Different studies have described a positive association between altitude and mortality from respiratory diseases.1,2 However the impact of altitude on mortality from COVID-19 has shown opposite results, as the altitude could be protective or a risk factor for mortality.3–5 On one side, SARS-Cov-2 interacts with human Angiotensin Converting Enzyme Receptor 2 (ACE2)6 expressing more in adults, in men and at low altitudes, and then it has been hypothesized that Covid-19 mortality could be reduced at higher altitudes.7–9 On the other hand, hypoxemia caused by lung diseases is expected to be more severe at higher altitudes, especially in those that have a sudden onset and therefore have a short period of time for adaptation. Hypoxemia is considered a risk factor for mortality in patients with COVID-19.10 We carried out an exploratory ecological study to evaluate the association between altitude and mortality from COVID-19, taking socioeconomic and demographic factors into account.
Mexico is divided into 32 states and 2468 municipalities but we only analyzed 1926, those with at least one COVID-19 death. We utilized public information from the COVID-19 database of the Ministry of Health of Mexico and collected data at the municipal level on confirmed deaths by PCR or antigen test, the number of persons with diabetes mellitus, chronic obstructive pulmonary disease (COPD), asthma, high blood pressure, cardiovascular disease, obesity, chronic kidney disease, pneumonia and smoking status, and the average number of tests (PCR or antigen) per person from March to December 2020.11 The frequency of deaths, comorbidities and tests per person were computed by age (5-year age groups) and stratified by gender. We also collected for each municipality, information on mid-year population for 2020,12 population density (population/km2),13 whether the municipality is a metropolitan zone,12 the altitude in meters above sea level divided into six 500-m categories,14 and two composite indicators of socioeconomic status: human development index15 (HDI) with four categories, and marginalization level12 (ML) with five categories. The reference categories were those with the worst socioeconomic status.
