Bronchiectasis patients may present a reduced functional capacity due to an increase in the ventilatory demand during exercise.
ObjectiveTo evaluate the effects of controlled voluntary hyperinflation and increased respiratory rate on the mechanics of the respiratory system, simulating what happens during exercise, in bronchiectasis and healthy subjects.
MethodsBronchiectasis (n=30) and healthy (n=16) subjects were evaluated by impulse oscillometry (IOS) during a baseline condition, and in controlled conditions with baseline (b) tidal volume (V) and hyperinflation (H), with respiratory rates at 30(R30) and 40(R40) bpm, in a random order. The mixed effects and a significance level at 0.05 were used for comparisons.
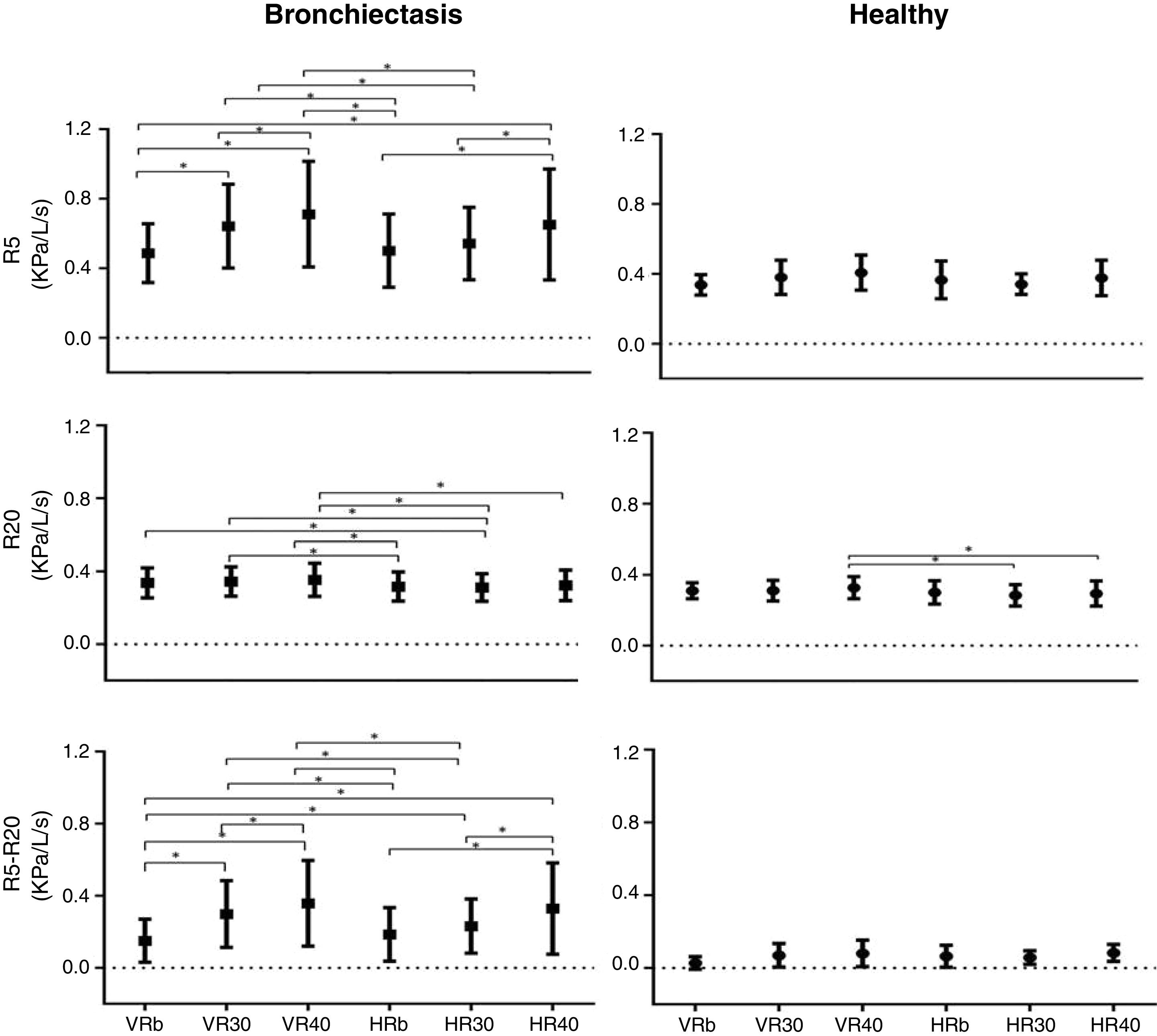
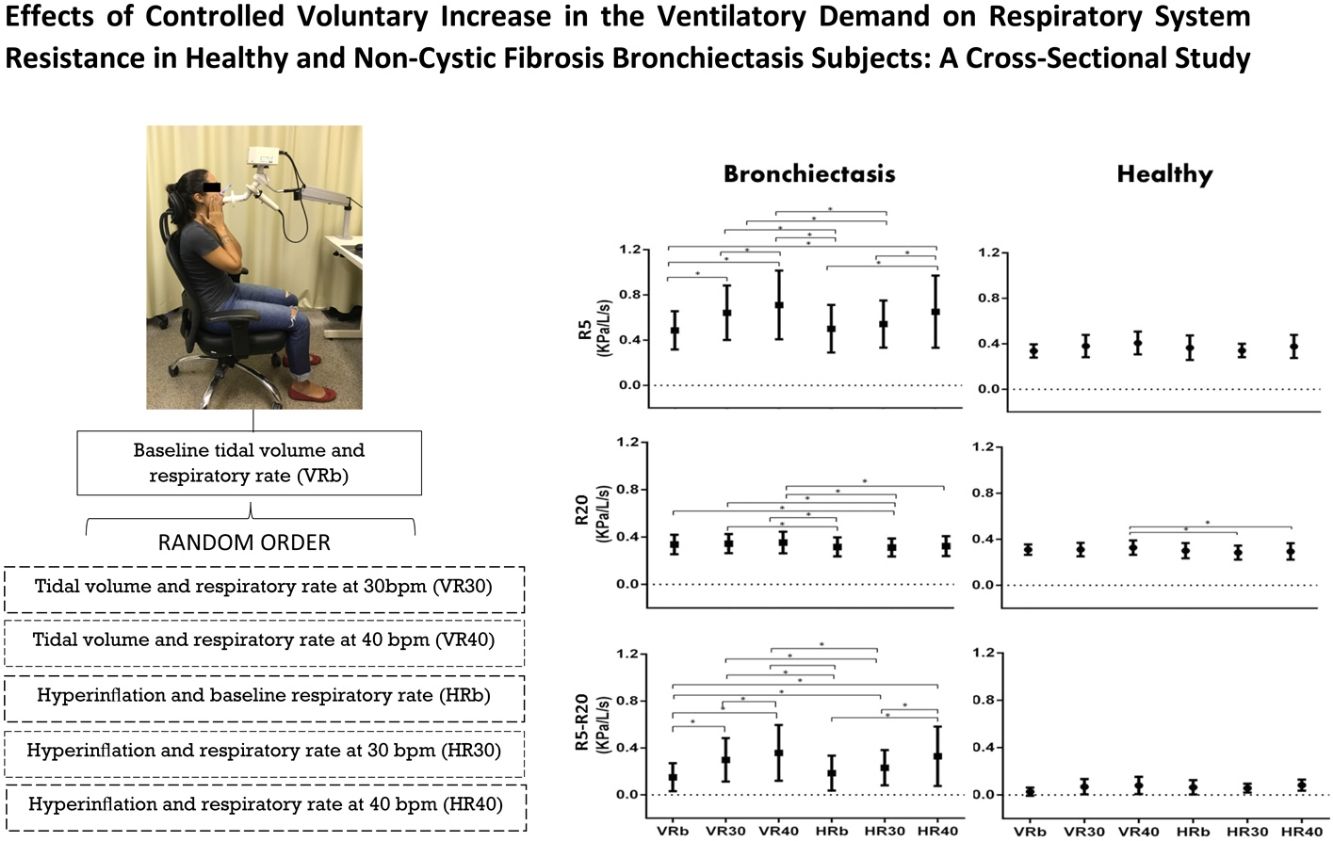
ResultsResistance at 5Hz (R5), and at minus 20Hz (R5–R20), in kPa/L/s, were higher in subjects with bronchiectasis in all experimental conditions (p<0.05). For the bronchiectasis group, R5 and R5-20 increased with R increase at V (VRb versus VR30 and VR40; VR30 versus VR40; R5, R20 and R5-20 increased with R increase at H (HRb versus HR40; HR30 versus HR40). For the same R, there was a decrease with H compared to V (HRb versus VR30 and VR40; and HR30 versus VR30 and VR40). For the healthy group, only R20 showed differences (HR30 versus HR40; HR40 versus VR40).
ConclusionThe tachypnea increases the resistance and reactance of the respiratory system in bronchiectasis patients, and the voluntary hyperinflation caused attenuates this increase. These results can guide the development of strategies to reduce the limitation of physical activity in patients with bronchiectasis.
