Carcinoid tumours of bronchial origin are rare in type 1 multiple endocrine neoplasia (MEN1) syndrome. The prevalence of histologically confirmed cases is approximately 5–8%, although in more recent studies it is estimated that it could be much higher and a possible relationship with the presence of hypergastrinaemia is suggested. We report a patient with a type 1 MEN syndrome, with no respiratory symptoms, with hypergastrinaemia, and in whom a 5 mm diameter nodule was detected in the wall of the left main bronchus by computed tomography. The bronchial biopsy confirmed that it was a typical bronchial carcinoid and the octreoscan showed a single focus of high uptake coinciding with this lesion. A bronchoplastic (sleeve) was performed with extirpation of 3 bronchial rings, which also demonstrated that it was a typical carcinoid.
Los tumores carcinoides de origen bronquial son raros en el síndrome de neoplasias endocrinas múltiples tipo 1 (MEN1). La prevalencia de casos con confirmación histológica es aproximadamente del 5%–8%, aunque en estudios más recientes se estima que podría ser más alta y, en ellos, se contempla una posible relación con la presencia de hipergastrinemía. Aportamos una paciente con síndrome de MEN1, sin síntomas respiratorios, con hipergastrinemia, y que en una tomografía computarizada se detectó un nodulo de 5 mm de diámetro en la pared del bronquio principal izquierdo. La biopsia bronquial confirmó que se trataba de un carcinoide bronquial típico y el Octeoscran® mostró un único foco de hipercaptación coincidente con esta lesión. Se realizó una broncotomía con extirpación de 3 anillos bronquiales, que también demostró que se trataba de un carcinoide típico.
Multiple endocrine neoplasia type 1 (MEN1) syndrome is transmitted as an autosomal dominant mutation of a suppressor gene located on chromosone 11q13. The sequencing of this gene identified more than 250 mutations but with no correlation with the phenotypic expression.1
MEN1 syndrome carcinoid tumours (CT) are located primarily in the gastrointestinal tract and infrequently in the thymus and lung.2 Bronchopulmonary prevalence is 5% although more recent studies have found that it can reach 38%. These studies suggest a relationship with the levels of gastrin in the blood.2,3
The role of gastrin in producing bronchial CT is believed to lie in the stimulation of bronchial neuroendocrine cells inducing hyperplasia, dysplasia and malignancy. A recent study found that hypergastrinemia was more frequent in patients with MEN1 syndrome and pulmonary nodules.2
The following is a report on a patient with MEN1 syndrome who presented a typical bronchial carcinoid that was detected early and had the peculiarity of being associated with hypergastrinemia.
Clinical observationThe patient was a 53-year-old female, with no toxic habits, who was diagnosed with MEN1 syndrome in 1990. The family genetic study confirmed involvement of the father and several brothers, and showed that the patient carried the molecular mutation of the MEN-1 gene (11q13) in exon 2360 insTG. Symptoms began at the age of 40 with hypoglycemia secondary to insulinoma, which was treated with resection of 80% of the pancreas. Subsequently, the patient presented primary hyperparathyroidism, which was treated in 2004 by total parathyroidectomy with reimplantation of parathyroid tissue in the supinator muscle of the right arm. Furthermore, secondary hyperprolactinemia was detected secondary to pituitary microadenoma and hypergastrinemia. Several years earlier she had had a lipoma removed from her armpit and a cutaneous neurofibroma removed from her right shoulder.