The relation between altitude and mortality from COVID-19 was evaluated by Poisson regression models stratified by gender and adjusted by age, socioeconomic status (HDI or ML), population density, the average number of tests (PCR or antigen) per individual, state of residence and if the residence was in a metropolitan area, the number of people with diabetes mellitus, chronic obstructive pulmonary disease (COPD), asthma, hypertension disease, cardiovascular disease, obesity, chronic kidney disease, pneumonia and smoking. We included municipal population as offset term in each model. Data were analyzed with Stata software version 12.
Municipalities located at <500m and [1500–2000m) registered the highest proportions of cases, 19.5% and 22.3%, respectively. Table S1 describes the population characteristics by altitude in the supplementary material. The cases confirmed by PCR or antigen test were 1,431,541 with 9% of deaths. Crude mortality rates per 100,000 population were estimated by altitude, and no pattern was found in women or men (Tables S2 and S3). Details on mortality rates by age (Tables S4 and S5) and comorbidities distribution by altitude could be found in the supplemental material (Tables S6–S11).
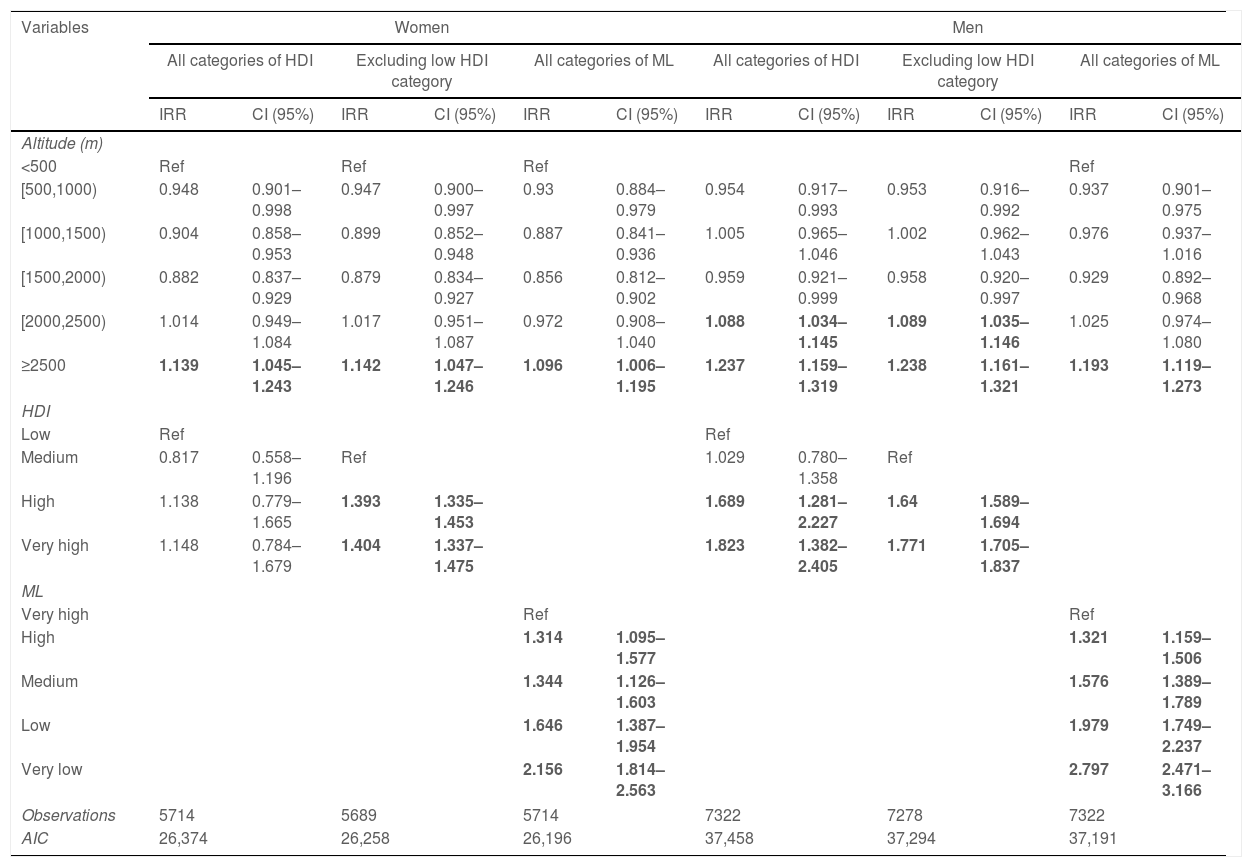
Poisson models (Table 1) depict that the risk of death was increased between 9.6% and 14.2% in women living at ≥2500m compared to those living at <500m. In models adjusted by HDI, the risk of death in men was increased 8.9% and 23.8% when living at (2000–2500m) and ≥2500m respectively. The risk of death was 19.3% higher in men living at ≥2500m if adjusted by ML.
Adjusted incidence rate ratios by altitude, human development index (HDI) or marginalization level (ML).
| Variables | Women | Men | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| All categories of HDI | Excluding low HDI category | All categories of ML | All categories of HDI | Excluding low HDI category | All categories of ML | |||||||
| IRR | CI (95%) | IRR | CI (95%) | IRR | CI (95%) | IRR | CI (95%) | IRR | CI (95%) | IRR | CI (95%) | |
| Altitude (m) | ||||||||||||
| <500 | Ref | Ref | Ref | Ref | ||||||||
| [500,1000) | 0.948 | 0.901–0.998 | 0.947 | 0.900–0.997 | 0.93 | 0.884–0.979 | 0.954 | 0.917–0.993 | 0.953 | 0.916–0.992 | 0.937 | 0.901–0.975 |
| [1000,1500) | 0.904 | 0.858–0.953 | 0.899 | 0.852–0.948 | 0.887 | 0.841–0.936 | 1.005 | 0.965–1.046 | 1.002 | 0.962–1.043 | 0.976 | 0.937–1.016 |
| [1500,2000) | 0.882 | 0.837–0.929 | 0.879 | 0.834–0.927 | 0.856 | 0.812–0.902 | 0.959 | 0.921–0.999 | 0.958 | 0.920–0.997 | 0.929 | 0.892–0.968 |
