The global incidence of coronavirus disease 2019 (COVID-19), the disease caused by severe acute respiratory syndrome-coronavirus-2, is increasing dramatically. Most COVID-19 cases present with only mild symptoms or no symptoms1; however, COVID-19 can also cause diffuse alveolar damage resulting in acute hypoxemic respiratory failure (AHRF), which requires intensive care unit admission and mechanical ventilation in a high percentage of cases.2 High-flow nasal cannula (HFNC) oxygen therapy is typically the prescribed respiratory therapy for AHRF. Recently both the Society of Critical Care Medicine (SCCM)3 and the World Health Organization (WHO)4 have recommended the use of HFNC oxygen therapy in AHRF caused by COVID-19. However, whether HFNC oxygen therapy is an aerosol generating medical procedure (AGMP) remains controversial—the WHO regards this therapy as an AGMP,4 whereas the SCCM does not.3
Respiratory aerosol generation and dispersion have been assessed by several methods, such as imaging with smoke light detection, schlieren imaging, and laser light scattering and aerosol particle sizer (APS) spectrometer.5 Most studies using these imaging methods examined exhaled breath by using smoke and a mannequin.5 Gaeckle et al. used an APS spectrometer and proposed that HFNC oxygen therapy did not increase the aerosol concentration.6 However, they collected exhaled breath in a negative pressure room through a funnel approximately 5cm away from the mouth; therefore, they could not evaluate the total aerosol concentration from respiratory tract. This current report represents two experimental trials with a novel fine particle visualization system and an APS spectrometer to evaluate whether particle dispersion from the respiratory tract increases while receiving HFNC oxygen therapy.
The first experimental trial was conducted using the same procedure as our previous study7 and four scenarios in which a healthy volunteer used devices such as nasal cannula at 4L/min and HFNC (AIRVO™2 device with an Optiflow™ nasal interface [Fisher & Paykel, Auckland, New Zealand]) without oxygen at a flow rate of 40L/min and temperature of 37°C. Particle dispersion was visualized by a video camera set at 29.97 frames per second (Eye Scope, Shin Nippon Air Technologies, Tokyo, Japan). This system used a light-emitting diode (wavelength 400–410nm; Parallel Eye D, Shin Nippon Air Technologies), which visualized particle ≥1μm in diameter. Images obtained were reconstructed as videos using a commercial software (Particle Eye, Shin Nippon Air Technologies).
The second experimental trial was performed with an APS spectrometer (model 3321, TSI Inc., St. Paul, MN, USA), which made it possible to measure the aerodynamic diameter of particles ranging from 0.52–20μm, in 5 healthy volunteers using devices such as nasal cannula at 4L/min and HFNC without oxygen at a flow rate of 40L/min and temperature of 37°C. Volunteers inhaled through the nose and exhaled through the mouth under normal breathing conditions, while holding a mouthpiece implanted in a box (size: 39.0×33.5×14.5mm) connected to the APS spectrometer. The APS spectrometer drew air at a flow of 5L/min with 1L/min directed to the measurement zone and 4L/min filtered and used as air for sheath flow in the instrument; 1-s spectra data were collected repeatedly for 60s.
These data were expressed as a median value with an interquartile range. All statistical analyses were performed using a statistical software package (JMP Pro 14 software; SAS Institute, Cary, NC, USA). For multiple comparisons, Kruskal–Wallis test was used. Differences were considered statistically significant at P<.05.
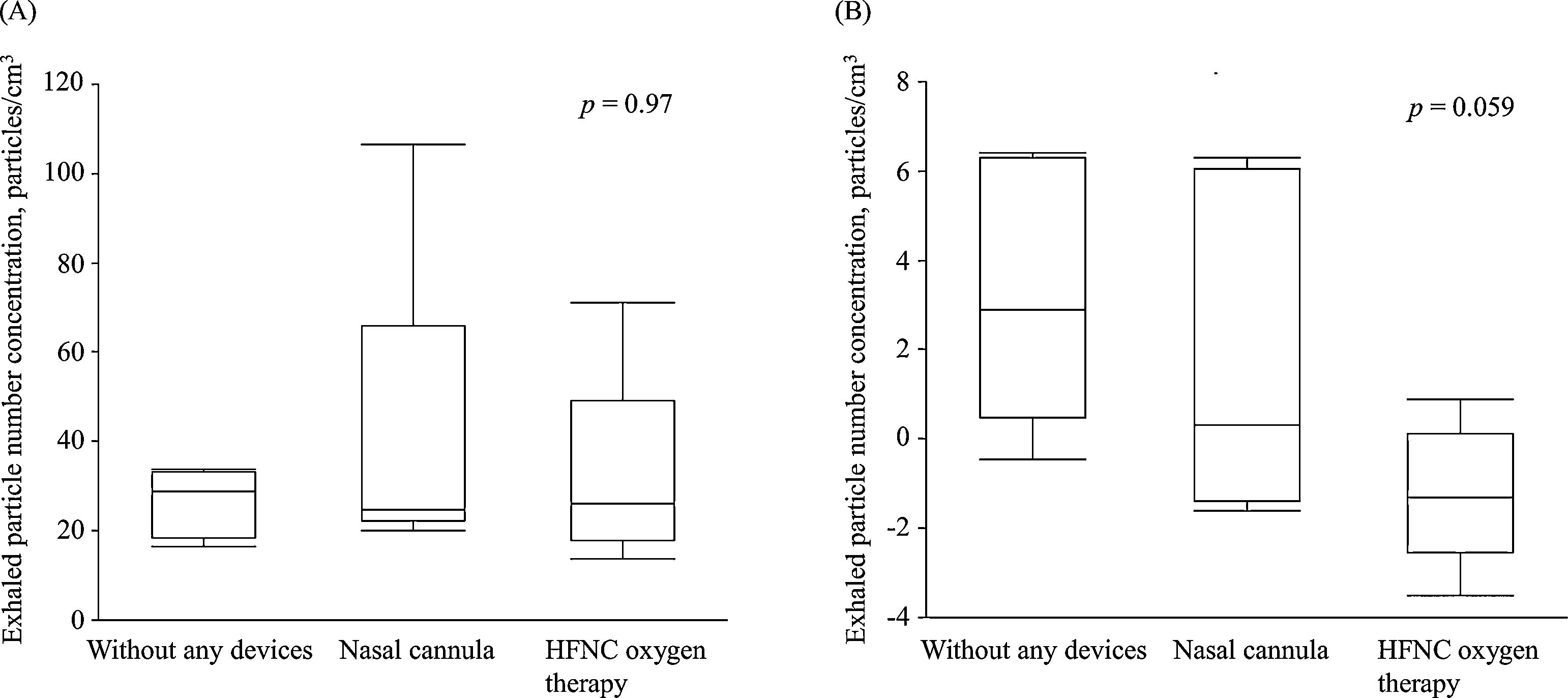