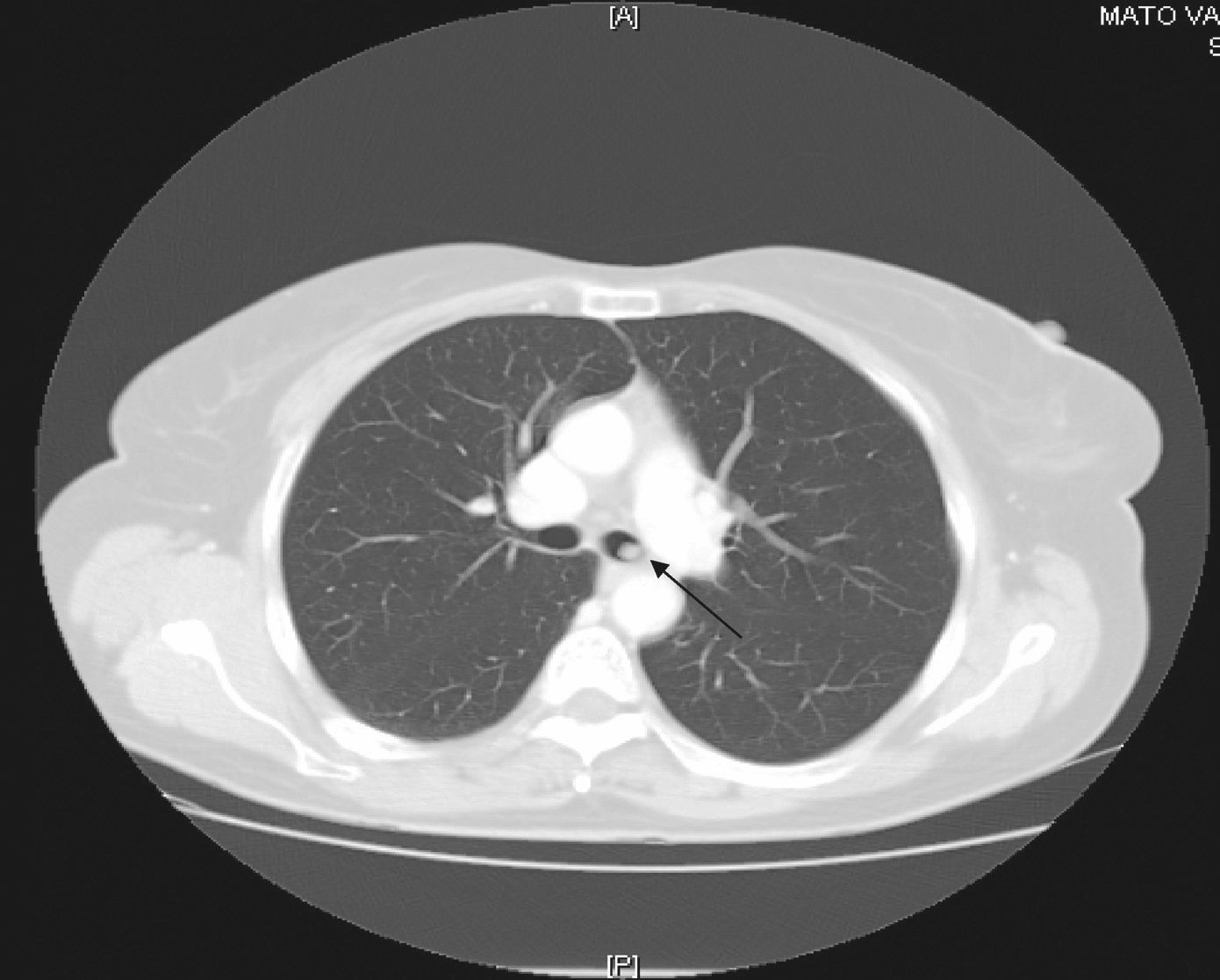
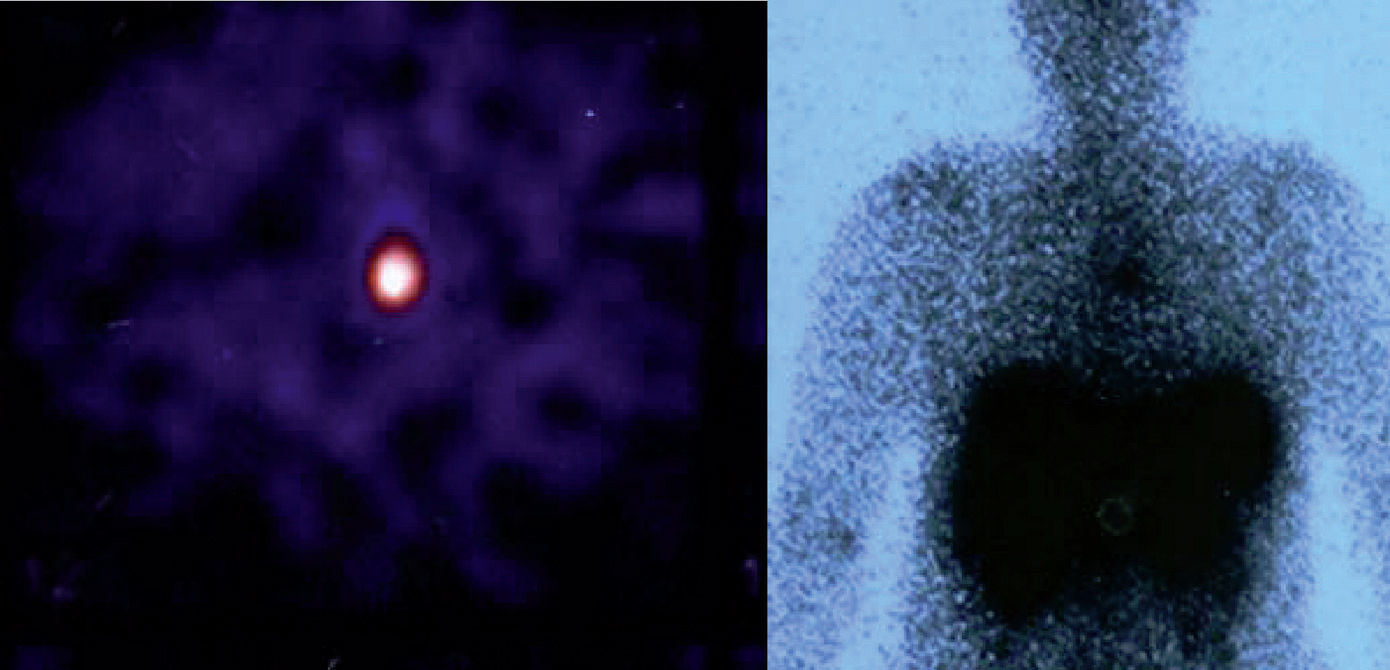
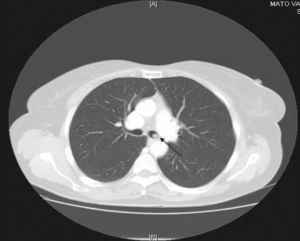
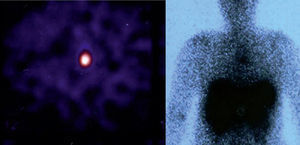
Since then, blood test have been carried out to monitor her, and on occasions imaging studies performed. During the last follow-up, blood tests were normal and gastrin levels were 717 pg/ml (VN<100) and chromogranin A was 340 ng/ml (VN 19.4–98.1). Thyroid hormones were normal. Calcium in urine for 24 hours was normal and 5-hydroxy-indoleacetic acid was 4.35 mg (VN 2.0–9.0). From 2000 to 2008, several measurements of blood gastrin were made and their values ranged from 700 to 900 pg. Thoracoabdominal tomography revealed the presence of a 5mm-diameter nodule in the left main bronchus (LMB) (fig. 1). Given this finding, a radiolabeled octreotide scintigraphy (octreoscan) was performed, which showed a focus with high uptake at the level of the mediastinum (fig. 2) coincident with the lesion observed on the CT. Bronchoscopy confirmed the existence of a clearly defined and highly vascular tumour in the LMB wall, more than 2 cm from the main carina. The bronchial biopsy and immunohistochemical studies confirmed that it was a typical carcinoid.
Pulmonary function was normal and in April 2008 resection of 3 LMB rings was performed with termino-terminal anastomosis. The histological study also confirmed the presence of a typical carcinoid.
