Since 1972, when Rick Demont lost his gold medal in Munich for using banned medication, anti-doping legislation in the field of asthma and sport has gradually become more fit-for-purpose, simplifying the lives of athletes with asthma, and consequently those of their doctors. Since January 2013, asthma can be treated without any significant restrictions, and the requirement to request permission from official sporting bodies has been lifted.1,2 However, the regulations must be read carefully, because authorizations for use are based on the administration of therapy as stipulated. Section S3 of the regulations reads as follows3: “All selective and non-selective beta-2 agonists and all their optical isomers are prohibited. These include, but are not limited to: fenoterol; formoterol; higenamine; indacaterol; olodaterol; reproterol; salbutamol; salmeterol; terbatuline; vilanterol. Exceptions: inhaled salbutamol: maximum 1,600 mcg over 24h, without exceeding 800 mcg over 12h; inhaled formoterol: maximum delivered dose 54 mcg over 24h; inhaled salmeterol: maximum 200 mcg over 24h. This corresponds to higher dose ranges than the therapeutic doses of the 3 products. The presence in urine of a concentration of salbutamol higher than 1000 nanograms per milliliter or formoterol higher than 40 nanograms per milliliter will be assumed to reflect use of the substance for non-therapeutic intentions and will be considered an adverse analytical finding (AAF), unless the athlete proves, in a controlled pharmacokinetic study, that this adverse finding was the consequence of the use a therapeutic dose (by inhalation) at the maximum dose indicated above”. That means, therapeutic doses of salbutamol, salmeterol and/or formoterol are permitted alone or in combination with any inhaled corticosteroids available on the pharmaceutical market. In these cases, no Therapeutic Use Exemption (TUE) is required, and the medical practitioner needs only to attend his/her patient. If one of the prohibited bronchodilators is considered essential due to the patient's characteristics or treatment history, or to improve adherence, wellbeing or ease of use, etc., the reason for this decision must be explained, and a TUE application must be submitted to demonstrate that this subject is asthmatic and experiences real bronchial hyperresponsiveness. In the application, the following points must be addressed: (1) the patient's disease must be demonstrated on the basis of detailed reports and validated clinical tests4; (2) it must be made clear that the prohibited substance or method in question is necessary to treat an acute or chronic medical problem which, if untreated, would cause a significant deterioration in the individual's health; (3) that there is no reasonable therapeutic alternative to the use of the prohibited substance; and (4), that is highly unlikely that this treatment will improve the individual's performance more than that what could be achieved from restoring them to their normal state of health.5 The reason for the prohibition of some beta-2-agonists is not a whim or a financial maneuver from pressure groups, but is based on point 4 of the TUE criteria listed above: these substances at therapeutic doses are useful for the treatment of asthma, but at high concentrations they also have performance-enhancing effects,6,7 and can be used by some individuals for unethical purposes.
