The increasing body of knowledge of chronic obstructive pulmonary disease acquired in recent decades has helped experts draw up more patient-focused treatment strategies. The latest versions of the Global Initiative for Chronic Obstructive Lung Disease (GOLD) document have moved toward a more multidimensional assessment, while in Spain, the assessment of COPD in the Spanish guidelines (GesEPOC) has evolved with the adoption of clinical phenotypes. Although both strategies have their advantages and disadvantages,1 they exemplify the progress made in patient classification as a step on the path toward personalized treatment.
This year has seen the publication of the new version of the GOLD 2017 proposal, which introduces important changes in several aspects of the disease.2 And now, this edition of Archivos de Bronconeumología publishes the executive summary of the major GesEPOC 2017 recommendations for the pharmacological treatment of stable COPD.3 This new version of the guidelines describes an approach with several features that bring it closer to the GOLD document, while maintaining its distinct philosophy in several areas.
In the diagnostic section, GesEPOC 2017 establishes a new patient classification, in which it replaces the term “severity”, measured by BODE–BODEx, with the concept of high or low risk. This is similar to the GOLD approach, but with 2 important differences. Firstly, a different cutoff point for dyspnea measured by the mMRC scale has been selected: GesEPOC defines the cutoff as more than 2 points for patients without treatment. This new cutoff point may have a lower degree of concordance with the CAT,4 but it has been proposed as a limit for identifying a patient with a high disease burden.5 Secondly, GesEPOC maintains lung function as a variable to be taken into account in the classification of risk. This difference with GOLD 2017 is a clear indication of the controversy surrounding the role of spirometry in monitoring patients. While it is true that lung function remains stable over time in some patients, and in these patients repeat spirometry during monitoring will provide little new prognostic information, spirometric data can be of value in patients with progressively deteriorating lung function. We understand by this that the need for regular lung function assessments could be personalized.
Another significant change in the diagnostic recommendations of GesEPOC is the incorporation of the asthma-COPD overlap (ACO). In this new version, GesEPOC adopts the term ACO as used in GOLD. However, diagnostic criteria have changed considerably since the previous version of GesEPOC, and are based on the SEPAR consensus published by the Asthma and COPD interest areas.6 Major and minor criteria that appeared in the previous version have been dropped, probably after the results of observational studies were examined.7 To date, GOLD 2017 has not yet published the annex mentioned in its section on ACO, so we do not know if the diagnostic approach will change.
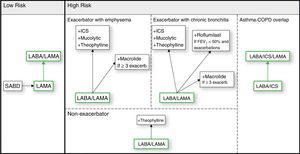
Regarding treatment, GesEPOC proposes a stepwise approach to the gradual intensification of therapy according to risk stratification and clinical phenotype. If we use the same type of figure that appears in the GOLD document, the GesEPOC treatment strategy would be similar to that of Fig. 1. We need to make 2 points here. Firstly, regarding the impact of long-term double bronchodilation, GesEPOC 2017, like GOLD 2017, proposes starting with one long-acting bronchodilator in patients with a low disease burden, and later stepping up to 2 bronchodilators. This approach makes good clinical sense, and for now it is the most logical approach. However, it should be remembered that disease onset is a good time to modify FEV1 decline,8 making it a window of opportunity for delaying functional progression of the disease. If it were confirmed that 2 bronchodilators could reduce FEV1 decline, the strategy should be reversed, and it would make more sense to administer 2 bronchodilators at the beginning of the disease.
Secondly, the current proposals assume that the indications for the use of inhaled corticosteroids are frequent exacerbations despite bronchodilator treatment and ACOS. This assumption needs to be clarified. First, the so-called persistent exacerbator,9 a patient who continues to present exacerbations despite receiving correct inhaled therapy, does not necessarily need inhaled corticosteroids. Several comorbidities have been identified that might influence the rate of exacerbations, and which cannot be treated with inhaled corticosteroids. GesEPOC 2017 establishes the use of inhaled corticosteroids as yet another option among a number of preventive treatments. It seems, then, that before debating the best way of scaling up to triple therapy,10 we should conduct a study on the factors that trigger these exacerbations, in order to determine the best preventive treatment. We also need to identify a marker for ACOS. GesEPOC 2017 proposes a definition based on the concomitant diagnosis of asthma, bronchoreversibility, and eosinophils in blood. The debate surrounding these criteria goes beyond the objectives of this editorial, but this proposal seems appropriate in the light of current knowledge. However, it must be mentioned that, while there is a correlation between these parameters and markers of response to inhaled corticosteroids, the degree of concordance between criteria associated with response to inhaled corticosteroids in individual patients is poor.7 We need, therefore, to identify candidate markers of response to these drugs that will allow us to take decisions at the patient level in the future.
Finally, other topics currently generating controversy in COPD should be addressed in the future, including, but not limited to, treatment of patients with few symptoms, classification of symptomatic patients with no spirometric obstruction, or the evaluation of comorbidities in patient management. Remembering rock and roll band Status Quo's hit “Cross that Bridge” from their 1988 studio album “Ain’t Complaining”, we will have to cross that bridge when we come to it, insofar as scientific progress permits. Meanwhile, the new GesEPOC 2017 document brings us closer to personalized, evidence-based treatment of COPD supported by a multidisciplinary committee of experts showing the best evidence available to date.
Please cite this article as: Lopez-Campos JL, Marquez-Martín E, Ortega-Ruiz F. Cambios mayores en la Guía española de la EPOC (GesEPOC) 2017: cruzando puentes. Arch Bronconeumol. 2017;53:291–292.