Los pacientes con bronquiectasias pueden presentar una capacidad funcional reducida debido a un aumento en la demanda ventilatoria durante el ejercicio.
ObjetivoEvaluar los efectos de la hiperinsuflación voluntaria y controlada y el aumento de la frecuencia respiratoria en la mecánica del sistema respiratorio, simulando lo que sucede durante el ejercicio, en sujetos sanos y en pacientes con bronquiectasias.
MétodosSe evaluó a sujetos con bronquiectasia (n=30) y sujetos sanos (n=16) mediante la oscilometría de impulso (IOS) en situación basal, y en condiciones controladas con basal (b), volumen corriente (V) e hiperinsuflación (H), con frecuencias respiratorias a 30 (R30) y 40 (R40) bpm, en orden aleatorio. Para las comparaciones se utilizaron el modelo de efectos mixtos y un nivel de significación de 0,05.
ResultadosLa resistencia a 5Hz (R5) y la diferencia con 20Hz (R5-R20), medida en kPa/l/s, fue mayor en sujetos con bronquiectasias en todas las condiciones experimentales (p<0,05). Para el grupo de bronquiectasias, R5 y R5-R20 aumentaron con el aumento de R en V (VRb versus VR30 y VR40; VR30 versus VR40; R5, R20 y R5-R20 aumentaron con el aumento de R en H (HRb versus HR40; HR30 versus HR40. Para el mismo R, hubo una disminución de H en comparación con V (HRb versus VR30 y VR40; y HR30 versus VR30 y VR40). Para el grupo sano, solo R20 mostró diferencias (HR30 versus HR40; HR40 versus VR40).
ConclusiónLa taquipnea aumenta la resistencia y la reactancia del sistema respiratorio en pacientes con bronquiectasias, y la hiperinsuflación voluntaria generada atenúa este aumento. Estos resultados pueden guiar el desarrollo de estrategias para reducir la limitación de la actividad física en pacientes con bronquiectasia.
Bronchiectasis is a chronic inflammatory condition that causes permanent dilatation of the bronchi and bronchioles by destruction of structural components, resulting in decreased mucociliary clearance and increased airway susceptibility to bacterial colonization and infection.1
The diagnosis is traditionally performed using high resolution computed tomography (HRCT) in association with clinical signs and spirometry.2,3 Because spirometry requires great cooperation from subjects, the functional evaluation of patients with bronchiectasis can be complemented with impulse oscillometry (IOS), which provides compartmentalized information of the central and peripheral airways, during tidal breathing, with good sensitivity to identify changes in respiratory system resistance.4
Clinically, the most frequent characteristic of this disease is an increase in the production of bronchial secretions. The mucus produced is thicker and results in slower transport, causing damage to the mucociliary transport,5 and patients may benefit from specific physiotherapy techniques for the removal of secretions from the respiratory system,6 as well as overall exercises within pulmonary rehabilitation programs.7
The functional exercise capacity of patients with respiratory diseases, such as bronchiectasis, may be reduced by several factors, due to increase in ventilatory demand, which causes an overload to the respiratory muscles. During the exercise, these patients have an increase in the respiratory rate which induces a dynamic hyperinflation.
Because it is difficult to use some measurements during exercise, the literature describes some controlled experimental situations to simulate the increased ventilatory demand that occur during exercises, using voluntary controlled respiratory rate in COPD patients,8 and using voluntary controlled hyperinflation in subjects with restrictive pattern.9 However, no similar studies were found in patients with bronchiectasis, to identify how this exercise respiratory pattern can affect the respiratory system resistance and to propose respiratory control strategies that might enable less exhaustive exercise.
In this study, we evaluated the effects of controlled voluntary hyperinflation and increased respiratory rate on the mechanics of the respiratory system, simulating what happens during exercise, in bronchiectasis and healthy subjects.
MethodsIn this cross-sectional study, the sample size was calculated based on pilot study by G*Power (3.1.9.2) using the resistance at 5Hz (R5), resulting in 16 volunteers per group. The project was approved by the Human Research Ethics Committee of the Clinical Hospital of the Ribeirão Preto Medical School, University of São Paulo, CAAE 66047717.2.0000.5440.
