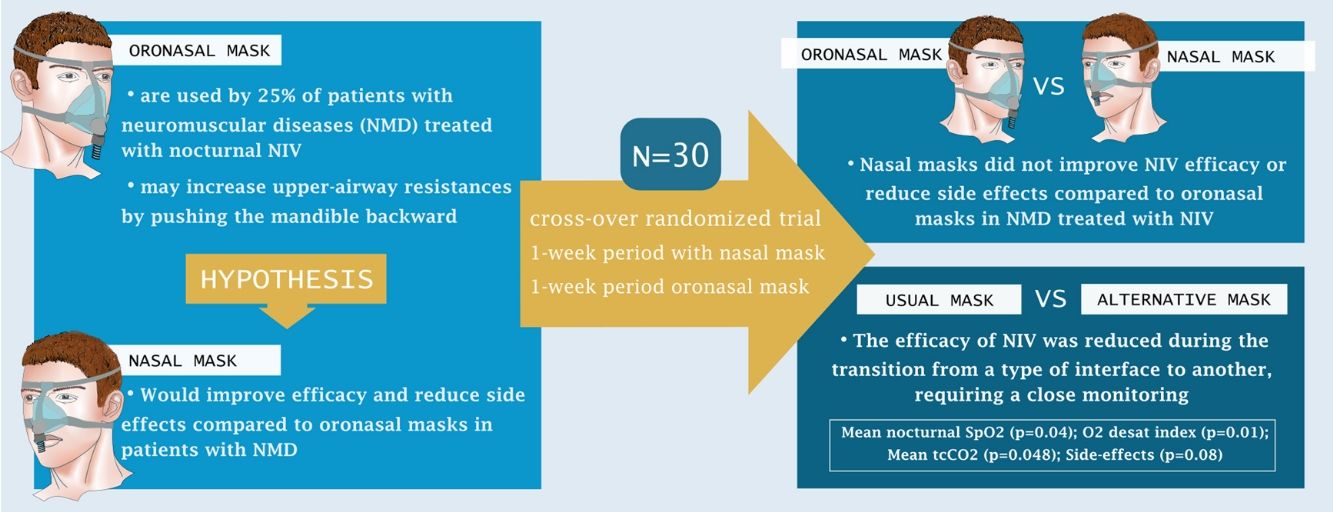
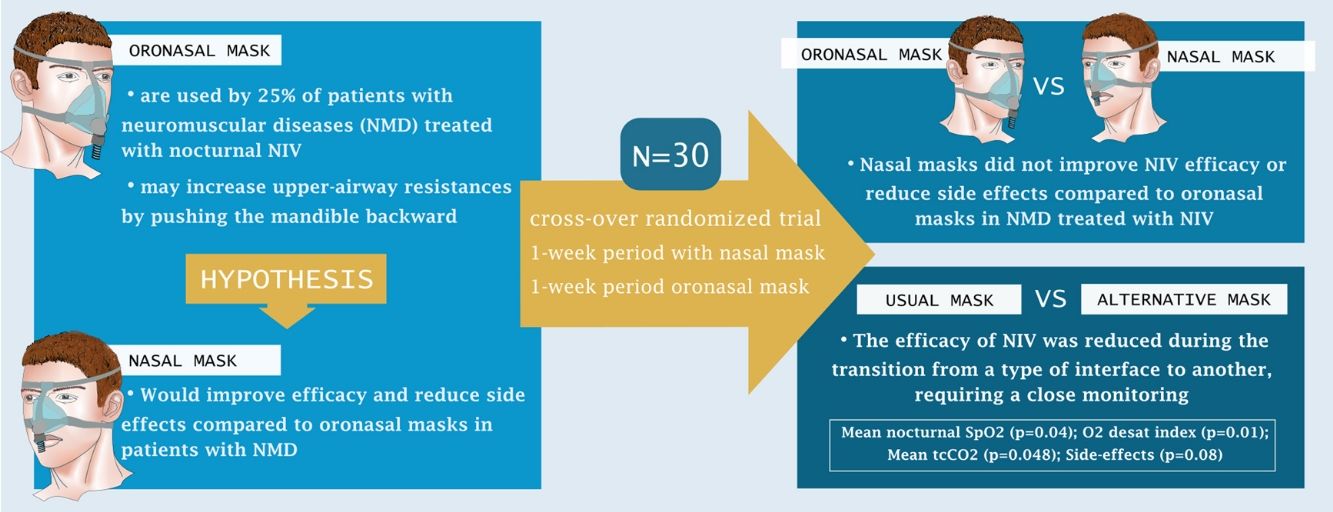
Around 25% of patients with neuro-muscular diseases (NMD) are treated by home noninvasive ventilation (NIV) through an oronasal mask. However, there is growing evidence that nasal masks require lower NIV pressures and result in fewer residual obstructive events. We hypothesized that nasal masks would improve efficacy and reduce side effects compared to oronasal masks in this population.
Methodsopen label, cross-over, randomized, study in 2 tertiary care hospitals. Patients with NMD treated by home NIV were randomized for one-week periods to nasal and oronasal interfaces respectively (cross-over). At the end of each period, nocturnal polygraphy (monitoring mouth opening) under NIV, synchronized with transcutaneous partial pressure in CO2 (tcCO2) was performed. Data were collected from the NIV built-in software and NIV side-effects were collected. Intention-to-treat and per protocol analyses were performed. The primary outcome was mean nocturnal SpO2. The secondary outcomes were: percentage of sleep with SpO2<90%, oxygen desaturation index (ODI), mean tcCO2, mean duration of mouth opening during sleep, level of non-intentional leaks and side-effects.
ResultsThirty patients with NMD were included. There were no between-group differences for either the primary or secondary outcomes. Post hoc comparisons showed that changing between interfaces reduced NIV efficacy: mean nocturnal SpO2 (p=0.04), ODI (p=0.01), mean tcCO2 (p=0.048), side-effects (p=0.008).
ConclusionNasal masks did not improve NIV efficacy or reduce side effects compared to oronasal masks in patients with NMD treated by home NIV. The efficacy of NIV is reduced during the transition to another interface, requiring close monitoring.
Registration number: NCT03458507.
