Species belonging to the genus Scedosporium/Pseudallescheria are opportunistic dematiaceous fungi that cause localized and disseminated infections, are difficult to treat and are almost always associated with immunosuppression. They are, however, rarely isolated from mycetomas in patients without severe immunosuppression.1
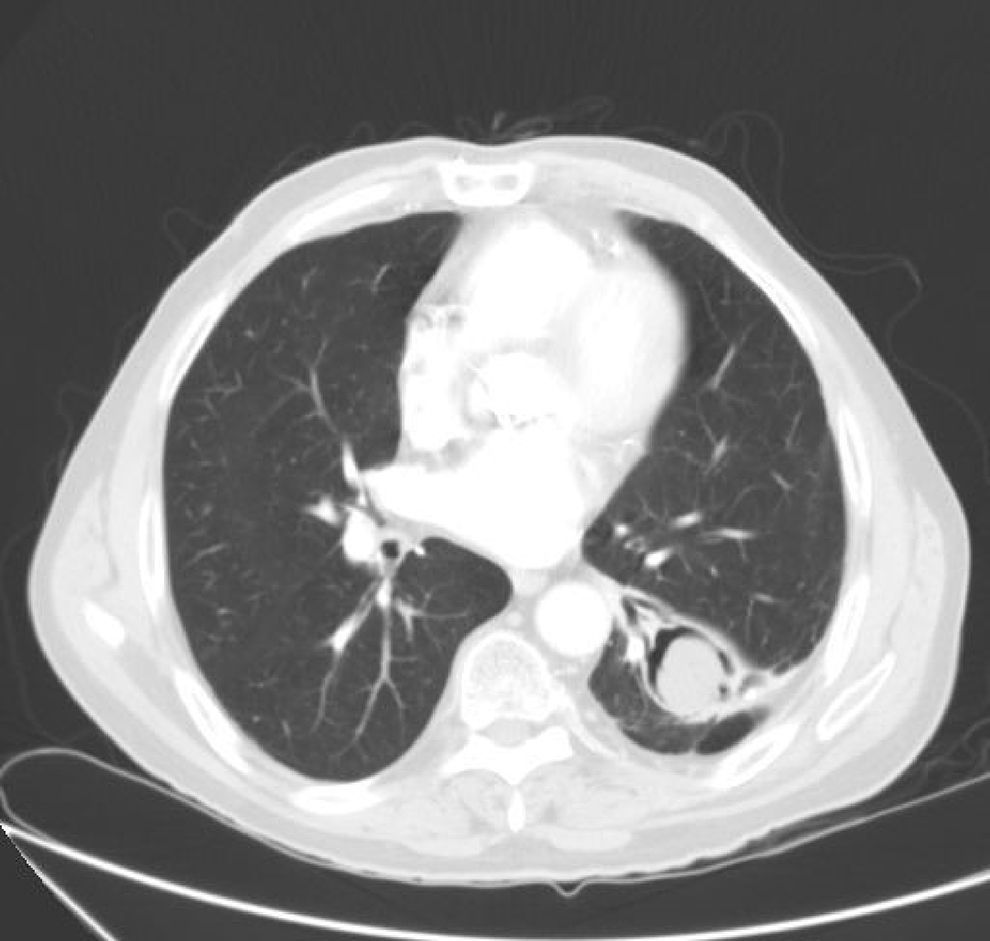
We report the case of an 84-year-old man with a history of pleural tuberculosis 50 years previously, with a biological aortic prosthesis, admitted to the hospital with an 8-h history of bloody expectoration. Physical examination showed him to be afebrile, tachypneic at 28 breaths/min, blood pressure 130/80mmHg, heart rate 80bpm, 95% oxygen saturation breathing room air, and crackles and rales in both bases on auscultation, with no other findings of interest. Clinical laboratory tests showed hematocrit 37%, and no other notable changes on complete blood count or serum biochemistry, including immunoglobulins. Chest radiograph revealed left basal infiltrate, sternal wires, and a biological aortic prosthesis. A chest computed tomography showed a cavity in the left lower lobe occupied by a solid mass consistent with mycetoma (Fig. 1).
Bronchoscopy performed after admission revealed inflammation of the mucosa in the left basal segments, with no neoplastic findings on histology. A Gram stain of the bronchial aspirate showed filamentous elements suggestive of fungi, and after 9 days of culture in appropriate medium for filamentous fungi, Scedosporium apiospermum/Pseudallescheria boydii was isolated from the sputum and bronchial aspirate.
