Sustained hyperuricemia (>7mg/dl), when it manifests as chronic tophaceous gout (CTG), can lead to the formation of granulomas (tophi) around the urate crystals, which have a high capacity for erosion.1 Laryngeal involvement in CTG is rare, and can cause upper airway obstruction and acute respiratory failure (ARF),2 and can affect the tracheobronchial tree.3 We report the case of a patient with CTG who developed ARF after an acute episode of laryngeal gout requiring tracheostomy, and who also presented tophi in the left main bronchus (LMB). The patient ultimately developed squamous cell carcinoma of the left upper lobe (LUL) bronchus. To the best of our knowledge, this is the first description in the literature of a patient with both lesions.
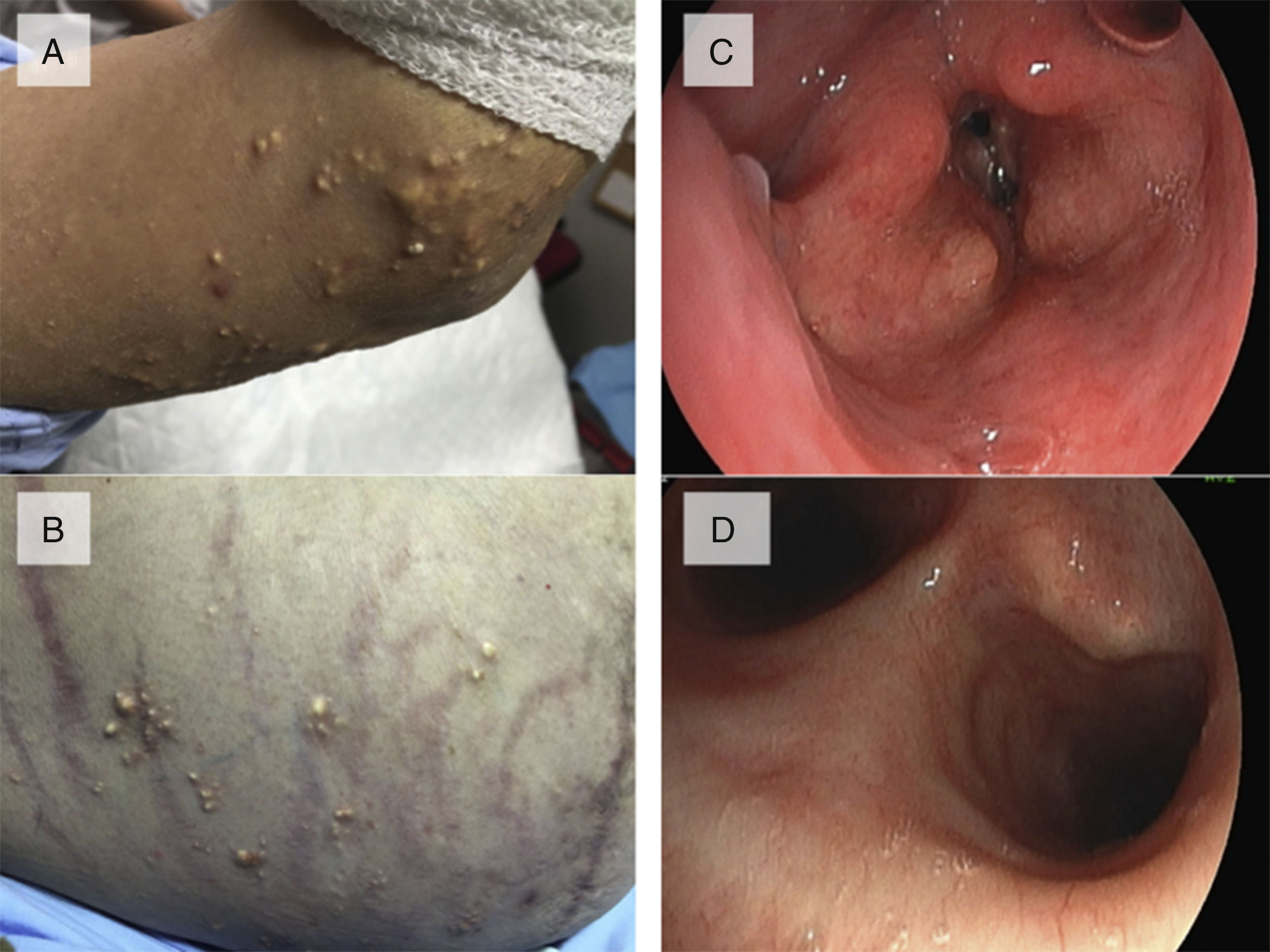
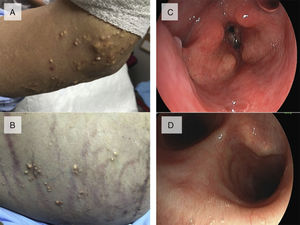
This was a 51-year-old man, smoker of 60 pack-years, with a clinical history of chronic bronchitis, obesity, symptomatic hyperuricemia treated with allopurinol 300mg/day (although compliance was irregular), arterial hypertension, and metabolic syndrome. One year previously, he had presented in the emergency department on repeated occasions with episodes of dyspnea, even at rest, attributed to COPD exacerbations, treated with bronchodilators and corticosteroids and discharged home with symptomatic treatment. Four months later, he returned to the emergency department with a severe attack of dyspnea. Examination showed increased work of breathing, central and peripheral cyanosis, stridor, and the following arterial blood gases: PaO2 55mmHg, PaCO2 60mmHg, pH 7, 20 and HCO3 22mmol. Chest X-ray revealed mild cardiomegaly and no other findings. An examination of the skin showed multiple giant tophi on the elbows, knees and hands, with deformed joints, on the eyelids, and on the abdominal wall, legs, and arms (Fig. 1A and B). He was assessed by the pulmonologist in the emergency department, and urgent evaluation by the ENT specialist was requested, in view of suspected upper airway stenosis. Fiberoptic laryngoscopy showed paralysis of the vocal cords (VC) in adduction, and whitish masses consistent with tophi on the VC and arytenoids (Fig. 1C), requiring urgent tracheostomy. Fiberoptic bronchoscopy showed whitish, excretory lesions in the LMB, similar to those observed in the larynx (Fig. 1D). Bronchial biopsies revealed fibrinous bloody material with isolated atypical epithelial cellularity and fragments of bronchial wall with no significant morphological changes. Laryngeal biopsies showed mucosa with squamous epithelium, with no changes and no evidence of malignancy. Of note in the clinical laboratory tests was uric acid 12mg/dl. After oxygen therapy, bronchodilators, and antibiotics, the patient was discharged with a permanent tracheostomy. During his hospital stay, a respiratory