Alrededor del 25% de los pacientes con enfermedades neuromusculares (ENM) son tratados mediante ventilación no invasiva (VNI) a través de una máscara oronasal. Sin embargo, existen crecientes indicios de que las máscaras nasales requieren presiones de VNI más bajas y resultan en menos eventos obstructivos residuales. Nuestra hipótesis fue que las máscaras nasales mejorarían la eficacia y reducirían los efectos secundarios en comparación con las máscaras oronasales en esta población.
MétodosEstudio abierto, cruzado, aleatorizado en 2 hospitales de atención terciaria. Los pacientes con ENM tratados mediante VNI domiciliaria fueron aleatorizados durante períodos de una semana de duración a las mascarillas nasales y oronasales, alternativamente (cruzado). Al final de cada período se realizó una polisomnografía nocturna (con monitorización de la apertura bucal) con VNI, sincronizada con la medición transcutánea de la presión parcial de CO2 (tcCO2). Los datos se recopilaron utilizando el software integrado en la VNI y se recogieron los efectos secundarios de la VNI. Se realizaron análisis por intención de tratar y por protocolo. El criterio de valoración principal fue la SpO2 nocturna media. Los criterios secundarios fueron: porcentaje de sueño con SpO2<90%, índice de desaturación de oxígeno (IDO), tcCO2 media, duración media de la apertura bucal durante el sueño, nivel de fugas no intencionales y efectos secundarios.
ResultadosSe incluyeron 30 pacientes con ENM. No hubo diferencias entre los grupos para los resultados primarios o secundarios. Las comparaciones a posteriori mostraron que cambiar mascarillas reducía la eficacia de la VNI: SpO2 nocturna media (p=0,04), IDO (p=0,01), tcCO2 media (p=0,048) y efectos secundarios (p=0,008).
ConclusiónLas máscaras nasales no mejoraron la eficacia de la VNI ni redujeron los efectos secundarios en comparación con las máscaras oronasales en pacientes con ENM tratados con VNI domiciliaria. La eficacia de VNI se reduce durante la transición a otra mascara, lo que requiere una estrecha vigilancia.
Home Noninvasive Ventilation (NIV) is the reference treatment for chronic alveolar hypoventilation in patients with neuro-muscular diseases (NMD).1,2 NIV can be provided through different types of interfaces, the most common of which are nasal masks and oronasal masks. The choice of interface is very important as it affects both the efficiency of the NIV and patient adherence, which is essential for effective treatment. Prior to initiation of NIV, clinicians must therefore determine which interface will provide the most efficient treatment but will also be comfortable for the patient so that they do not abandon the treatment.
Nasal masks are the most commonly used interface for home NIV in patients with NMD.3 However, persistent unintentional leaks, especially through the mouth trigger nasal congestion and/or nasopharyngeal and mouth dryness. Mouth leaks can also reduce NIV efficacy and impair sleep quality.4 Oronasal masks covering the nose and mouth are thus commonly proposed to patients in these situations.5
The decision to choose an oronasal mask is most often based on self-reported symptoms (especially leaks and side effects). Data collected from the NIV built-in software may also lead clinicians and home care providers to suspect leakage due to mouth opening. However, the proportion of real sleep time spent with an open mouth is usually not objectively documented6 and once an oronasal mask has been prescribed, a shift back to a nasal mask is almost never considered during follow-up as mouth leaks are considered to be a permanent problem.
About 25% of patients with NMD who are treated with nocturnal NIV at home use an oronasal mask3,7,8 but clinical reality shows that there is great heterogeneity of use between different countries, regions or units. A recent analysis showed that NMD patients who used oronasal interfaces were older and had higher BMI than patients who used nasal mask; two factors which are linked to obstructive apneas. Accordingly, positive expiratory pressures (PEP) set on the NIV were also higher with oro-nasal masks.9 This type of interface have been reported to increase upper-airway (UA) resistance by pushing the mandible backward, which in turn facilitates occurrence of upper airway obstruction.10,11
Very few studies have compared, using appropriate methodology, the efficacy of nasal mask versus oronasal mask in patients treated by long term nocturnal NIV.12,13 The aim of this study was to compare the efficacy and rate of side effects between nasal and oronasal masks in patients with NMD. We hypothesized that the application of an oronasal mask might compromise nocturnal mechanical ventilation and gas exchange by favoring pharyngeal closure or increased resistance in patients already prone to upper airway obstruction due to their underlying disease or to their maxillofacial profile. Thus, the use of a nasal mask (that does not reduce upper airway stability) would provide more effective NIV with fewer side effects compared to an oronasal mask.
MethodsTrial design (see details in online supplement)We performed an open label, cross-over, randomized, study in 2 tertiary care hospitals in France. Both hospitals had substantial experience in the management of NMD and treatment with NIV. The study was approved by our ethics committee (CPP Nord-ouest II) following French legislation, and written informed consent was obtained from all patients. The trial was registered in ClinicalTrials.Gov (number NCT03458507).
ParticipantsPatients with NMD treated with home NIV who were followed by the participating centers were screened for eligibility between May 2018 and May 2019. The inclusion criteria were: adults (>18 years old) treated with nocturnal non-invasive ventilation <15h/day (to exclude patient with severe respiratory insufficiency in which transition period should have been at risk) and diagnosed with a NMD (Becker muscular dystrophy, facio-scapulo-humeral dystrophy, limb-girdle dystrophy, myotonic dystrophy, Duchenne muscular dystrophy). Exclusion criteria were: occurrence of an acute cardiorespiratory or ear-nose-throat event during the month prior to inclusion, rapidly progressive NMD (such as ALS), severe nasal obstruction and maxillofacial deformities or previous upper airway surgery preventing the usage of one type of mask (nasal or oronasal).
InterventionsInclusion visit. Patients tested the interface they did not usually use (i.e. nasal mask for those treated with oronasal masks or oronasal mask for those treated with nasal masks) during a one-hour diurnal NIV session. The most appropriate alternative mask (in terms of brand and size) was chosen for each patient in order to minimize leakage and maximize comfort.
Follow up. At the end of the inclusion visit, patients were randomized to start either with their usual or alternative interface. During the week with the alternative interface, patients were asked to use this new interface with the aim to sleep overnight with it. During the week with the usual interface, patients were asked to pursue their treatment as usual. The number and nature of interface-related problems reported by patients and/or the need for a home care provider visit were collected during both weeks.