DiscussionMEN1 syndrome or Wermer syndrome includes parathyroid, pancreatic and pituitary neoplasms and, less frequently, anterior intestine carcinoids, pheochromocytomas, visceral or subcutaneous lipomas, angiofibromas and cutaneous collagenomas.1 It is inherited in an autosomal dominant manner and occurs as a consequence of “inactivating” mutations in a supposed suppressor gene (MEN-1) located on chromosone 11q13, which encodes a protein of 610 amino acids, called menin. This protein functions as a regulator of transcription, although its exact role is unknown.1,2
Bronchopulmonary neuroendocrine tumours represent 20% of pulmonary neoplasms and arise from neuroendocrine cells of the bronchial epithelium.3 The World Health Organisation classifies them into four subgroups: typical carcinoid tumour (low-grade malignancy), atypical carcinoid tumour (intermediate-grade malignancy), and two high-grade neoplasms: large-cell neuroendocrine carcinoma and small-cell neuroendocrine carcinoma. The first two subtypes are encompassed as carcinoid tumours.3,4
The typical carcinoid accounts for 80–90% of sporadic carcinoid tumours5 and is also the most frequent in this syndrome although atypical carcinoids with high potential for malignancy in the mediastinum and lung have also been reported.2,6 Presentation occurs most often in patients in their 50s and 60s but it can also appear in children and is the most frequent pulmonary neoplasm in childhood. When associated with MEN1 syndrome, there is a preponderance of women and it occurs at younger ages.2
Unlike other pulmonary neoplasms, these are not related to smoking.3,5 They generally occur in isolation, however, as occurred in this case, they can be observed within the MEN1 syndrome. Several cases within the same family have also been reported in which there were no genetic alterations nor MEN syndrome,7,8 and additionally, some genetic mutations of the MEN1 syndrome have been observed in patients with sporadic carcinoid and in large-cell neuroendocrine carcinoma, the significance of which is still unknown.9,10
Most typical CTs have a central location while the atypical CTs may be central or peripheral and tend to be larger.5 Nodal metastases are more common in atypical carcinoids (57 vs. 13%5) and also in the liver, bone and central nervous system (21 vs. 3%). The differential diagnosis between typical and atypical carcinoid is a challenge for the pathologist,11 one that is very important for deciding the type of resection during surgery.
Symptoms related to the release of serotonin (carcinoid syndrome) are rare in these tumours.3,5 A differential hormonal characteristic of the carcinoid associated with MEN1 syndrome is that secretion of serotonin by the tumour has not been reported.12,13
The diagnosis of CT, as well as the serum markers such as chromogranin A, and urinary acids such as 5-hydroxy-indoleacetic acid, is performed with imaging tests. CT scan, MRI, PET and scintigraphy with octreotide (somatostatin analog labeled by octreoscan)6,14 are important for localisation and the extension study. Eighty percent of bronchopulmonary CTs express somatostatin receptors (SST2) and thus the sensitivity of the octreoscan in carcinoid tumours is 73–89%, but it shows false positives for granulomas, thyroid diseases and areas with aggregates of activated lymphocytes, thus requiring confirmation of the octreoscan findings through imaging and biopsy.
Curative treatment of CT is through surgery. In patients with typical carcinoids, another therapy option is endoscopic treatment with Nd-YAG laser and cryotherapy, but this therapeutic option should be reserved for patients in whom surgery is not feasible.15 However, in patients with MEN1 syndrome who present tumours in other locations, this treatment would be more justified than in patients with sporadic carcinoids.
The typical CT prognosis is good, with survival rates at 5 years for 87–89%, although there may be late recurrences16 and development of another second metachronous carcinoid tumour.17 We do not know if early diagnosis by imaging techniques is important in the prognosis but we do know that in 129 patients the CT did not have a worse prognosis, although further studies are needed in this area.2
As a conclusion to this case, we emphasise the rarity of a bronchial carcinoid tumour in MEN 1 syndrome, its association with hypergastrinemia and the importance of imaging techniques for early diagnosis.