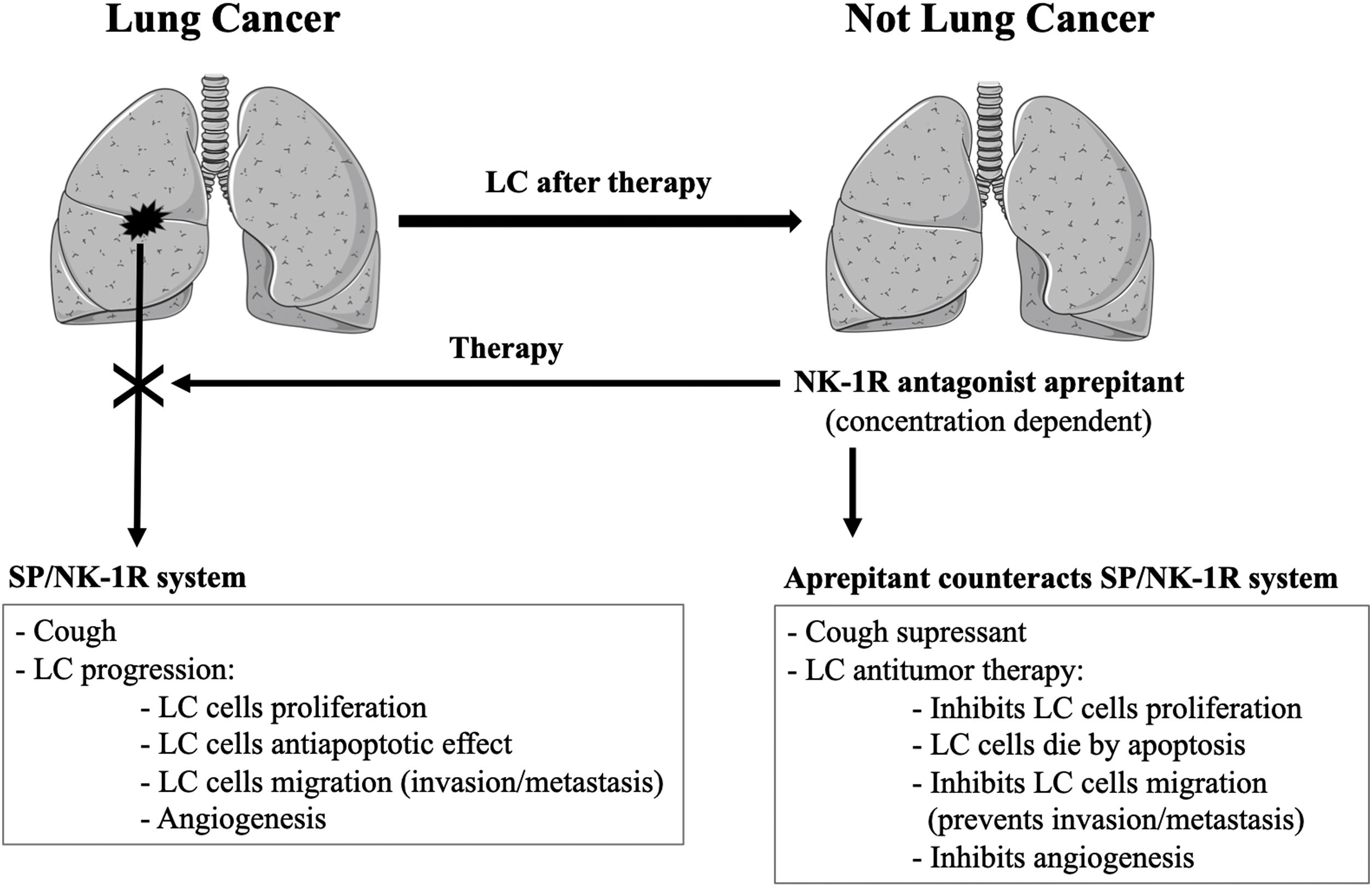
Effective antitussive cough treatments are an important need in patients with lung cancer (LC). Aprepitant drug, a selective neurokinin-1 receptor (NK-1R) antagonist and used for the treatment of nausea/vomiting, blocks the activation of the receptor by substance P (SP); this activation also mediates cough.1 Two randomized trials have clearly shown that aprepitant (day 1: 125mg; 2−7/2−3: 80mg) suppresses cough in LC patients.1,2 Preclinical/clinical studies have also shown the involvement of the SP/NK-1R system in cancer progression and the potential use of aprepitant as a new antitumor drug has been suggested.3 The key-points of these studies are the following: LC cells synthesize/release SP which acts through autocrine, paracrine and neuroendocrine (tumor mass) mechanisms; LC tumor mass increases SP level in lung and blood; compared to healthy subjects, SP plasma levels are higher in subjects with cough and in patients with cancer; SP increases both cough and the proliferation of LC cells; LC cells overexpress the NK-1R; this receptor is involved in the viability of these cells; in a concentration-dependent manner, NK-1R antagonists (including aprepitant) suppress cough and exert an antitumor action because they decrease LC cell proliferation and induce the death of LC cells by apoptosis.4 NK-1R antagonists also inhibit the migration of tumor cells (counteracting invasion/metastasis) and angiogenesis. Aprepitant crosses the blood-brain barrier and could be useful to treat LC metastasis.3 For the first time, a combination therapy using aprepitant (1140mg/day for 45 days; compassionate use) and palliative radiotherapy to treat a patient suffering from LC has been reported5: the tumor mass (8×7cm) disappeared six months after treatment and no severe side-effect was found. The patient showed a general good health with weight gain and no biochemical analytical alteration.5 Before surgery, radiotherapy is currently used to partially decrease the size of large tumors. Thus, it seems that the whole disappearance of the LC tumor mass was due to the combination therapy of radiotherapy and aprepitant, although this must be confirmed in future studies.5 Importantly, the antitumor action of aprepitant is against any type of LC cells (small and non-small LC cells) because these cells overexpress the NK-1R.4 It has been suggested that the clinical antitumor dose of aprepitant to be administered could be 20−40mg/kg/day.3 In humans, the safety of aprepitant (300mg/day for 45 days; 375mg/day for two weeks) was excellent and it was well tolerated.3 Accordingly, an important question arises: Why use aprepitant only as a cough suppressant in LC when at higher doses it could also exert an antitumor action against LC? (Fig. 1) Thus, in LC patients: (1) aprepitant, in a concentration-dependent manner, could exert a dual therapeutic effect: cough suppressant and antitumor action against LC; (2) aprepitant must be repurposed as an antitumor drug, alone or in combination therapy, and (3) phase I and II clinical trials are urgently needed to assess its safety/efficacy. What are we waiting for when it could just be a question of dosage?
Not declared.
Author's contributionsAll authors participated equally.
Conflict of interestNot declared.
We would like to thank Mr. Javier Muñoz (Seville University) for technical assistance.