At the end of each week, an unattended type III nocturnal polygraphy under NIV was performed at home using a Somnolter® (Nomics, Liege, Belgium) device synchronized with transcutaneous partial pressure in CO2 (PtcCO2) monitoring by SenTec V-Sign™ System. This polygraph also records mandibular mouth opening.
Assessment of NIV side-effects was performed with the Modified SECI questionnaire14 (for each commonly reported side effect, the patient is asked to rate the frequency, magnitude and perceived impact on adherence on a five-point Likert-type scale).
OutcomesPatients were evaluated on two occasions, at the end of each week with either the usual or the alternative interface (cross-over). The primary objective was to compare mean nocturnal oxygen saturation between oronasal masks and nasal masks (Mean nocturnal SpO2, measured by polygraphy, Somnolter®). We also assessed the following exploratory secondary outcomes: percentage of sleep with SpO2<90% (polygraphy, Somnolter®); oxygen desaturation index (polygraphy, Somnolter®); mean nocturnal transcutaneous partial pressure in CO2 (PtcCO2, SenTec V-Sign™); mean mouth opening during sleep (polygraphy, Somnolter®); level of non-intentional leaks (percentage of recording time spend with leaks assessed by SomnoHolter®); the side-effects for each type of mask (Modified SECI).
RandomizationParticipants were randomized (computer-generated random numbers list) to either begin the trial with their usual interface (for one week) or the alternative interface (for one week) in order to allow a balance between the numbers of patients who would begin with their usual interface versus with an alternative interface, the randomization was stratified for the type of interface usually used by the patient.
Sample sizeVery few studies have compared nasal and oronasal masks for noninvasive ventilation in chronic respiratory failure.10,12,13 Therefore, in this pilot study, the sample size estimation was based on our inclusion capability and was based on the following: 1) at least 100 eligible patients were followed in the two participating centers, 2) 25% to 30% of patients with NMD use oronasal masks and we wanted to recruit equal numbers of patients who usually used oronasal masks and nasal masks and 3) we estimated that about 50% of eligible patients would accept to participate. Therefore, we planned to 30 patients (15 with nasal mask and 15 patients with oronasal mask). This sample size was sufficiently powered to detect a mean difference of mean nocturnal SpO2 of 2±2% (considered as clinically significant) between oronasal versus nasal mask (alpha=0.05, power=80%).
Statistical analysisA treatment effect test, a period effect test, and a test for the interaction between treatment and period were used to assess respective effect of treatment (nasal versus oronasal), treatment sequence, and the first-order carryover risk.
Paired student t-tests were conducted to assess differences between the two interfaces for normally distributed outcomes. Wilcoxon signed-rank tests for paired values were used to compare non-normally distributed outcomes. Significance was set at p<0.05. Missing data were not imputated.
All randomized patients were analyzed in intention to treat (ITT): if a patient did not tolerate the alternative interface, polygraphy was performed with their usual interface and data collected during this evaluation was used in the analysis. A per-protocol (PP) was secondarily performed using data from patients who completed both polygraphies with the appropriate masks without any protocol deviation. Finally, a post hoc comparison using the same statistical models was performed to compare the usual and alternative interfaces to assess the impact of mask change among those patients.
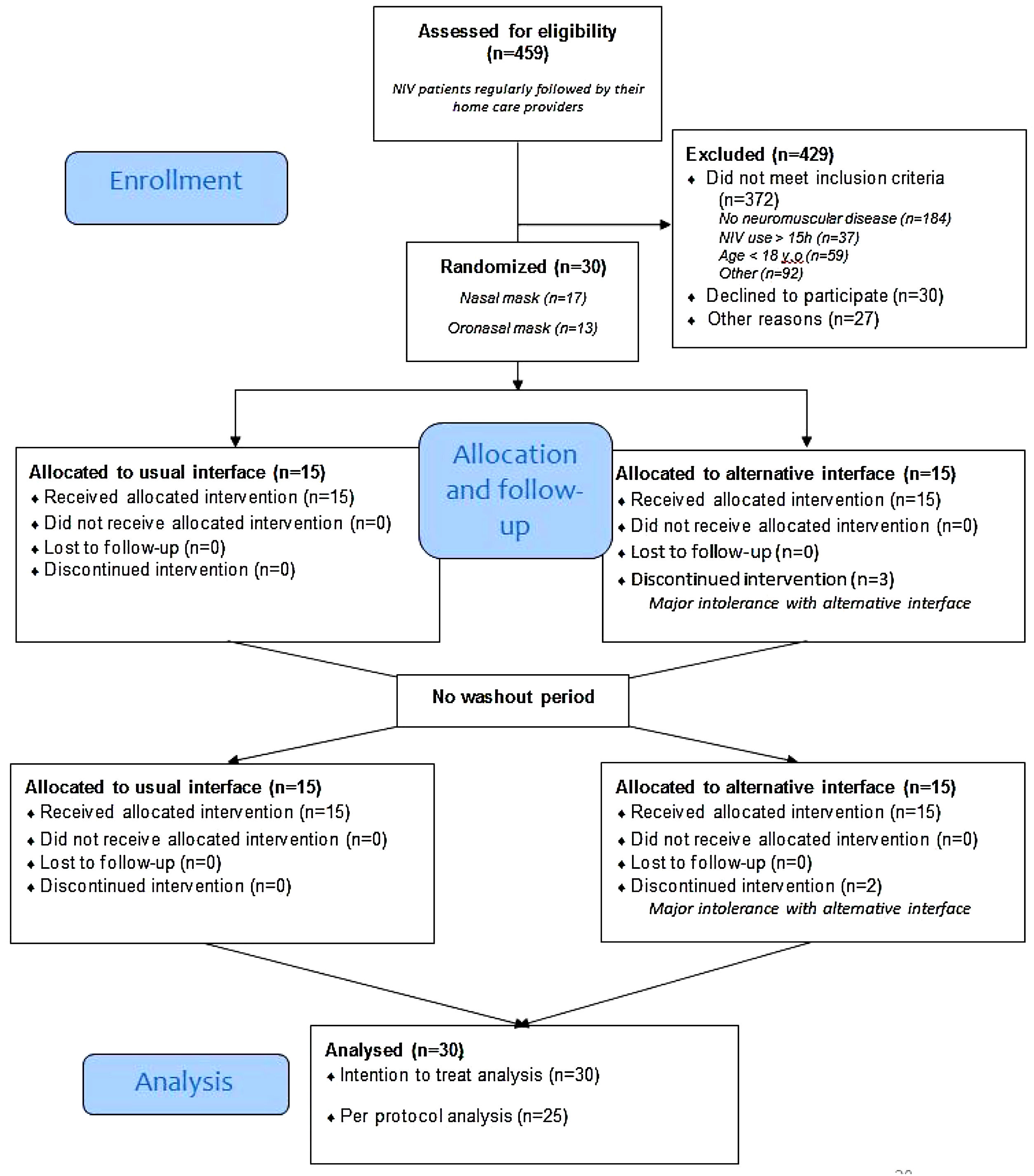
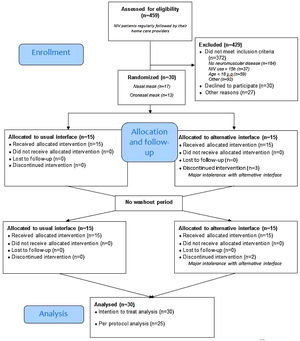
ResultsRecruitmentThe study flow is depicted in Fig. 1, 30 patients with NMD were included. Among them, 13 used an oronasal mask at home. During the week with the alternative interface, 5 patients had major intolerances (2 with the nasal mask and 3 with the oro-nasal) despite home care provider interventions and thus resumed the use of their usual interface (Fig. 1).