Arterial embolization was performed and treatment was started with voriconazole, but hemoptysis persisted, so a lower left lobectomy was performed. The surgical piece revealed a cystic cavity measuring 3cm×2cm×2cm. Hyphae were visualized on fluorescence microscopy using calcofluor-white. S. apiospermum/P. boydii were identified from the culture. Results of the amplification and sequencing of ITS regions were consistent with P. boydii. The patient has remained asymptomatic since the intervention and there has been no recurrence of hemoptysis.
The genus Scedosporium/Pseudallescheria is composed of filamentous fungi belonging to the Microascaceae family. These are pleomorphic, dematiaceous fungi, with both sexual and asexual reproduction.1 Six species have been identified to date using molecular phylogeny techniques: Scedosporium prolificans, S. apiospermum, P. boydii, Scedosporium aurantiacum, Pseudallescheria minutispora and Scedosporium dehoogii, the last 5 constituting the S. apiospermum/P. boydii complex. Species in which sexual reproduction has been identified are called Pseudallescheria.1–4
S. prolificans causes infections with high mortality rates, since it is a more virulent species and resistant to practically all available antifungal agents. It is found all over the world, but fewer S. prolificans infections are reported in the north of Spain, Australia and the United States.5–7 It is not part of the human microbiota, and is acquired spontaneously from exogenous sources. It has been isolated from soil samples, stagnant water, streams, and from contaminated environments in general.5 The most common routes of entry are the inhalation and aspiration of conidia. Both the S. apiospermum/P. boydii complex and S. prolificans are important emerging opportunistic pathogens that have been related with a growing number of cases of infection and can colonize surfaces, ducts or cavities.
A study conducted in patients admitted to 29 Spanish hospitals concluded that the prevalence of colonization or infection by filamentous fungi was 1.6 per million inhabitants, and that the second most common fungus after Aspergillus spp. is Scedosporium spp.7
Depending on the patient's immune status, this pathogen, like Aspergillus spp., can colonize the respiratory system, causing bronchiectasis and cavities with formation of mycetomas, or severe forms of disseminated invasive disease.2
The case we describe involved a non-invasive infection-colonization of an existing cavity, possibly caused by tuberculosis. This clinical form has been called mycetoma or pseudallescherioma by different authors,8,9 and several studies have been published on Scedosporium mycetoma in patients with immune deficiency due to HIV, insulin-dependent diabetes, cancer, solid organ transplants and/or immunosuppressive treatments or corticosteroids. More exceptional cases have been reported in immunocompetent patients with an accumulation of predisposing factors, such as bronchiectasis or tuberculosis. This was the case in our patient, who, despite his advanced age, presented no other evidence of severe immune deficiency.2,10
In addition to the inflammatory response with vascular proliferation observed in mycetomas, Scedosporium possesses proteases and peptidases that can degrade human fibrinogen and cleave the fibronectin and laminin of the basement membrane of the lung tissue, favoring vascular invasion, dissemination and the development of hemoptysis, which can be persistent – as observed in our patient.11
A specific diagnosis of Scedosporium colonization or infection cannot be determined from imaging studies, clinical examinations, and histopathological findings.12S. apiospermum/P. boydii colonies are lighter in color and more cottony in texture than S. prolificans in standard culture media. It is essential to identify the species, as they differ in terms of virulence and sensitivity to antifungal agents. Newly described species are practically indistinguishable in morphological terms, so they must be differentiated using molecular techniques or proteomics. The targets are sequences that encode the ribosomal RNA complex (18s, 5.8s and 28s), ITS regions, and due to their greater resolving power, genes that encode proteins such as calmodulin or β-tubulin.13,14
Mycetomas must be treated with a combination of surgery and antifungal treatment when possible. S. apiospermum/P. boydii responds better to voriconazole, caspofungin, micafungin and anidulafungin. S. prolificans is resistant to azoles and amphotericin B. Several publications have highlighted the value of the synergistic action of several anti-fungal agents whenever possible.1,15 In our patient, we decided to perform a lobectomy with resection of the mycetoma, in view of the repeated episodes of hemoptysis despite embolizations and treatment with voriconazole. The patient has remained asymptomatic without recurrence for 18 months.
We thank Dr Luis Torres of the Microbiology Department of Hospital San Jorge and Dr Antonio Rezusta of the Microbiology Department of Hospital Miguel Servet for their help with the microbiological studies.
Please cite this article as: Colmenarejo C, Milagro A, Briz E, Borderías L. Hemoptisis persistente en un paciente anciano con micetoma por Pseudoallescheria boydii. Arch Bronconeumol. 2017;53:527–528.