There were included volunteers aged 18–70 years, diagnosed with bronchiectasis not due to cystic fibrosis, recruited at the pulmonology outpatient clinic of the Hospital das Clínicas of the Medical School of Ribeirão Preto, as well as healthy volunteers who were not active or ex-smokers without lung disease. Participants with COPD, asthma, lung cancer, and lobectomy, active or ex-smokers, and individuals with cardiovascular disease or decompensated metabolic, neuromuscular, or musculoskeletal diseases were excluded. All participants have signed the Informed Consent Form.
The medical diagnoses were based on clinical history, HRCT, and spirometry. In the Laboratory of Assessment Respiratory, they were evaluated by sociodemographic and anthropometric data, Bronchiectasis Severity Index (BSI) for assess the severity, ventilometry, spirometry, and impulse oscillometry system (IOS).
ProtocolAll the measurements were performed in a seated position.
The IOS were firstly performed using the subject normal breathing pattern, called baseline tidal volume and respiratory rate (VRb). After this, they were instructed to perform the following conditions, in a random order:
- -
Tidal volume and respiratory rate at 30bpm (VR30);
- -
Tidal volume and respiratory rate at 40bpm (VR40);
- -
In hyperinflation (at 50% of the inspiratory capacity) and baseline respiratory rate (HRb);
- -
In hyperinflation (at 50% of the inspiratory capacity) and respiratory rate at 30bpm (HR30);
- -
In hyperinflation (at 50% of the inspiratory capacity) and respiratory rate at 40bpm (HR40).
The hyperinflation was controlled using a ventilometer (FERRARIS Wrigth Mark) and to control the respiratory rate the volunteers were instructed to follow the sound of a metronome (Metrônomo n-Track (versão 1.2.1. © n-Track Software).
SpirometryAll patients underwent spirometry to detect respiratory changes using the Jaeger MasterScreen spirometer (Jaeger Co, Wurzburg, Germany), according to the ATS/ERS guidelines.10 The following variables were analyzed: forced vital capacity (FVC), forced expiratory volume in one second (FEV1), FEV1/FVC and mean forced expiratory flow (FEF25–75%).
Impulse oscillometryImpulse oscillometry was performed by using a Jaeger impulse oscillometry system (Jaeger, Wurzburg, Germany) calibrated daily for volume and pressure by using a 3L syringe with a reference resistance of 0.2kPa/L/s. A freeflow mouthpiece was used to minimize the effect of malposition of the tongue.11 The mouthpiece contains a depressor to keep the tongue on the floor of the mouth, stabilize it, and reduce oral resistance. Pressure pulses were applied to the airways 5 times per second for at least 60s while the subject was breathing at the tidal volume level. The pulses were performed to calculate the mean of 3 technically acceptable measurements.