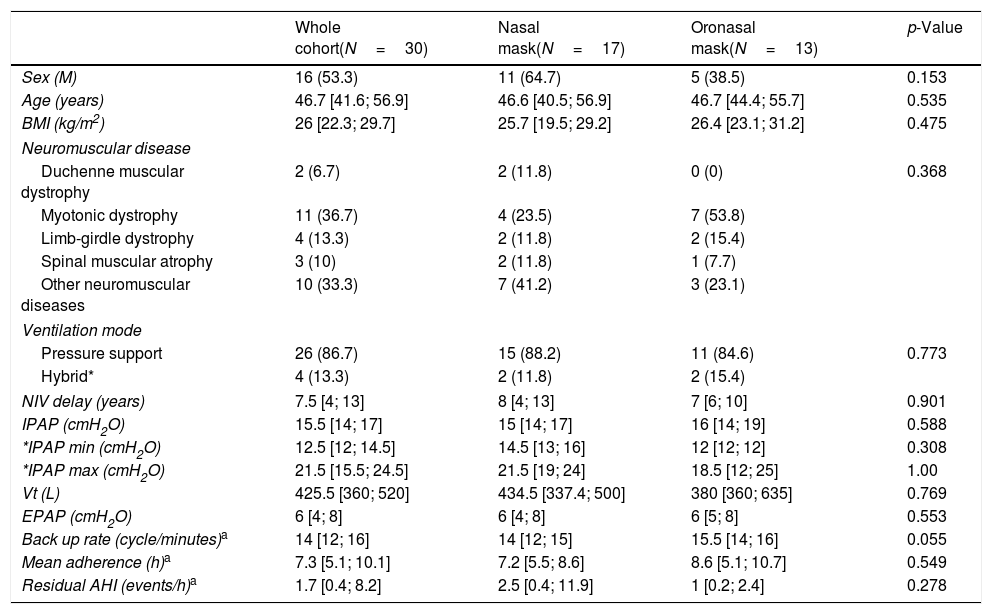
Table 1 presents the anthropometric characteristics of patients and ventilator settings. Patients were middle aged and equally distributed in terms of sex. Myotonic dystrophy was the most represented neuromuscular disorder (36.7%). Pressure support mode was used by 86.7% of patients with no differences in IPAP and EPAP settings between those who usually used nasal and those who used oronasal masks. There was no difference in mean NIV adherence, residual AHI or tidal volume estimated by NIV built-in software between patients who used nasal and those who used oronasal interfaces.
Demographics and ventilator settings.
| Whole cohort(N=30) | Nasal mask(N=17) | Oronasal mask(N=13) | p-Value | |
|---|---|---|---|---|
| Sex (M) | 16 (53.3) | 11 (64.7) | 5 (38.5) | 0.153 |
| Age (years) | 46.7 [41.6; 56.9] | 46.6 [40.5; 56.9] | 46.7 [44.4; 55.7] | 0.535 |
| BMI (kg/m2) | 26 [22.3; 29.7] | 25.7 [19.5; 29.2] | 26.4 [23.1; 31.2] | 0.475 |
| Neuromuscular disease | ||||
| Duchenne muscular dystrophy | 2 (6.7) | 2 (11.8) | 0 (0) | 0.368 |
| Myotonic dystrophy | 11 (36.7) | 4 (23.5) | 7 (53.8) | |
| Limb-girdle dystrophy | 4 (13.3) | 2 (11.8) | 2 (15.4) | |
| Spinal muscular atrophy | 3 (10) | 2 (11.8) | 1 (7.7) | |
| Other neuromuscular diseases | 10 (33.3) | 7 (41.2) | 3 (23.1) | |
| Ventilation mode | ||||
| Pressure support | 26 (86.7) | 15 (88.2) | 11 (84.6) | 0.773 |
| Hybrid* | 4 (13.3) | 2 (11.8) | 2 (15.4) | |
| NIV delay (years) | 7.5 [4; 13] | 8 [4; 13] | 7 [6; 10] | 0.901 |
| IPAP (cmH2O) | 15.5 [14; 17] | 15 [14; 17] | 16 [14; 19] | 0.588 |
| *IPAP min (cmH2O) | 12.5 [12; 14.5] | 14.5 [13; 16] | 12 [12; 12] | 0.308 |
| *IPAP max (cmH2O) | 21.5 [15.5; 24.5] | 21.5 [19; 24] | 18.5 [12; 25] | 1.00 |
| Vt (L) | 425.5 [360; 520] | 434.5 [337.4; 500] | 380 [360; 635] | 0.769 |
| EPAP (cmH2O) | 6 [4; 8] | 6 [4; 8] | 6 [5; 8] | 0.553 |
| Back up rate (cycle/minutes)a | 14 [12; 16] | 14 [12; 15] | 15.5 [14; 16] | 0.055 |
| Mean adherence (h)a | 7.3 [5.1; 10.1] | 7.2 [5.5; 8.6] | 8.6 [5.1; 10.7] | 0.549 |
| Residual AHI (events/h)a | 1.7 [0.4; 8.2] | 2.5 [0.4; 11.9] | 1 [0.2; 2.4] | 0.278 |
BMI, body mass index; NIV, noninvasive ventilation; IPAP, inspiratory positive airway pressure; Vt, tidal volume; EPAP, expiratory positive airway pressure; AHI, apnea hypopnea index.