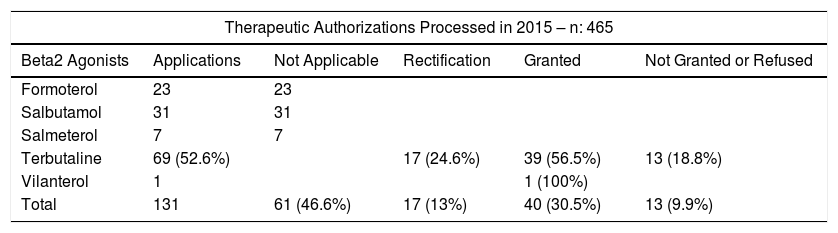
If, as doctors, we perform our work efficiently, this will impact the efficacy of everyone involved: not only the patient, but also the entire community, the family, the sporting bodies, colleagues who help us perform the additional tests, and the TUE assessors. A TUE is not a mere formality, and a submission involves a lot more than simply completing the form. It is a serious, official application for permission to use a particular medication, and must be properly completed and well justified. Additional tests conducted by various professionals must be attached, the results of which may not always be as desired,8 the application sections listed above must be addressed in detail, and the official form must also be completed. In short, the application requires quality work and plenty of time. It must also be emphasized that a TUE application is not a document that should be submitted “just in case” or processed because the word is out that “these medications can give positive doping results”. Medications for asthma and other diseases may either constitute doping or not or they may under certain conditions result in an adverse analytical finding. All applications are evaluated by all members of the Therapeutic Use Exemption Committee (TUEC) of the Spanish Agency for the Protection of Health in Sport, a process which involves the dedication of several individuals and specific administrative procedures. The TUEC does not judge the treatments per se, but it does evaluate if the application for the use of that treatment, endorsed by a medical specialist, meets the criteria to permit the athlete to use a prohibited substance that, while allowing them to train and compete in health conditions similar to their peers on a level playing field, might also enhance their performance.9 However, this standard of quality is not always achieved. As shown in Table 1, a total of 465 applications were made in 2015, 131 (28.2%) of which were for beta-agonists, and of these, 61 (46.6%) were unnecessary. Furthermore, rectification was requested for 25% of the 69 applications for the use of terbutaline, in the form of a request for a more comprehensive report or for a better rationale, particularly regarding points 2 and 3 mentioned previously. We would draw attention to this, since point 3 requires the most justification, in view of the availability of alternative treatment, and point 4 is the criterion which allows the use of some bronchodilators, but under the terms of a TUE.10,11 With regard to the overall number of applications for the use of corticosteroids in the context of asthma, nearly all are unnecessary, since they refer to permitted treatments and routes of administration. In summary, of the 202 applications related with the treatment of asthma submitted by pulmonologists, allergologists, pediatricians, family doctors, and sports physicians, 132 (65%) were unnecessary, and 21(10%) had to be rectified. We can surely do better than that.
Decisions on Therapeutic Use Exemption applications assessed by the Therapeutic Use Exemption Commission of the Spanish Agency for the Protection of Health in Sport in 2015.
| Therapeutic Authorizations Processed in 2015 – n: 465 | |||||
|---|---|---|---|---|---|
| Beta2 Agonists | Applications | Not Applicable | Rectification | Granted | Not Granted or Refused |
| Formoterol | 23 | 23 | |||
| Salbutamol | 31 | 31 | |||
| Salmeterol | 7 | 7 | |||
| Terbutaline | 69 (52.6%) | 17 (24.6%) | 39 (56.5%) | 13 (18.8%) | |
| Vilanterol | 1 | 1 (100%) | |||
| Total | 131 | 61 (46.6%) | 17 (13%) | 40 (30.5%) | 13 (9.9%) |
| Corticosteroids | Applications | Not Applicable | Rectification | Granted | Not Granted or Refused |
|---|---|---|---|---|---|
| Prednisone | 36 | 2 | 31 | 5 | |
| Budesonide | 27 | 24 (88.9%) | 1 | 3 | |
| Fluticasone | 27 | 26 (96.3%) | 1 | 1 | |
| Methylprednisolone | 25 | 1 | 23 | 2 | |
| Dexamethasone | 23 | 2 | 21 | 2 | |
| Betamethasone | 15 | 10 (66.7%) | 2 | 4 | 1 |
| Deflazacort | 15 | 15 | |||
| Triamcinolone | 9 | 6 | 2 | 3 | |
| Beclomethasone | 8 | 7 (87.5%) | 1 | ||
| Mometasone | 3 | 3 (100%) | |||
| Ciclesonide | 1 | 1 (100%) | |||
| Methylprednisone | 1 | 1 | |||
| Others | 2 | 2 | |||
| Total | 192 | 77 (40.1%) | 11 (5.7%) | 103 (53.6%) | 12 (6.3%) |
The percentages of the most significant applications are shown. Those for betamethasone and budesonide are not limited to asthma and also apply to anti-inflammatory therapies for other systems. No applications for use in asthma were denied.
Please cite this article as: Drobnic F, Blanco-Alfaro J. La solicitud de autorización de uso terapéutico para agonistas betaadrenérgicos y corticoides inhalados en el deportista con asma. Arch Bronconeumol. 2018;54:223–225.