The aim of this study is to analyze the expression of the main oxidant scavenger superoxide dismutase (EC-SOD), its main binding protein Fibulin-5 and several oxidative and nitrosative-derived products in the lung of COPD patients and controls.
Materials and methodsLung tissue samples from 19 COPD patients and 20 control subjects were analyzed. The architecture of elastic fibres was assessed by light and electron microscope histochemical techniques, and levels of EC-SOD and fibulin-5 were analyzed by immunohistochemistry and RT-PCR. The impact of oxidative stress on the extracellular matrix was estimated by immunolocalization of 4-hydroxynonenal (4-HNE), malondialdehyde (MDA) and 3-nitrotyrosine (3-NYT) adducts.
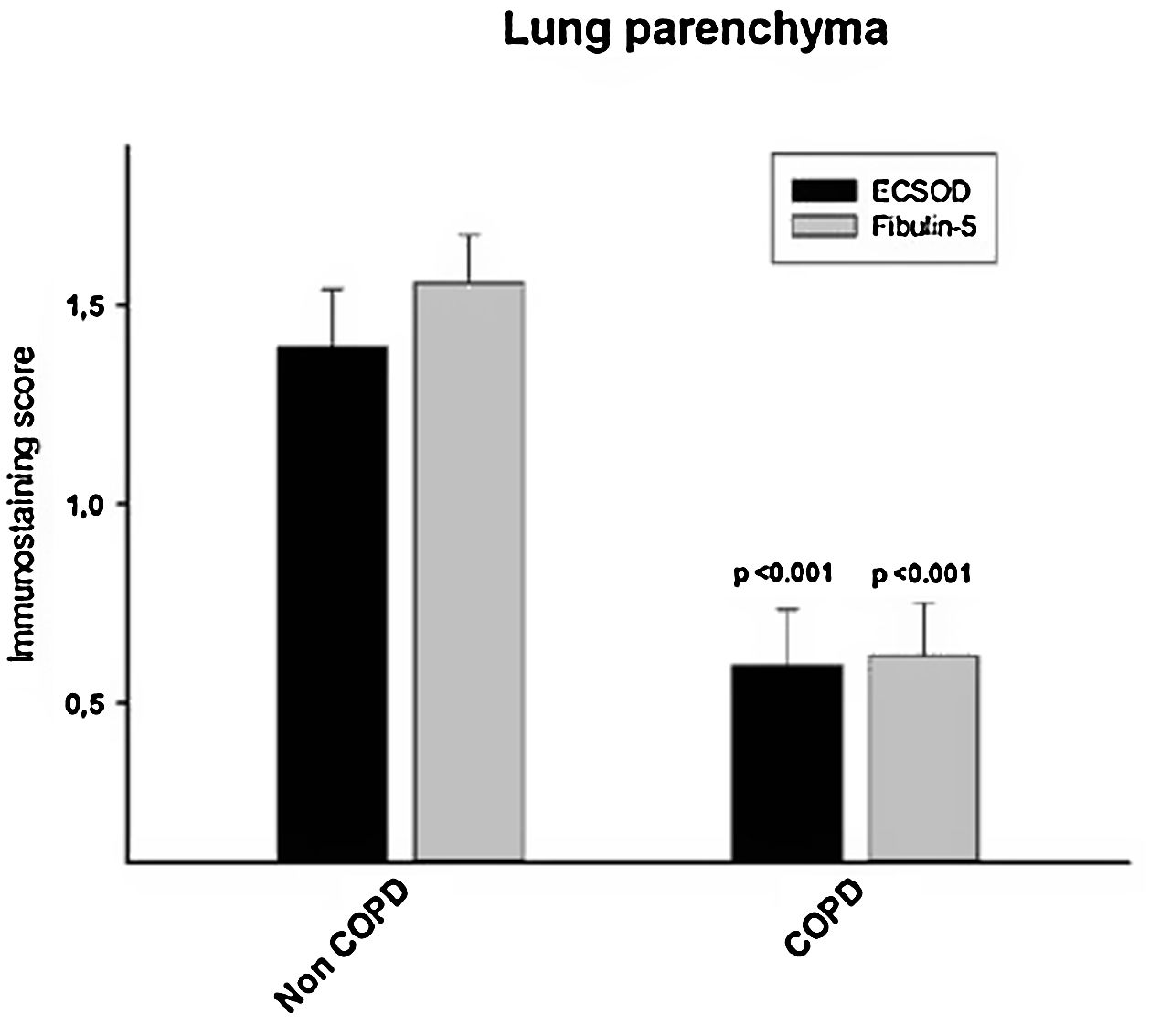
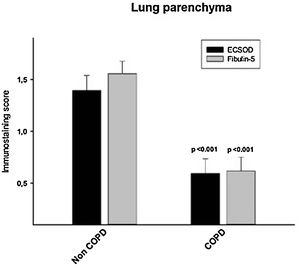
ResultsAlveolar walls of COPD patients exhibited abnormal accumulations of collapsing elastic fibres, showing a pierced pattern in the amorphous component. The semiquantitative analysis revealed that COPD patients have a significantly reduced expression of both EC-SOD and fibulin-5 (0.59±0.64 and 0.62±0.61, respectively) in alveolar, bronchiolar and arteriolar walls compared to control subjects (1.39±0.63 and 1.55±0.52, respectively, p<0.05). No significant changes in mRNA levels of these proteins were observed between groups. Among the oxidation markers, malondialdehyde was the best in distinguishing COPD patients.
ConclusionsCOPD patients show a reduced expression of EC-SOD and fibulin-5 in the lung interstitium. Paralleling the reduction of EC-SOD levels, the decrease of fibulin-5 expression in COPD lungs supports the hypothesis of an impaired pulmonary antioxidant response in COPD patients.
Chronic obstructive pulmonary disease (COPD) is a global health problem, which has been projected to produce 4.7 million deaths worldwide in 2020.1 Prolonged exposure to tobacco smoking and less frequently to other particles and gases produce a chronic inflammatory pattern leading to different pulmonary histopathological features, the two most predominant being small airways narrowing and emphysematous parenchymal destruction.2
Oxidative stress, which is defined as an imbalance between oxidizing molecules and antioxidant defences, play a pivotal role in COPD pathogenesis.3 Oxidants activate the transcription of the pro-inflammatory factor NF-kB in macrophages and bronchial and alveolar epithelial cells.4 This leads to the recruitment and activation of other inflammatory cells such as neutrophils, which can also induce the additional release of reactive oxygen intermediates and proteases5 involved in emphysematous destruction.6 In addition to this, it has also been reported that COPD patients have diminished antioxidant defences, which can enhance this oxidative stress-driven inflammatory response.7 Specifically, it has been shown that the levels of Nuclear factor erythroid-2-related factor-2 (Nrf2), the master transcription factor involved in the antioxidant defence, and its stabilizer DJ1, are reduced in lungs of COPD patients.7