Results are expressed as number (%) or median [IQR].
p-Value are given for the differences at baseline between patients using nasal compared to those using oronasal interface.
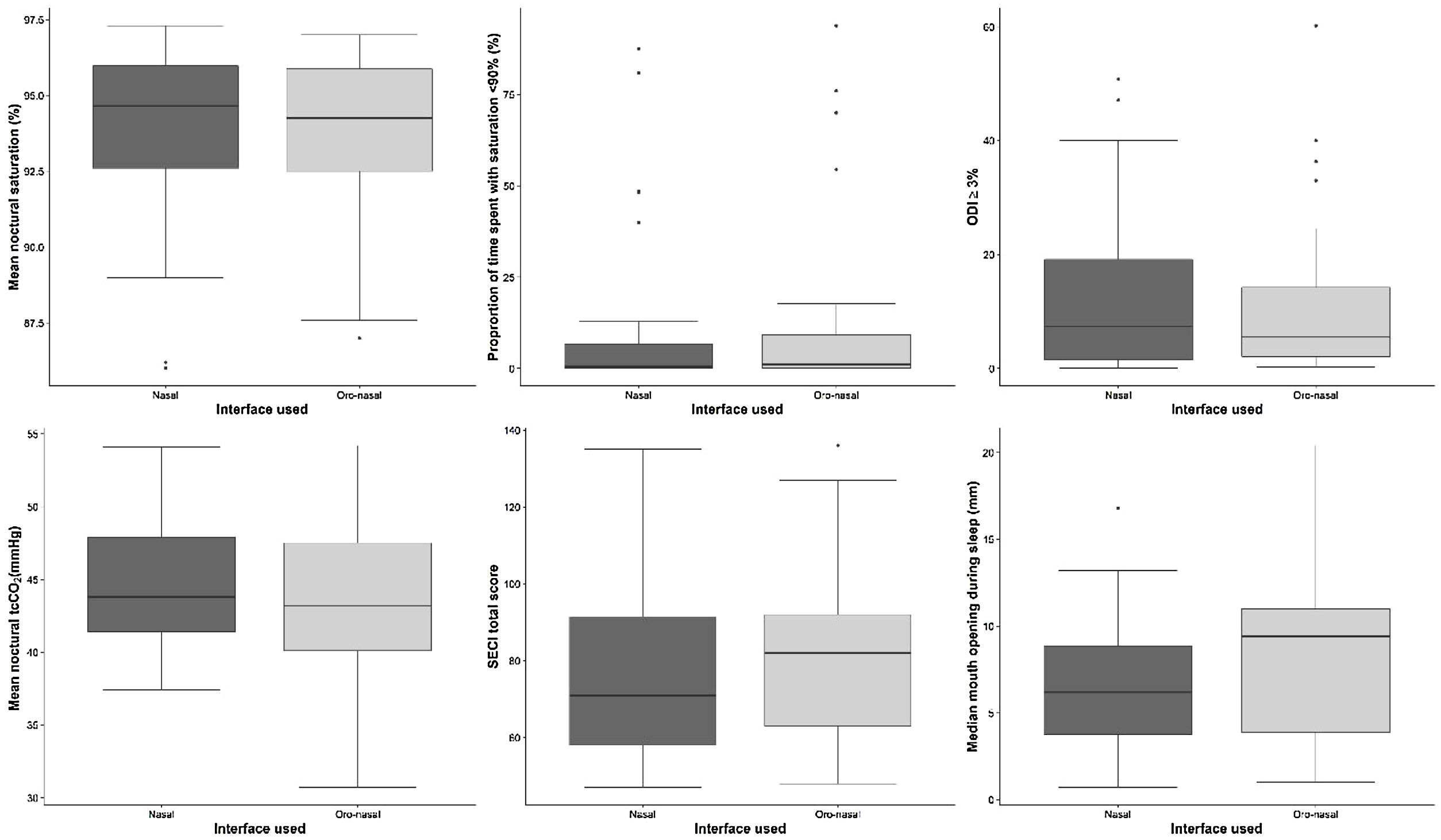
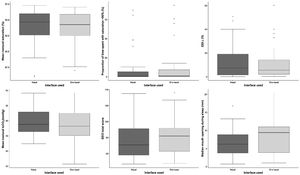
There were no significant differences in mean nocturnal SpO2 between the interfaces (ITT, p=0.73; PP, p=0.59) (Fig. 2).
Secondary outcomesThere were no differences in secondary outcomes between the interfaces (Fig. 2): percentage of sleep recording spent with SpO2<90% (ITT, p=0.33; PP, p=0.57); oxygen desaturation index (ITT, p=0.74; PP, p=0.59); mean nocturnal transcutaneous partial pressure in CO2 (ITT, p=0.13; PP, p=0.11); mean mouth opening during sleep (ITT, p=0.1931; PP, p=0.054); level of non-intentional leaks (ITT, p=0.58; PP, p=0.80) or side-effects according to each type of mask (ITT, p=0.46; PP, p=0.98). Additionally, no differences were found between masks in terms of mean NIV adherence, residual AHI, nadir SpO2, time spent with leaks or time spent with tcCO2>55mmHg.
