To the Editor: The Mycobacterium tuberculosis complex includes the species M. tuberculosis, M. bovis, M. africanum, M. microti and M. canetti. While M. africanum is the cause of more than 60% of pulmonary tuberculosis cases in central Africa, it is rare in the European context.1-6
We report a case of disseminated tuberculosis in a 78-year-old Caucasian male smoker suffering from chronic obstructive pulmonary disease and prostatism. In the four months preceding hospital admission, his prostate symptoms had become sufficiently acute as to prevent him from sleeping. He therefore consulted a urologist, who prescribed two cycles of treatment that were unsuccessful. At the time of his admission, in addition to prostatism, the patient also exhibited anorexia, asthenia, and cough with yellowish sputum. Physical examination revealed a fever of 38.2 ºC and a generally deteriorated physical state.
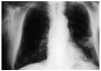
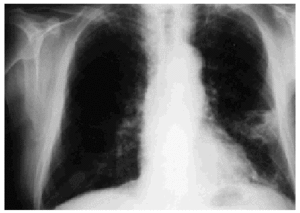
Noteworthy lab test results were as follows: an erythrocyte sedimentation rate of 48 mm/h, creatinine 3.70 mg/dL, urea 231 mg/dL, and serum gamma glutamyl-transferase 303 IU/L. A chest x-ray revealed irregular opacities in both apices and a cavitated infiltrate in the lower left lobe (Figure 1).
Fig. 1. Chest x-ray showing cavitated pulmonary infiltrate in the lower left lobe.
Innumerable leukocytes per field were observed in the urine sediment, with cultures in the usual media yielding negative results. Acid-fast bacteria tests of sputum and urine were positive (>50 resistant acid-fast bacilli per line), which permitted a diagnosis of active pulmonary and renal tuberculosis. A nonchromogenic bacterium that displayed slow dysgonic growth was isolated from the three sputum cultures. It was negative for nitrate and catalase reduction at 68ºC. Tests for urease, nicotinamidase, and pyrazinamidase activities, and for susceptibility to thiophen-2-carboxylic acid hydrazide were positive. The strain was delivered to the Spanish National Center of Microbiology in Majadahonda for identification and determination of antibiotic sensitivity. It was identified as M. africanum, susceptible to isoniazid, rifampin and pyrazinamide. Urine cultures for mycobacteria proved to have been contaminated. A treatment regimen of 2 months with rifampin, isoniazid, and pyrazinamide and 7 months with rifampin and isoniazid was initiated. The patient's progress was satisfactory, and sputum and urine cultures were negative at the end of treatment.
M. africanum was first described in 1968 and biochemically variant strains have been found in different regions of Africa.3,5,6 It has rarely been isolated in Spain and in most such cases it has been found in immigrants or in Spaniards who have traveled to Africa.1,2 The patient in the present case had taken part in armed conflict in North Africa and had worked on a cargo ship, where he may have been infected. In the epidemiological study of his contacts no further cases were detected.
Signs and symptoms caused by M. africanum are indistinguishable from those produced by any other mycobacterium of the M. tuberculosis complex.1-3,5 Precise identification is of some interest from an epidemiological standpoint, given that it allows the detection of strains and outbreaks and the application of the most appropriate control measures. Disseminated tuberculosis caused by this mycobacterium is rare in Spain, and although we were unable to isolate it from urine, the combination of acid-fast bacteria smear tests, clinical presentation, and the patient's progress indicate dissemination in this particular case.
In conclusion, we wish to highlight the need for proper identification of this species and call attention to the ability of M. africanum to cause disseminated infections.