In the first experiment, we did not detect any remarkable particles exhaled through the mouth without any devices (Supplemental Video A) and with nasal cannula device (Supplemental Video B) and HFNC device (Supplemental Video C) during normal breathing, whereas we identified exhaled particles dispersed from coughing without any devices (Supplemental Video D). In the second experiment, the concentration of the exhaled particles subtracted from the background particle concentration ranging from ≥0.52 to <1.037μm did not differ among conditions, without any devices and with nasal cannula device and HFNC device (28.7 [18.35–33.3] without any devices vs. 24.7 [22.15–65.9] with nasal cannula vs. 26.1 [17.85–49.05]particles/cm3 with HFNC; P=.97) (Fig. 1A). In contrast, the concentration ranging from ≥1.037 to <20μm with HFNC device tended to be lower than without any device and with nasal cannula (2.9 [0.48–6.3] without any devices vs. 0.32 [−1.4 to 6.05] with nasal cannula vs. −1.3 [−2.55 to 0.12]particles/cm3 with HFNC; P=.059) (Fig. 1B). The median of the geometric mean diameter of particles was as follows: 0.73 (0.72–0.76) without any devices; 0.68 (0.68–0.7) with nasal cannula device; and 0.67 (0.66–0.71)μm with HFNC device.
To the best knowledge of these authors, this study is the first to describe visual and total quantitative evidence that HFNC oxygen therapy could not be an AGMP at least under normal breathing conditions in healthy subjects. These results reinforced those of Gaeckle et al. as previously described6 and supported the conclusion by the SCCM that HFNC oxygen therapy could not be an AGMP.3
The present data showed that the concentration of exhaled particles ranging from ≥1.037–20μm tended to be low in the condition with HFNC device at a flow rate of 40L/min (Fig. 1B). Gaeckle et al. reported that the use of HFNC device with flow rates of ≥30L/min decreased the particle number concentration in the setting of deep breathing.6 They suggest that HFNC oxygen therapy could decrease aerosol production by increasing positive end expiratory pressure and preventing closure of small bronchioles.6
This study had three major limitations. First, two experimental trials were performed in a small number of participants, particularly only one in the first trial. Second, we performed two trials in healthy participants with normal respiratory pattern. We should be cautious to apply the present findings to patients with AHRF, because different physical conditions, such as respiratory rate, temperature, and expiratory effort may influence aerosol generation. Third, we evaluated only one flow rate of 4L/min and 40L/min for nasal cannula device and HFNC device, respectively, although HFNC device could generate flow rates of up to 60L/min. Therefore, further studies with large numbers of patients with AHRF and different experimental settings such as 60L/min for HFNC device are needed to evaluate the effect of HFNC oxygen therapy on aerosol generation.
Financial ConflictsThis study was funded in part by the JSPS KAKENHI19K17634 (SH).
The Department of Advanced Medicine for Respiratory Failure is a Department of Collaborative Research Laboratory funded by Teijin Pharma.
Conflict of InterestSatoshi Hamada reports grants from Teijin Pharma, outside the submitted work.
The authors are grateful to Mr. Ryuta Okamoto, Mr. Taro Furukawa, and Mr. Kozo Takahashi, who are members of Shin Nippon Air Technologies, and Mr. Susumu Matsumoto, who is a member of Tokyo Dylec Corp, for technical assistance. Also, the authors are grateful to Dr. Tomohiro Handa, Dr. Hiroshi Shima, Dr. Satoru Terada, and Dr. Yusuke Shiraishi for performing the experimental trial with an aerosol particle sizer spectrometer.