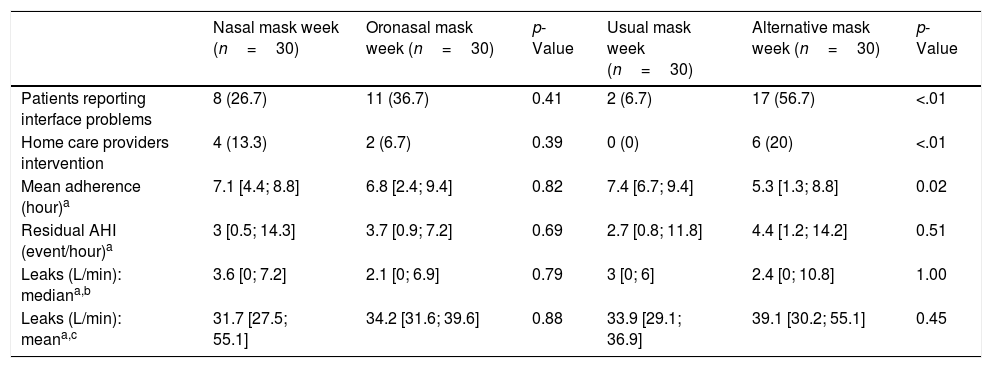
There were no differences in the ratio of patients who reported interface problems or the number of home care provider interventions between interfaces (Table 2).
Follow-up variables during the week with both interfaces.
| Nasal mask week (n=30) | Oronasal mask week (n=30) | p-Value | Usual mask week (n=30) | Alternative mask week (n=30) | p-Value | |
|---|---|---|---|---|---|---|
| Patients reporting interface problems | 8 (26.7) | 11 (36.7) | 0.41 | 2 (6.7) | 17 (56.7) | <.01 |
| Home care providers intervention | 4 (13.3) | 2 (6.7) | 0.39 | 0 (0) | 6 (20) | <.01 |
| Mean adherence (hour)a | 7.1 [4.4; 8.8] | 6.8 [2.4; 9.4] | 0.82 | 7.4 [6.7; 9.4] | 5.3 [1.3; 8.8] | 0.02 |
| Residual AHI (event/hour)a | 3 [0.5; 14.3] | 3.7 [0.9; 7.2] | 0.69 | 2.7 [0.8; 11.8] | 4.4 [1.2; 14.2] | 0.51 |
| Leaks (L/min): mediana,b | 3.6 [0; 7.2] | 2.1 [0; 6.9] | 0.79 | 3 [0; 6] | 2.4 [0; 10.8] | 1.00 |
| Leaks (L/min): meana,c | 31.7 [27.5; 55.1] | 34.2 [31.6; 39.6] | 0.88 | 33.9 [29.1; 36.9] | 39.1 [30.2; 55.1] | 0.45 |
AHI, apnea hypopnea index.
Results are expressed as number (%) or median [IQR].
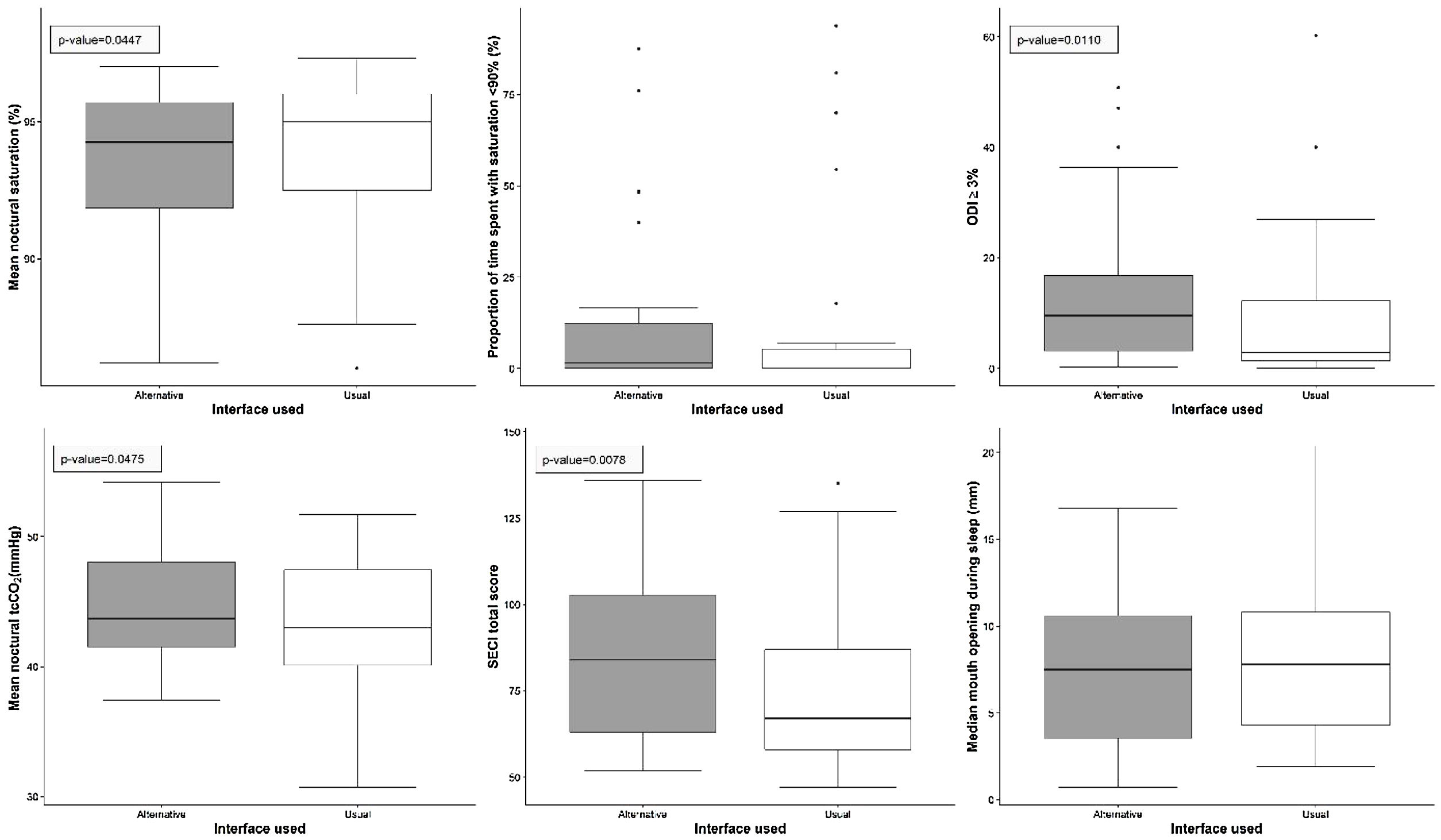
Post hoc comparisons showed that changing from one interface to another reduced NIV efficacy: oxygen desaturation index (ITT, p=0.01; PP, p=0.01); mean nocturnal transcutaneous partial pressure in CO2 (ITT, p=0.048; PP, p=0.09); side-effects (ITT, p=0.008; PP, p=0.02).