The activity of antioxidant enzymes is crucial against the action of free radicals, either by stabilizing or deactivating them before they damage cellular components.8 Among them, extracellular superoxide dismutase (EC-SOD, EC 1.15.1.1) is a main oxidant scavenger in extracellular space, including lung interstitium.9 Thus, high levels of EC-SOD can be found in lung extracellular matrix,10 where it binds with heparin sulphate proteoglycan and fibulin-5, an integrin-binding matricellular protein involved in elastogenesis.11,12 There, EC-SOD catalyzes the reduction of superoxide radical to hydrogen peroxide, a less reactive molecule.13 Moreover, several preclinical studies have also shown that EC-SOD can effectively inhibit lung inflammation.14,15 In this regard, it has been suggested that the anti-inflammatory activity of EC-SOD could be achieved either by reducing the release of pro-inflammatory cytokines from phagocytes or by inhibiting the production of extracellular matrix fragments that act as chemoattractants for inflammatory cells.14,16
As the only antioxidant scavenging superoxide in the pulmonary extracellular compartment, EC-SOD is thought to play a relevant role in COPD. Hence, it has been shown that smokers carrying the EC-SOD polymorphism R213G have a reduced risk of developing COPD.17,18 Moreover, it has been reported that COPD patients have higher levels of EC-SOD in induced sputum but not in BALF as compared to control non-smokers.19 Regarding EC-SOD levels in lung tissue of COPD patients, data available are scarce and contradictory. Whereas one study found decreased expression of EC-SOD in lung vessels and bronchioles of COPD patients,19 another reported no differences among subjects with COPD and controls.20
The aim of the present study is to analyze the antioxidant expression in COPD lungs not only by determining levels of EC-SOD, but also those of its binding protein fibulin-5. Expression of these two molecules was analyzed through immunohistochemistry and RT-PCR in lung tissue from COPD patients and control subjects. The localization of EC-SOD in emphysematous walls was also assessed at an ultrastructural level. Lung expression of 4-hidroxynonennal (4-HNE), malondialdehyde (MDA) and 3-nitrotyrosine (3-NTS) was also analyzed by immunochemistry.