Parameters were calculated by using frequencies between 5 and 35Hz, including the resistance at 5Hz (R5) or total respiratory system resistance; resistance at 20Hz (R20) or central resistance; R5–R20 or peripheral resistance; reactance at 5Hz (X5), which reflects the combined effect of tissue elastance and inertance; reactance area (AX), at which is a useful index related to respiratory compliance and therefore of small airways patency.12,13
Data analysisFor statistical analysis, the model with mixed effects, using the R Core Team software (Vienna, Austria, 2016), SAS Statistical Software (version 9.3, SAS Institute, Inc. Cary, NC) were used to compare baseline and experimental conditions, with a significance level set at p<0.05.
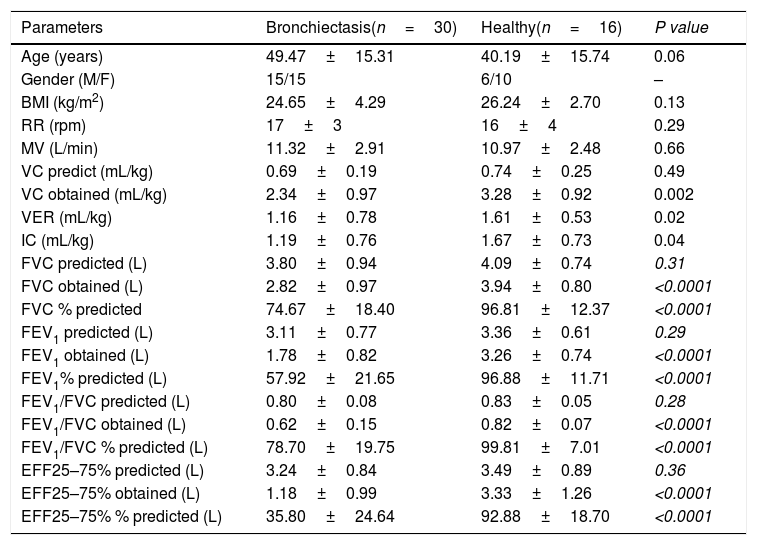
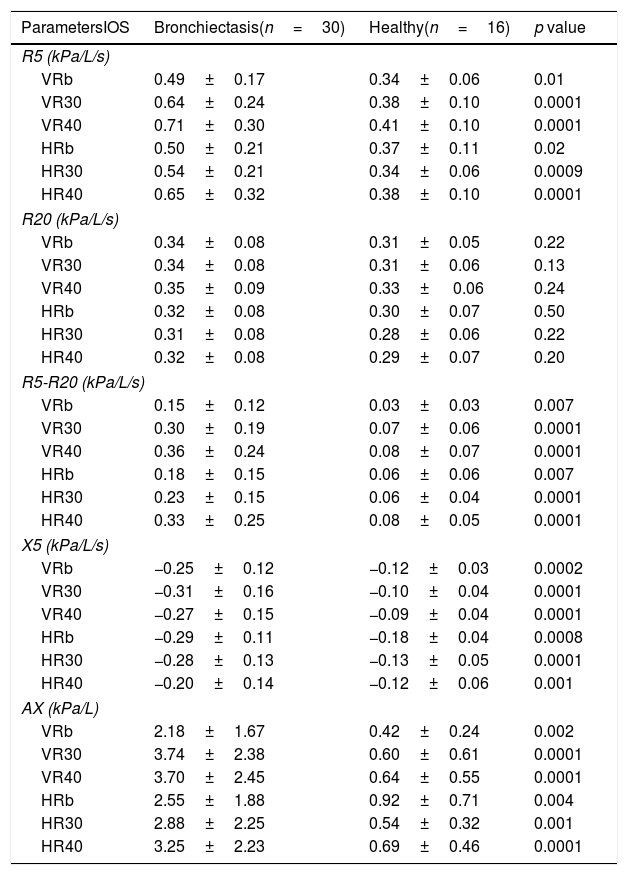
ResultsThe anthropometric and evaluation data of the subjects are described in Table 1. In the BSI score the patients were classified as intermediate, the mean of the group is 5 points. The values obtained for R5 and R5-20 were higher in subjects with bronchiectasis in all experimental conditions (Table 2).
Basal data of subjects.
| Parameters | Bronchiectasis(n=30) | Healthy(n=16) | P value |
|---|---|---|---|
| Age (years) | 49.47±15.31 | 40.19±15.74 | 0.06 |
| Gender (M/F) | 15/15 | 6/10 | – |
| BMI (kg/m2) | 24.65±4.29 | 26.24±2.70 | 0.13 |
| RR (rpm) | 17±3 | 16±4 | 0.29 |
| MV (L/min) | 11.32±2.91 | 10.97±2.48 | 0.66 |
| VC predict (mL/kg) | 0.69±0.19 | 0.74±0.25 | 0.49 |
| VC obtained (mL/kg) | 2.34±0.97 | 3.28±0.92 | 0.002 |
| VER (mL/kg) | 1.16±0.78 | 1.61±0.53 | 0.02 |
| IC (mL/kg) | 1.19±0.76 | 1.67±0.73 | 0.04 |
| FVC predicted (L) | 3.80±0.94 | 4.09±0.74 | 0.31 |
| FVC obtained (L) | 2.82±0.97 | 3.94±0.80 | <0.0001 |
| FVC % predicted | 74.67±18.40 | 96.81±12.37 | <0.0001 |
| FEV1 predicted (L) | 3.11±0.77 | 3.36±0.61 | 0.29 |
| FEV1 obtained (L) | 1.78±0.82 | 3.26±0.74 | <0.0001 |
| FEV1% predicted (L) | 57.92±21.65 | 96.88±11.71 | <0.0001 |
| FEV1/FVC predicted (L) | 0.80±0.08 | 0.83±0.05 | 0.28 |
| FEV1/FVC obtained (L) | 0.62±0.15 | 0.82±0.07 | <0.0001 |
| FEV1/FVC % predicted (L) | 78.70±19.75 | 99.81±7.01 | <0.0001 |
| EFF25–75% predicted (L) | 3.24±0.84 | 3.49±0.89 | 0.36 |
| EFF25–75% obtained (L) | 1.18±0.99 | 3.33±1.26 | <0.0001 |
| EFF25–75% % predicted (L) | 35.80±24.64 | 92.88±18.70 | <0.0001 |
Data expressed as mean and standard deviation; BMI: body mass index; M: man; F: female: RR: respiratory rate; MV: minute volume; TV: tidal volume; VC: vital capacity; VER: volume of expiratory reserve; IC: inspiratory capacity; FVC: forced vital capacity; FEV1: forced expiratory volume in first second; FEV1/FVC: ratio of forced expiratory volume in first second to forced vital capacity; EFF25–75%: expiratory mean forced flux.