polygraphy with open tracheostomy was performed, showing episodes of severe hypoventilation, and an AHI of 8.5/h. Treatment began with invasive mechanical ventilation via the tracheostomy in the hospital, followed by night time ventilation at home (VIVO 50 VA/C: Vt 1000ml, Fr 12bpm, EPAP 6cmH2O, Ti 1.5s, inspiratory trigger 3, and descending ramp), with good compliance. One year later, the patient was admitted for hemoptysis, with soft tissue density visualized on CT around the LMB, origin of the intermediate bronchus and the LUL bronchus. In addition to the LMB lesions, fiberoptic bronchoscopy showed almost complete stenosis of the lingular bronchus and partial stenosis of the culmen with necrotic mucosa. Biopsies revealed well-differentiated squamous cell carcinoma.
Head and neck tissue involvement in CTG is exceptional, and VC paralysis due to acute gouty arthritis is even rarer. Moreover, bronchial involvement has only been described in 1 patient,3 and there are no more than 20 published cases of laryngeal involvement, none of which appear in the Spanish literature.4 Although very few studies have reported laryngeal involvement in gout, which is more commonly observed in association with the cricothyroid joint, Garrod, in 1863, described a few “specks” of sodium urate in the arytenoid cartilage in the autopsy of a man with CTG.5 Years later, Virchow reported the presence of monosodium urate in the VC of a patient with extensive tophaceous deposits.6 VC paralysis is a potential cause ARF, and exceptionally occurs with acute arthritis.2 As in our case, VC paralysis is caused by atrophy and denervation of some of the laryngeal muscles of the cricoarytenoid joint, rather than the direct deposit of tophi, causing fibrosis and inflammation on the perineuronal tissue, that ultimately leads to atrophy of the innervated muscle fibers and hypertrophy of the healthy tissue.7,8 Our patient also presented endobronchial tophi, although he ultimately developed squamous cell carcinoma. Of course, no cause–effect relationship between gout and the carcinoma caused by smoking can be established. The diagnosis of cricoarytenoid gout can be established by signs and symptoms observed in patients with extensive CTG, such as dysphagia, dysphonia, dyspnea, etc.2 Gouty laryngeal involvement should be considered in a case of history of hyperuricemia and acute or chronic gouty arthritis, accompanied by snoring, odynophagia, dysphagia, stridor, dyspnea, and/or dysphonia. Sometimes the upper airway is acutely compromised, requiring urgent tracheostomy, and bronchial lesions can also develop.3
Please cite this article as: Arlandis M, Molina V, Vañes S, Chiner E. Insuficiencia respiratoria aguda secundaria a gota tofácea crónica con afectación laríngea y bronquial: una complicación excepcional. Arch Bronconeumol. 2018;54:399–400.