Materials and methodsSubjectsThe study included thirty-nine patients who underwent lung resection for non-obstructive peripheral lung tumours or were subjected to double lung transplantation for very severe COPD at Vall d’Hebron University Hospital (Barcelona, Spain). The Hospital's ethics committee approved the study and written informed consent was obtained from all subjects. Pulmonary function was assessed through spirometry according to ERS recommendations21 using standard equipment (Masterlab; Jaeger, Würzburg, Germany).
Subjects were divided into COPD (N=20) and non-COPD patients (N=19). All of them were free of acute lung infections in the last three weeks and none had received chemotherapy before surgery.
Tissue processingThe excised lungs or lobes were inflated immediately after surgery at 30cm of water pressure for 1h with 4% (w/v) formaldehyde in 0.1M phosphate-buffered saline solution (pH 7.4). In patients with lung cancer, tissue samples were taken from the non-malignant lung tissue as demonstrated by pathologist examination.
Samples to be analyzed by light microscopy were then immersed in fixative for 24h. After that, 2cm×2cm×0.3cm randomly selected tissue blocks were excised, embedded in paraffin, cut into 4-μm sections and mounted on positive charged slides Starfrost Plus (Menzel-Gläser, Braunschweig, Germany). Four consecutive serial sections from a representative block from each patient were selected for histologic, histochemical and immunohistochemical analyses. Every first section was stained with haematoxylin and eosin (H&E) to reveal lung histology. The second and third sections were reserved for immunolocalization of EC-SOD and fibulin-5. The last section in each set was stained with Verhoeff's haematoxylin to detect pulmonary elastic fibres.22
On another front, after initial fixation, samples to be studied by transmission electron microscopy were cut into 1–2-mm thick pieces and fixed in 1% (w/v) paraformaldehyde, 0.1% (v/v) glutaraldehyde in 0.1M phosphate-buffered saline (PBS) solution for 2h at room temperature. After that, a post-fixation with 1% (w/v) osmium tetroxide in cacodylate buffer was performed for 1h. Embedding was performed in Lowycril K4M resin according to standard procedures.23
ImmunohistochemistrySamples to be analyzed by light microscopy were deparaffinised and processed at 121°C in a 2100 Antigen Retriever (PickCell Laboratories, Leiden, Netherland) in Tris–HCl 10mM-EDTA 0.5mM pH 9.0 buffer for EC-SOD slides and in Citrate 10mM pH 6.0 for fibulin-5 slides. After antigen unmasking, sections were washed three times in PBS and submerged in 3% hydrogen peroxide, 10% methanol in PBS for 15min. After washing three times with PBS, nonspecific binding was suppressed by incubation with blocking solution. Following that, samples were incubated at 4°C overnight with polyclonal anti-EC-SOD antibody (1:500, Abnova, Taipei, China) monoclonal anti-fibulin 5 antibody (1:750, Abcam, Cambridge, UK), rabbit polyclonal anti-4-HNE antibody (1:300; Calbiochem, San Diego, CA, USA), rabbit polyclonal anti-MDA antibody (1:400; Abcam Ltd., Cambridge, UK) or with rabbit polyclonal anti-NT antibody (1:90; Upstate Biotechnology, Inc., Lake Placid, NY, USA) in blocking solution. Negative controls were obtained by incubating a slide with rabbit IgG (1:500, ab37415, Abcam, Cambridge, UK) or mouse IgG (1:200, ab37355, Abcam). On the following day, sections were washed in PBS and incubated with biotinylated anti-Rabbit or ant-Mouse IgG antibody (Vector Laboratories; Burlingame, CA) for 1h at room temperature. Immunostaining was performed using the avidin–biotin complex (ABC) immunoperoxidase method (Vectastain Elite ABC kit; Vector Laboratories, Burlingame, CA, USA) with a 3,3′-diaminobenzidine tetrahydrochloride (DAB) reaction. Finally, the slides were counterstained in haematoxylin, dehydrated, and mounted. For analysis of immunostained lung tissue, two independent observers (JGV and JO) assessed the intensity and extent of immunoreactivity of EC-SOD in a double-blind fashion through a semi quantitative score (0=no staining; 1=weak/intermediate staining; 2=strong staining). At least twelve fields of each sample were randomly and systematically sampled to assess EC-SOD and fibulin-5 staining in alveolar interstitium.