IOS measurements in different breathing patterns.
| ParametersIOS | Bronchiectasis(n=30) | Healthy(n=16) | p value |
|---|---|---|---|
| R5 (kPa/L/s) | |||
| VRb | 0.49±0.17 | 0.34±0.06 | 0.01 |
| VR30 | 0.64±0.24 | 0.38±0.10 | 0.0001 |
| VR40 | 0.71±0.30 | 0.41±0.10 | 0.0001 |
| HRb | 0.50±0.21 | 0.37±0.11 | 0.02 |
| HR30 | 0.54±0.21 | 0.34±0.06 | 0.0009 |
| HR40 | 0.65±0.32 | 0.38±0.10 | 0.0001 |
| R20 (kPa/L/s) | |||
| VRb | 0.34±0.08 | 0.31±0.05 | 0.22 |
| VR30 | 0.34±0.08 | 0.31±0.06 | 0.13 |
| VR40 | 0.35±0.09 | 0.33± 0.06 | 0.24 |
| HRb | 0.32±0.08 | 0.30±0.07 | 0.50 |
| HR30 | 0.31±0.08 | 0.28±0.06 | 0.22 |
| HR40 | 0.32±0.08 | 0.29±0.07 | 0.20 |
| R5-R20 (kPa/L/s) | |||
| VRb | 0.15±0.12 | 0.03±0.03 | 0.007 |
| VR30 | 0.30±0.19 | 0.07±0.06 | 0.0001 |
| VR40 | 0.36±0.24 | 0.08±0.07 | 0.0001 |
| HRb | 0.18±0.15 | 0.06±0.06 | 0.007 |
| HR30 | 0.23±0.15 | 0.06±0.04 | 0.0001 |
| HR40 | 0.33±0.25 | 0.08±0.05 | 0.0001 |
| X5 (kPa/L/s) | |||
| VRb | −0.25±0.12 | −0.12±0.03 | 0.0002 |
| VR30 | −0.31±0.16 | −0.10±0.04 | 0.0001 |
| VR40 | −0.27±0.15 | −0.09±0.04 | 0.0001 |
| HRb | −0.29±0.11 | −0.18±0.04 | 0.0008 |
| HR30 | −0.28±0.13 | −0.13±0.05 | 0.0001 |
| HR40 | −0.20±0.14 | −0.12±0.06 | 0.001 |
| AX (kPa/L) | |||
| VRb | 2.18±1.67 | 0.42±0.24 | 0.002 |
| VR30 | 3.74±2.38 | 0.60±0.61 | 0.0001 |
| VR40 | 3.70±2.45 | 0.64±0.55 | 0.0001 |
| HRb | 2.55±1.88 | 0.92±0.71 | 0.004 |
| HR30 | 2.88±2.25 | 0.54±0.32 | 0.001 |
| HR40 | 3.25±2.23 | 0.69±0.46 | 0.0001 |
Data presented in mean±standard derivation.
R5: resistance to 5Hz; R20: resistance to 20Hz; R5-R20: peripheral resistance.
VRb: tidal volume and basal frequency; VR30: tidal volume and respiratory rate of 30 breaths/min; VR40: tidal volume and respiratory rate of 40breaths/min; HRb: hyperinflated and respiratory rate basal; HR30: Hyperinflated and respiratory rate of 30breaths/min; HR40: Hyperinflated and respiratory rate of 40breaths/min.