During the week with the alternative interface, mask problems ratio and home care provider intervention numbers were higher (p<0.01) and mean adherence was lower (p=0.02) (Table 2). Accordingly, patients perceived an impact on adherence during the week with the alternative interface (Table 3).
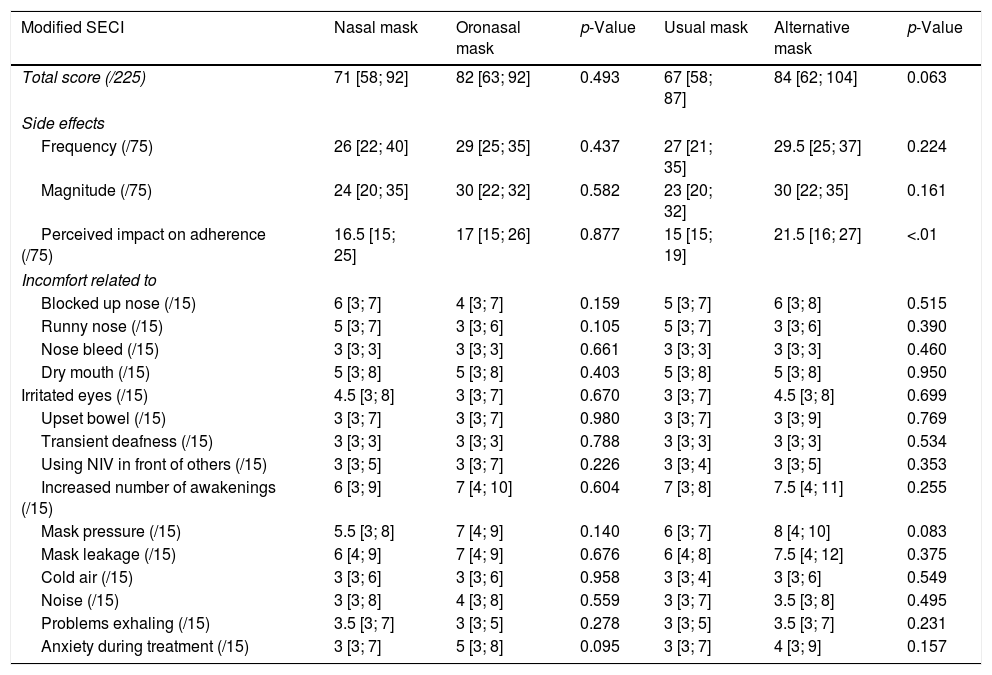
Side effects according to the type of mask (details of the SECI score).
| Modified SECI | Nasal mask | Oronasal mask | p-Value | Usual mask | Alternative mask | p-Value |
|---|---|---|---|---|---|---|
| Total score (/225) | 71 [58; 92] | 82 [63; 92] | 0.493 | 67 [58; 87] | 84 [62; 104] | 0.063 |
| Side effects | ||||||
| Frequency (/75) | 26 [22; 40] | 29 [25; 35] | 0.437 | 27 [21; 35] | 29.5 [25; 37] | 0.224 |
| Magnitude (/75) | 24 [20; 35] | 30 [22; 32] | 0.582 | 23 [20; 32] | 30 [22; 35] | 0.161 |
| Perceived impact on adherence (/75) | 16.5 [15; 25] | 17 [15; 26] | 0.877 | 15 [15; 19] | 21.5 [16; 27] | <.01 |
| Incomfort related to | ||||||
| Blocked up nose (/15) | 6 [3; 7] | 4 [3; 7] | 0.159 | 5 [3; 7] | 6 [3; 8] | 0.515 |
| Runny nose (/15) | 5 [3; 7] | 3 [3; 6] | 0.105 | 5 [3; 7] | 3 [3; 6] | 0.390 |
| Nose bleed (/15) | 3 [3; 3] | 3 [3; 3] | 0.661 | 3 [3; 3] | 3 [3; 3] | 0.460 |
| Dry mouth (/15) | 5 [3; 8] | 5 [3; 8] | 0.403 | 5 [3; 8] | 5 [3; 8] | 0.950 |
| Irritated eyes (/15) | 4.5 [3; 8] | 3 [3; 7] | 0.670 | 3 [3; 7] | 4.5 [3; 8] | 0.699 |
| Upset bowel (/15) | 3 [3; 7] | 3 [3; 7] | 0.980 | 3 [3; 7] | 3 [3; 9] | 0.769 |
| Transient deafness (/15) | 3 [3; 3] | 3 [3; 3] | 0.788 | 3 [3; 3] | 3 [3; 3] | 0.534 |
| Using NIV in front of others (/15) | 3 [3; 5] | 3 [3; 7] | 0.226 | 3 [3; 4] | 3 [3; 5] | 0.353 |
| Increased number of awakenings (/15) | 6 [3; 9] | 7 [4; 10] | 0.604 | 7 [3; 8] | 7.5 [4; 11] | 0.255 |
| Mask pressure (/15) | 5.5 [3; 8] | 7 [4; 9] | 0.140 | 6 [3; 7] | 8 [4; 10] | 0.083 |
| Mask leakage (/15) | 6 [4; 9] | 7 [4; 9] | 0.676 | 6 [4; 8] | 7.5 [4; 12] | 0.375 |
| Cold air (/15) | 3 [3; 6] | 3 [3; 6] | 0.958 | 3 [3; 4] | 3 [3; 6] | 0.549 |
| Noise (/15) | 3 [3; 8] | 4 [3; 8] | 0.559 | 3 [3; 7] | 3.5 [3; 8] | 0.495 |
| Problems exhaling (/15) | 3.5 [3; 7] | 3 [3; 5] | 0.278 | 3 [3; 5] | 3.5 [3; 7] | 0.231 |
| Anxiety during treatment (/15) | 3 [3; 7] | 5 [3; 8] | 0.095 | 3 [3; 7] | 4 [3; 9] | 0.157 |
Complaints reported by participants about the alternative interface were predominantly mask intolerance, leaks, mouth dryness and cutaneous lesions. Problems relating to the underlying condition and impaired autonomy were reported especially with the use of an oronasal interface (impaired communication leading to the incapacity to call for help if needed, compromised vocal command device use, compromised hydration autonomy at night, etc.)