| [2000,2500) | 1.014 | 0.949–1.084 | 1.017 | 0.951–1.087 | 0.972 | 0.908–1.040 | 1.088 | 1.034–1.145 | 1.089 | 1.035–1.146 | 1.025 | 0.974–1.080 |
| ≥2500 | 1.139 | 1.045–1.243 | 1.142 | 1.047–1.246 | 1.096 | 1.006–1.195 | 1.237 | 1.159–1.319 | 1.238 | 1.161–1.321 | 1.193 | 1.119–1.273 |
| HDI | ||||||||||||
| Low | Ref | Ref | ||||||||||
| Medium | 0.817 | 0.558–1.196 | Ref | 1.029 | 0.780–1.358 | Ref | ||||||
| High | 1.138 | 0.779–1.665 | 1.393 | 1.335–1.453 | 1.689 | 1.281–2.227 | 1.64 | 1.589–1.694 | ||||
| Very high | 1.148 | 0.784–1.679 | 1.404 | 1.337–1.475 | 1.823 | 1.382–2.405 | 1.771 | 1.705–1.837 | ||||
| ML | ||||||||||||
| Very high | Ref | Ref | ||||||||||
| High | 1.314 | 1.095–1.577 | 1.321 | 1.159–1.506 | ||||||||
| Medium | 1.344 | 1.126–1.603 | 1.576 | 1.389–1.789 | ||||||||
| Low | 1.646 | 1.387–1.954 | 1.979 | 1.749–2.237 | ||||||||
| Very low | 2.156 | 1.814–2.563 | 2.797 | 2.471–3.166 | ||||||||
| Observations | 5714 | 5689 | 5714 | 7322 | 7278 | 7322 | ||||||
| AIC | 26,374 | 26,258 | 26,196 | 37,458 | 37,294 | 37,191 | ||||||
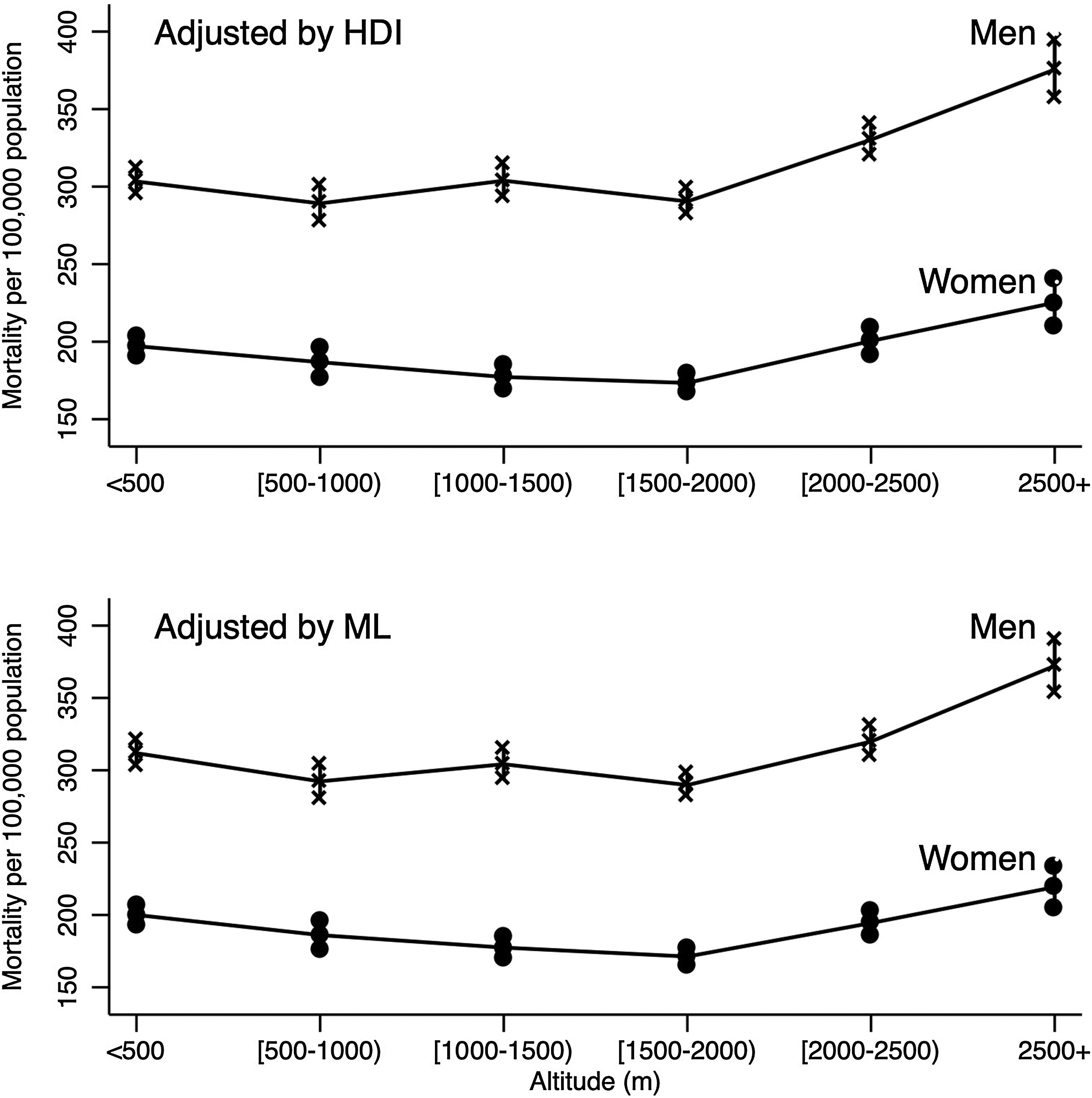
Fig. 1 describes the adjusted mortality rates per 100,000 inhabitants for men and women controlling for HDI and ML estimated by Poisson models. From [1500–2000m) to ≥2500m a positive trend in mortality rate was detected in women and men.
Human development index-adjusted and marginalization level-adjusted mortality rates from COVID-19 as a function of altitude*. *Mortality rates were estimated using Poisson models, which were adjusted by: age, population density, number of test (PRC or antigen) per individual, metropolitan zone, state of residence, the number of people with diabetes mellitus, chronic obstructive pulmonary disease (COPD), asthma, hypertension disease, cardiovascular disease, obesity, chronic kidney disease, pneumonia and smoking. HDI: human development index; ML: marginalization level.
Studies have described several discordant mechanisms by which exposure to moderate or high altitude can modify the severity of respiratory diseases. On one hand, a drop in partial or absolute barometric pressure may decrease the absorption of pulmonary fluid leading to pulmonary edema16 and more likely respiratory infections. In addition, chronic hypoxia induces leukocyte-mediated tissue damage17 and by itself is a risk of adverse outcomes.