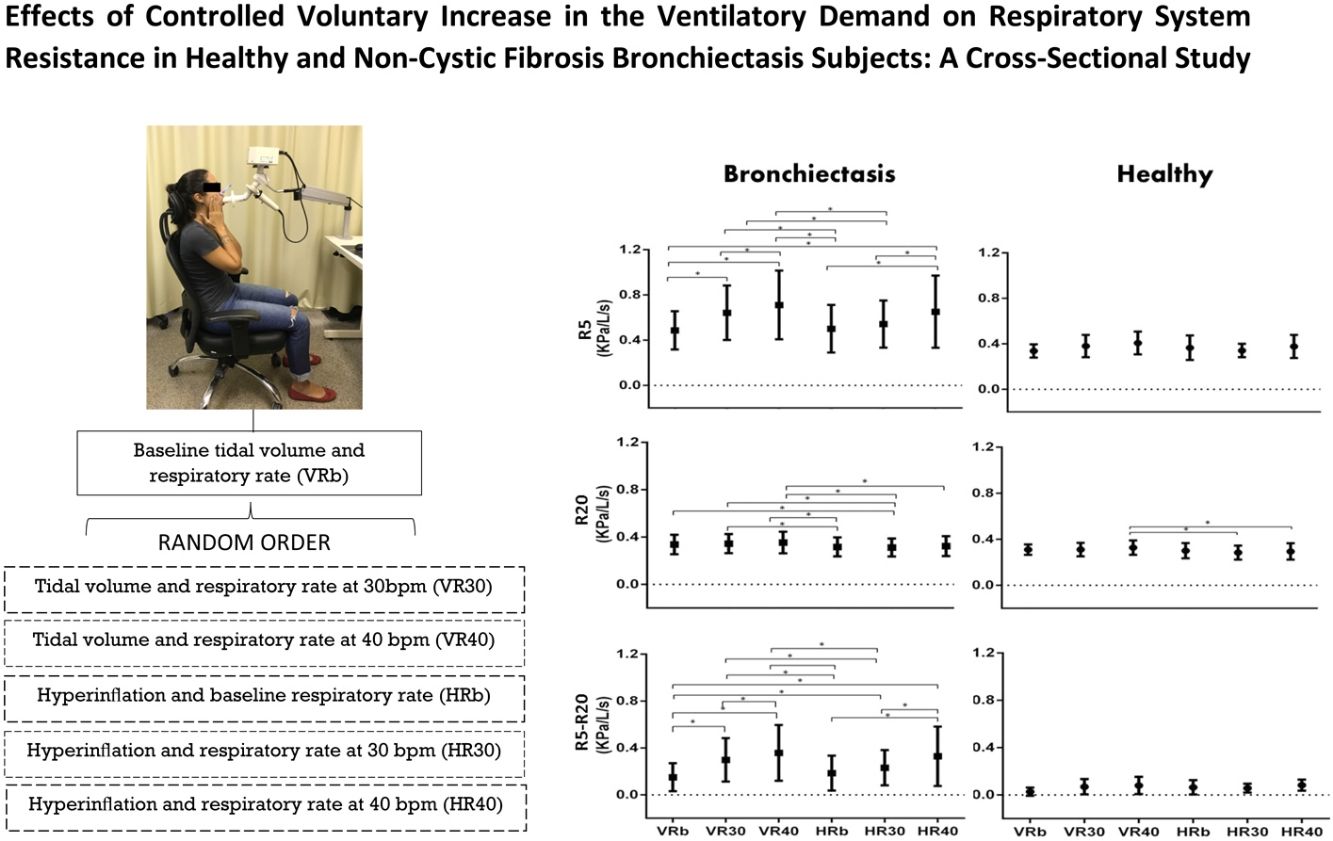
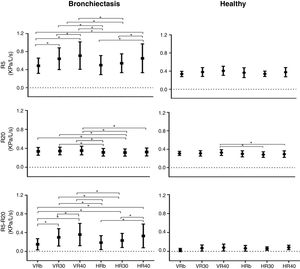
In the bronchiectasis group, R5 (kPa/L/s) showed differences between VRb versus VR30 (p=0.0001), VR40 (p=0.0001), and HR40 (p=0.0001); VR30 versus VR40 (p=0.02); HRb versus HR40 (p=0.0001); HR30 versus HR40 (p=0.0005); HRb versus VR30 (p=0.0001), and VR40 (p=0.0001); and HR30 versus VR30 (p=0.001) and VR40 (p=0.0001).
For R20 (kPa/L/s), there were differences between HRb versus VR30 (p=0.01), and VR40 (p=0.001); HR30 versus VRb (p=0.02), VR30 (p=0.005), and VR40 (p=0.0004); and HR40 versus VR40 (p=0.009).
In R5-R20 (kPa/L/s), there were differences between VRb versus VR30 (p=0.0001), and VR40 (p=0.0001); VR30 versus VR40 (p=0.0001); HRb versus HR40 (p=0.0001); HR30 versus HR40 (p=0.0001); VRb versus HR30 (p=0.0008), and HR40 (p=0.0001); HRb versus VR30 (p=0.0001), and VR40 (p=0.0001); and HR30 versus VR30 (p=0.005), and VR40 (p=0.0001) (Fig. 1, Table 2).
In the healthy group, only R20 (kPa/L/s) showed a difference when compared the HR30 versus HR40 (p=0.007); and HR40 versus VR40 (p=0.004) (Fig. 1, Table 2).