DiscussionThis original randomized cross-over study found no differences between nasal and oronasal masks in terms of nocturnal NIV efficacy and NIV tolerance. However, during the transition between the interfaces mean nocturnal SpO2 and adherence decreased and oxygen desaturation, mean nocturnal tcCO2 and side effects increased.
In a previous RCT that included a heterogeneous group of patients with chronic respiratory failure, Willson et al. reported poorer quality of sleep, impaired tolerance (less comfort, more perceived leaks) with the use of oronasal masks compared to nasal masks.13 However, similarly to the present study, they also found no differences in terms of nocturnal gas exchange or sleep disordered breathing between the two interfaces. In Willson et al.’s study, all subjects were treated with nocturnal NIV at home via a nasal mask.13 Therefore, the reduced tolerance reported with oronasal mask may simply reflect difficulty switching from the usual interface to another. This hypothesis is supported by the results of the post hoc analyses in the present study.
We hypothesized that the oronasal mask might increase upper airway obstruction and jeopardize NIV efficacy. This was based on a recent case report in a patient with ALS (without bulbar involvement) that showed that use of an oronasal mask with NIV caused persistent obstructive respiratory events, with oxygen desaturations, sleep fragmentation and persistent sleep hypercapnia.10 In the field of obstructive sleep apnea, the impact of the mask type has been more extensively assessed in patients treated with continuous positive airway pressure (CPAP). Similar reports of persistent obstructive respiratory events with the use of oronasal masks have been published.15–17 Although, the exact mechanisms have not been completely elucidated, two main hypotheses have been put forward: (i) mechanical constraint of the chin induced by the oronasal mask and traction of straps may push the mandible posteriorly during sleep18; (ii) air pressure blowing into the mouth and oral breathing may push the tongue backward and reduce pharyngeal patency.15,19,20 The results of the present study did not suggest that oronasal masks increased upper airway obstruction, despite the fact that this is a common occurrence in patients with neuro-muscular diseases. Although mask-related upper airway obstruction has been shown to be highly prevalent in patients with neuromuscular disorders, it may be more prevalent in patients with ALS not included in the present study.11 Indeed, the level of EPAP was relatively low both in patients with nasal and oronasal mask suggesting that the patients included may be unlikely to have severe upper airway collapsibility. Finally, our specialized nurses are strongly aware of this issue and thus teach patients and primary caregivers to avoid any over tightening of mask straps during the positioning.
The patients included in this study, were all treated with home nocturnal NIV and the median adherence was greater than 7h/night. The post hoc analysis showed that during the transition to a new interface, NIV efficacy was reduced and side effects increased. This information is important for clinicians since in practice, interfaces are often changed by home care providers or on the patient's own initiative with no systematic medical supervision. In the present study, despite close follow up during the week of change (and in-hospital initial mask choice and adjustment), mean nocturnal SpO2 decreased and mean tcCO2 increased as did the oxygen desaturation index and the number of side effects occurs. To our knowledge, no study has previously addressed the acute consequences and risks related to mask change in patients with neuromuscular diseases with nocturnal NIV. Our results suggest that information regarding changing masks should be provided during patient and caregiver education to ensure that appropriate medical supervision is sought, should they decide to change interface.
LimitationsAlthough our study was powered to detect a mean difference of mean nocturnal SpO2 of 2±2% between oronasal and nasal masks, it is possible that the study was under powered due to the fact 5 patients did not tolerate the alternative interface.
The patients were not naive to NIV and the heterogeneity of the underlying diseases and level of autonomy could also have impacted the results. Yet, this recruitment reflects daily practice in tertiary care hospitals who deals with respiratory care in NMD which remain rare diseases (therefore it would have not been possible to complete this study in a reasonable time-frame with naive patients).
In our study, oronasal mask patients appeared to not have significant enough issues with upper airway resistance. We should have focused on more carefully selected patients with high upper airway resistance, residual events or low efficacy of oronasal NIV and assess the benefits to switch interface in those patients.
Finally, the period of adaptation to the alternative interface might have been too short.
Because of the high specificity of the sample included, the results of this study should not be extrapolated to other populations such as people with obesity hypoventilation syndrome and chronic obstructive pulmonary disease.
ConclusionThis study found no difference between oronasal and nasal masks in patients with neuromuscular diseases under long term nocturnal NIV in terms of treatment tolerance and efficacy. The results showed that treatment efficacy may be reduced during transition between interfaces, thus changes in masks should be carried out under close medical supervision.
Data sharing statementIndividual participant data that underlie the results reported in this article after deidentification (including data dictionaries) will be shared beginning 3 months and ending 5 years following article publication with researchers who provide a methodologically sound proposal. Data will be shared for analyses to achieve aims in the approved proposal. Proposals should be directed to antoine.leotard@aphp.fr. To gain access, data requestors will need to sign a data access agreement.
Authors’ contributionsSubstantial contributions to study conception and design, acquisition of data, or analysis and interpretation of data: A.L., JC.B., JL.P., F.L., H.P., M.L., R.T., M.D., C.SR,A.S., K.L. Statistical analyses: N.D. Drafting the article or revising the article critically for important intellectual content: A.L., JC.B., F.L., JL.P., M.L. Final approval of the version to be published: all co-authors. Guarantor of the paper, taking responsibility for the integrity of the work as a whole: A.L.
FundingThis study received a grant from “Association Française contre les Myopathies – AFM”.
Conflict of interestThe authors declare that they have no conflict of interest.
We would like to thank Richard Yvinec, Vincent Delord, Mickael Berlusconi, Steffy Masucco, Galina Penkov, Sebastien Bailly, Marie Peeters, Mélanie Lopez, Pierre Ansay to have contribute to this work with human or technical assistance.