Multiple myeloma is the second most common cancer of the blood. Despite significant advances in treatment, it is considered an incurable disease, response to treatment is limited in duration, and the clinical course is generally relapsing. The combination of lenalidomide and dexamethasone is the therapeutic regimen of choice in relapse. Few cases of long-term lung toxicity presenting as organizing pneumonia in patients receiving this treatment have been reported in the literature. We report the case of a patient with a diagnosis of multiple myeloma, receiving lenalidomide, who developed dyspnea and pulmonary infiltrates. This is the first description of lenalidomide-induced lung toxicity in Spain.
This was a 64-year-old man, former smoker, who was diagnosed with IgA kappa multiple myeloma in 2012. He initially received 6 cycles of bendamustine, Velcade® and prednisone, in addition to autologous stem cell transplantation. In 2014, he relapsed, and began second-line treatment with monthly lenalidomide and dexamethasone.
In 2015, he was admitted on 3 occasions with a diagnosis of pneumonia, treated with empirical antibiotics and short-term tapering corticosteroids. At the time of the last admission, he presented with a clinical picture of dyspnea, low-grade fever, and cough. The patient was admitted to the ward with a diagnosis of community-acquired pneumonia; PCR for influenza A and B, and antigens in blood for Legionella and pneumococcus were negative. Empirical treatment was started with levofloxacin, corticosteroids, and oxygen therapy.
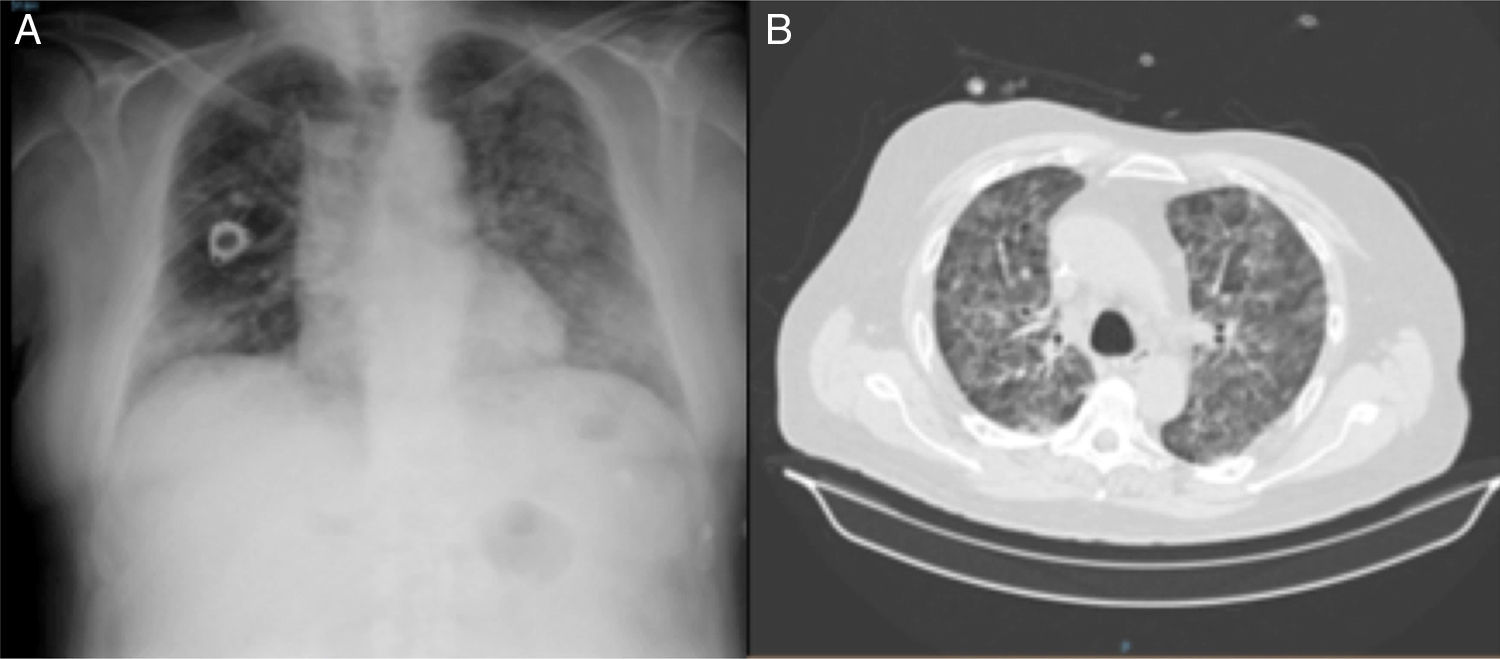
Seven days after admission, coinciding with discontinuation of steroid treatment, the patient's clinical situation worsened, and he developed severe respiratory failure, and bilateral pulmonary infiltrates on chest radiograph, so a high-resolution CT (HRCT) was performed, showing extensive bilateral, diffuse, ground glass involvement, with patchy septal thickening and dilated bronchi (Fig. 1).
Fiberoptic bronchoscopy with bronchoalveolar lavage (BAL) was performed, showing predominantly lymphocytic cellularity (with a CD4/CD8 ratio <0.9), and increased percentages of neutrophils and eosinophils. Microbiological cultures were negative. Transbronchial biopsy was not performed, given the worsening respiratory failure observed during the test.
In view of these findings, the patient's clinical records were reviewed, showing previous episodes of pneumonia and administration of chemotherapy.
The relationship between treatment administration and onset of symptoms, HRCT findings and BAL results, as well as the improvement in symptoms after reintroduction of corticosteroids, led us to a diagnosis of lenalidomide-induced interstitial lung disease.
Lenalidomide is a less toxic and more potent immunomodulatory agent than thalidomide. It is indicated in the treatment of multiple myeloma relapses in combination with dexamethasone, and in other hematological diseases. Only 7 cases of lenalidomide-induced lung toxicity have been described in the literature.1–4
The mechanism of action of the drug is based on inhibition of prostaglandin E (PGE2). If PGE2 synthesis of the fibroblasts is affected in the lung, leukotriene mitogen activity is activated, triggering fibroblast proliferation and collagen synthesis, leading to interstitial lung disease.2
Another pathway that may cause lung disease may be an immunological response similar to that which occurs in patients with hypersensitivity pneumonitis.
According to Zagouri et al.,5 the onset of symptoms occurs 3–5 months after starting treatment. In our patient, symptoms began to appear 1 year after the first dose of lenalidomide.
The treatment of choice is to discontinue the drug. The development of severe respiratory failure in our patient required the long-term use of corticosteroids.
In conclusion, while lenalidomide-induced lung toxicity is a rare adverse effect, it should be considered in the differential diagnosis of patients receiving this drug who show symptoms of interstitial disease, after other alternative causes of lung disease have been ruled out.
Our thanks to the Respiratory Medicine and Hematology Departments of the Hospital Universitario Virgen de la Victoria.
Please cite this article as: Cabrera César E, Fernández Aguirre MC, González Fernández A. Toxicidad pulmonar después del tratamiento con lenalidomida en un paciente con mieloma múltiple. Arch Bronconeumol. 2017;53:355–356.