On the other hand, chronic exposure to moderate or high altitudes may enhance adaptive features in the lungs at the organ and cellular level, as well as modulation of ACE2 protein expression in the lung cell membrane6 and be a protective factor for both COVID-19 incidence and mortality due to a multisystemic adaptation process to hypoxia.4,5
Studies carried out in Peru and Colombia reported that altitude is a protective factor for the risk of death from COVID-19.4,5 In the Peruvian study, the mortality comparisons were made for areas at different altitudes matched by population density adjusting for only one confounder at a time. Thus, the reported relative risk (RR) could be different if multiple adjustment had been made. In Colombia, multiple linear regression models including population density and altitude were fitted for the number of deaths and fatality rate but it was not clear if altitude was or not a risk for death. Our study incorporated adjustment by confounding factors such as population density, clinical conditions, and belonging to a metropolitan area, which were not considered in a previous study performed in Mexico but which nevertheless also concluded that altitude could be a risk factor for COVID-19 mortality.3
The main limitation of our study is the ecological design, and therefore the results may not apply at the individual level. In addition, we did not consider confounding factors that may modify the relationship between altitude and mortality from COVID-19 such as silent hypoxemia, health care resources and availability of services, climate, and air pollution.18–20 Additionally, in Mexico firewood is still used as a source of energy to cook food, especially in rural areas, and inhalation of wood smoke is associated with impaired pulmonary function21; this factor was also not considered.
In conclusion, after adjusting by several confusion factors, our results suggest that above 2000m in men and above 2500m in women, the risk of mortality increases between 8.9% and 23.8% compared to residents living at <500m above sea level.
However, these results should be considered with caution and individual studies are recommended to confirm these findings.
Financial/non-financial disclosureThis research did not receive any specific grant from funding agencies in the public, commercial, or non-profit sectors. None of the authors has any financial interest or other conflicts of interest.