DiscussionOur results demonstrated that an increased respiratory rate promotes increases in the total, central and peripheral resistances, in subjects with bronchiectasis and an increase in the central resistance in healthy subjects. When they were instructed to perform a voluntary controlled hyperinflation, the high resistance and reactance, induced by the respiratory rate increases was attenuated, close to the baseline values, in the bronchiectasis group. Comparing bronchiectasis to the healthy group, there was an increase in the total and peripheral resistance of the respiratory system in all experimental conditions, which could be explained to the more compromised peripheral airways in the subjects’ with bronchiectasis.14
People with bronchiectasis experience reduction in both exercise capacity and health-related quality of life.15 Reduction in exercise capacity has been associated with structural alterations to lung tissue, progressive airflow obstruction, dyspnea secondary to dynamic hyperinflation, and psychological morbidity.15–17
In general, dynamic hyperinflation is associated with a mechanically disadvantageous position for the respiratory muscles, causing increased respiratory work and exercise limitation.18 Nevertheless, the hyperinflation mechanism, as the increase in the lung volume, promotes a reduction in the airways resistance because the bronchi are supported by the radial traction of surrounding lung tissue, and their caliber increases as the lung expands.19
The use of strategies to reduce respiratory work during exercise is widespread, for example, the pursed-lips breathing. This maneuver maintains positive expiratory pressure on the airways, increasing alveolar pressure and central displacement of the point of equal pressure, avoiding alveolar collapse.20
However, the effects of pursed-lips breathing during exercise capacity are still controversial. Studies with COPD showed a reduction in dynamic hyperinflation and an increase in SpO2, in the endurance and incremental tests, after the use of a bronchodilator plus pursed-lips breathing.21 Another study using pursed-lips breathing without bronchodilator, reported a reduction of dynamic hyperinflation in the Glittre test, but not in the six-minute walk test distance.22
Physical activities and exercises are recommended for patients with chronic respiratory diseases because they are associated with clinical patient outcomes improvements. Pulmonary rehabilitation programs for people with bronchiectasis, that increases the ventilator demand during aerobic exercises, aims to improve exercise capacity, through effects on aerobic capacity and peripheral muscle, as well as to enhance disease management and improve quality of life.7 Programs of pulmonary rehabilitation for stable patients have clinically significant benefits but the improvements in exercise capacity and healthy related quality of life are of a short duration. Further exploration to identify the effects of these interventions on disease severity and optimal approaches to maintain positive outcomes are needed.23
Our findings have shown that hyperinflation is not detrimental when we observe it effects on airway resistance. In our protocol, hyperinflation was beneficial even without the use of the bronchodilator, reducing resistance values that were elevated by increased respiratory rate. Based on these results, we hypothesize that, during exercise, individuals with increased resistance values may benefit from voluntary control of respiratory frequency and volume of exhaled air, and that this strategy could contribute to clinical improvements in dyspnea.
The exertional dyspnea for people with respiratory diseases is usually multifactorial in origin, partly reflecting peripheral muscle dysfunction, the consequences of dynamic hyperinflation, increased respiratory load, or defective gas exchange.24–26 Although dyspnea is not the primary symptom of bronchiectasis, a reduction in dyspnea may partially contribute to the improvement in the exercise capacity and healthy related quality of life.23
In this study we had a controlled situation, without other adaptations to exercise, and we need to know if these physiological strategies could contribute to reduce the exercise limitation in bronchiectasis patients. It is important to identify strategies that may improve exercise tolerance, which need to be evaluated during exercise in patients with bronchiectasis, contributing to a reduction of sedentary behavior and an improvement in the quality of life. It is also important to identify whether other patients with increased respiratory system resistance, such as asthma sufferers for example, may also benefit, since there was a decrease in central resistance in subjects with bronchiectasis and in subjects without lung disease. Because the evaluation occurred in a controlled experimental setting, it is necessary to know if these results could be reproduced during exercises for aerobic training.
In conclusion, the tachypnea increases the resistance and reactance of the respiratory system in bronchiectasis patients, and the voluntary hyperinflation caused attenuates this increase. These results can guide the development of strategies to reduce the limitation of physical activity in patients with bronchiectasis
FundingThis work was supported by São Paulo Research Foundation (FAPESP process numbers 2016/20077-6), and National Council for Scientific and Technological Development (CNPq). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES).
Conflicts of interestThe authors declare no conflicts of interest.
The authors wish to thank the São Paulo Research Foundation for their financial support and Laboratory of Assessment Respiratory for equipment available for this study.