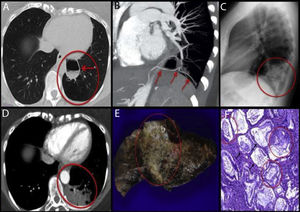
We report the case of a 29-year-old woman, former smoker, with congenital pulmonary airway malformation (CPAM) diagnosed previously using chest computed tomography (CT) (Fig. 1A). The malformation consisted of anomalous arterial vascularization (Fig. 1B), giving rise to a hybrid lesion: CPAM+pulmonary sequestration.
A: High-resolution CT, axial plane. Left lower lobe shows an area of destruction of the pulmonary parenchyma with less vascularity (red circle). The image of the larger cyst shows thickened walls and air-fluid level (arrow). B: Chest angio-CT scan with MIP and MPR reconstructions. An arterial branch is seen emerging from the celiac trunk (arrows) that supplies blood to the cystic mass and to the area of destruction. C: Chest X-ray, lateral projection. Consolidation seen in the left lower lobe (red circle), which in the clinical context of the patient was suggestive of pneumonia. D: Chest CT with intravenous contrast and imaging in arterial phase. The larger cystic lesion remains stable (arrow). Hypodense consolidation containing vessels seen within the CPAM (red circle). E: Macroscopic view of the surgical specimen. A whitish, indurated, condensation (red circle) is seen, extending through the pulmonary parenchyma in the form of nodules of variable size. F: Microscopic view of the lesion. Extensive architectural malformation with cystic areas (black arrows) associated with CPAM. Neoplastic proliferation of atypical mucinous epithelial cells covering the alveolar septa, with areas of pseudopneumonic pattern (red circles).
Three and a half years later, the patient presented in the emergency department due to fever, cough, and pain in the left hemithorax. Chest radiograph was performed (Fig. 1C), which revealed consolidation in the left lower lobe, which in the clinical context of the patient was suggestive of pneumonia. Given her persistent pain and dry cough after antibiotic treatment, we decided to expand the study with a chest CT (Fig. 1D), which showed consolidation within the CPAM. The differential diagnosis of this consolidation was determined to be either a bacterial superinfection of the CPAM or a neoproliferative process. Bacterial superinfection was the most probable diagnosis given the visualization of consolidation, accompanied by a concomitant predisposing lesion. A neoproliferative process, on the other hand, was less likely, taking into account the uncommon pattern of presentation and the young age of the patient. Due to the suspicion of superinfection and the high probability of recurrence, we decided to perform lower left lobectomy. The surgical specimen was sent for pathology study (Fig. 1E and F), which gave a definitive diagnosis of invasive mucinous adenocarcinoma in CPAM, with a pseudopneumonic pattern.
CPAMs form a heterogeneous group of cystic lesions and non-cystic lesions caused by early changes in the development of the pulmonary airway. The estimated incidence is 1/25000–35000 newborns.1 There are 5 subtypes, Type I being the most frequent and the one associated with the largest cysts.2 Most are supplied by blood from the pulmonary circulation, with the exception of hybrid lesions that may receive blood directly from the systemic arterial circulation.3
Malignant transformation has been reported in some cases of type I CPAM, a situation that is associated with a mutation in the K-ras gene. In 2013, Ishida et al. published a case report with a review of the literature,4 in which the authors described the fourth documented case to date of this type of CPAM-associated neoplasm with K-ras mutation. The case we describe also had the K-ras mutation. Radiological management is complex, since the similarity of this entity and bacterial pneumonia on imaging studies may delay diagnosis.5 Pseudopneumonic pattern of the tumor is associated with a worse prognosis, while treatment of choice is surgical resection.
Please cite this article as: Soler-Perromat A, Vollmer I, Ramírez J, Sánchez M. Adenocarcinoma mucinoso invasivo sobre malformación congénita de la vía aérea pulmonar (MCVAP): a propósito de un caso. Arch Bronconeumol. 2019;55:384–385.