only found for neuron-specific enolase in PF (NSEPF) (P=.046) and for PEL (P=.000). The diagnostic yield of these 2 variables for predicting pleurodesis failure is shown in the same table [areas under the ROC curves for NSEPF and PEL of 0.717 (0.526–0.908) and 0.935 (0.842–1.027), respectively].
Characteristics of the Total Number of Patients by Pleurodesis Response and Diagnostic Yield of the Variables with Significant Differences Between the Pleurodesis Response Groups to Predict Failure [Percentages (95% CI)].
| Pleurodesis Successa | Pleurodesis Failurea | P | CO | Sen (%) | Spec (%) | PPV (%) | NPV (%) | PLR | NLR | |
|---|---|---|---|---|---|---|---|---|---|---|
| n (%) | 26 (72.2) | 10 (27.8) | ||||||||
| Age | 65.7±13.9 | 64±10.9 | ||||||||
| Men/women | 11/15 | 5/5 | .677 | |||||||
| Time (days)b | 20 (8–40) | 37.5 (27.5–60) | .172 | |||||||
| PF right/left/bilateral | 9/10/7 | 6/3/1 | .334 | |||||||
| PF size <1/3/>1/3–<2/3/>2/3 | 3/16/7 | 0/3/7 | .051 | |||||||
| Chest pain (yes/no) | 12/14 | 2/8 | .149 | |||||||
| PF appearance (serous/serous-bloody/bloody/milky) | 14/9/2/1 | 3/5/0/0 | .493 | |||||||
| Red blood cells | 9700 (7000–22,500) | 65,000 (9125–260,000) | .077 | |||||||
| Leukocytes | 1095 (710–2080) | 955 (635–2112) | .711 | |||||||
| Segmented (%) | 13.5 (3.5–38.25) | 22 (9.3–50.5) | .230 | |||||||
| Lymphocytes (%) | 45 (29.3–85) | 42 (15–68.8) | .425 | |||||||
| PF pH | 7.40 (7.37–7.45) | 7.39 (7.30–7.45) | .764 | |||||||
| PF glucose (mg/dl) | 102.5 (88–123) | 89.5 (79.5–113.8) | .525 | |||||||
| PF cholesterol (mg/dl) | 93 (68.8–111) | 79.5 (63.3–113.8) | .777 | |||||||
| PF amylase (U/l) | 37 (23.8–51.5) | 40.5 (22.5–129.5) | .633 | |||||||
| PF total protein (g/dl) | 4.4 (4.2–5.1) | 4.1 (3.8–4.9) | .173 | |||||||
| PF albumin (g/dl) | 2.8 (2.6–3) | 2.5 (2.3–2.7) | .088 | |||||||
| PF LDH (IU/l) | 480 (291.5–1092) | 675.5 (366.8–2769.5) | .061 | |||||||
| PF CEA (ng/ml) | 5 (0.6–124.4) | 56.8 (1–1067) | .394 | |||||||
| PF CA 125 (U/ml) | 535.5 (345–1098) | 311.5 (177–2000) | .158 | |||||||
| PF CA 15–3 (U/ml) | 30.1 (13.1–105.4) | 106.2 (15.3–1282.6) | .273 | |||||||
| PF CA 19–9 (U/ml) | 9 (5.1–28.8) | 6.8 (1.3–11,080) | .860 | |||||||
| PF CYF 21–1 (ng/ml) | 35.8 (9.4–234.1) | 213.9 (25.2–1094.8) | .090 | |||||||
| PF NSEPF (ng/ml) | 7.2 (3.3–13.1) | 31.5 (6.9–74.4) | .046 | >52.2ng/ml | 50 (18.7–81.3) | 92.3 (74.9–99) | 71.4 (38.3–91.6) | 82.8 (52.5–97.7) | 6.5 (1.5–28.2) | 0.54 (0.29–1.01) |
| PEL (cm H2O/l) | 11.7 (9.8–11.3) | 22.3 (16.7–26.2) | .000 | ≥8cmH2O/l | 80 (44.4–97.5) | 100 (86.8–100) | 100 (66.9–100) | 93 (72.2–100) | NA | 0.20 (0.06–0.69) |
[Median (25th–75th percentiles)].
CA 125, cancer antigen 125; CA 15–3, carbohydrate antigen 15–3; CA19-9, carbohydrate antigen 19–9; CEA, carcinoembryonic antigen; CO, cut-off point; CYF 21–1, cytokeratin fragment 21–1; LDH, lactate dehydrogenase; NA, not applicable; NPV, negative predictive value; NSE, neuron-specific enolase; PEL, pleural elastance; PF, pleural fluid; PPV, positive predictive value; PVR, positive likelihood ratio; RVN, negative likelihood ratio; Sen, sensitivity; Spec, specificity.
This study confirms that PEL is a useful parameter for predicting the response to pleurodesis in MPE, and that values ≥18cmH2O/l suggest that the procedure will fail, due to poor apposition of the two pleural membranes12 and the inability of the lung to return to its natural position as the PF is extracted. Pleurodesis can also fail in patients with an expandable lung and PEL below this cut-off point (2/28; 7.1%). This seems to indicate that other factors may cause pleurodesis failure, such as insufficient inflammatory response to cause fibrosis and produce pleural symphysis, or time elapsed between diagnosis and starting drainage.13
The only biochemical parameter that showed some discriminant value for predicting the failure of pleurodesis was NSEPF, which was not significantly different from that of PEL (P=.07564), probably because of the small size of the series and the width of the confidence intervals. Thus, the lower limit of the positive likelihood ratio of NSEPF (1.5) has little effect on the probability of diagnosing pleurodesis failure. The reason why NSEPF is high in these patients is still unknown. Perhaps, as in rheumatoid arthritis, a situation of hypoxia is generated that activates anaerobic glycolysis and causes this marker to rise.14,15 The main limitations of the study are its retrospective nature, the small number of cases in the study, and the fact that all patients were recruited in a single center.
Our study suggests that NSEPF and PEL measurements can predict response to pleurodesis in MPE. In patients with raised PEL (≥18cmH2O/l), the probability of pleurodesis failure is very high and, therefore, other therapeutic alternatives that may offer a chance of success should be considered for the prompt control of the patient's symptoms.
Please cite this article as: Ferreiro L, San José E, Gude F, Valdés L. Análisis del líquido y elastancia pleurales como predictores de respuesta a la pleurodesis en los derrames pleurales malignos. Arch Bronconeumol. 2018;54:163–165.