Samples to be analyzed by transmission electron microscopy (TEM) were hydrated in a PBS 0.1M glycine 0.1M solution and non-specific binding was suppressed by incubation in 2% ovalbumin in PBS 0.1M. After that, samples were incubated with the primary antibody (anti-EC-SOD, 1:150, Abnova, Taipei, China) in blocking solution for2h. Then, samples were washed three times in PBS 0.1M glycine 0.1M solution and incubated with Goat Anti-Rabbit IgG with 10nm Gold preadsorbed (1:10, ab27234, Abcam) for1h. Finally, samples were washed in PBS 0.1M and jet-washed in 10ml of milli-Q water prior to counterstaining and mounting.
Quantitative real-time PCRIsolation of total RNA from tissue samples was performed using the AllPrep DNA/RNA Mini Kit (QIAGEN, Hilden, Germany). After reverse transcription of mRNA into complementary DNA (cDNA), qRT-PCR was performed using primer and probe sets for ECSOD and fibulin-5 from TaqMan Gene Expression Assays purchased from Applied Biosystems (Foster City, CA, USA). Data from qRT-PCR reactions was analyzed by the comparative Ct (2-ΔΔCt) quantification method. Relative expression levels of ECSOD and fibulin-5 genes were determined using 18S mRNA as an endogenous control. Three independent experiments were performed and results were expressed as the normalized mean of the relative quantification (RQ) of the tested transcripts. Non-template controls exhibited no signal.
Statistical analysisThe descriptive statistical analysis included mean±SEM or SD for each parameter and patient group. Data were compared between the two groups using the Chi-square and Mann–Whitney U-tests. The significance level was set at p<0.05. All analyses were performed using Statgraphics Centurion XV (StatPoint, Inc., Warrenton, VA, USA).
ResultsDemographic and clinical findingsThe two clinical groups were similar regarding age and body-mass index, with a predominance of males and tobacco smoking in the COPD group (Table 1). As expected, the mMRC dyspnoea score, the number of previous exacerbations in the last year and the use of inhaled corticosteroids were higher in COPD patients. In this group, the microscopic (LM) and macroscopic emphysema (MED) degree was increased and the spirometry variables were reduced compared with non-COPD patients.
Clinic and demographic characteristics of patients.
| Non-COPD (n=19) | COPD (n=20) | p-For trend | |
|---|---|---|---|
| Age (years) (SD) | 59.26 (12.16) | 61.00 (9.20) | 0.8219 |
| Sex. male vs. Female | 8/11 | 18/2 | 0.002 |
| BMI (kg/m2). (SD) | 27.21 (4.11) | 25.17 (3.81) | 0.1774 |
| Smokers (n, Pack-years (SD)) | 10, 28.84 (33.86) | 20, 58.05 (23.64) | 0.0044 |
| Dyspnoea score (mMRC>2). n(%) | 2/19 (10.53) | 11/20 (55.00) | 0.02 |
| Any exacerbations in the last year. n(%) | 3/19 (15.79) | 14/20 (70.00) | <0.001 |
| Patients treated with ICS. n(%) | 2/19 (10.53 | 14/20 (70.00) | <0.001 |
| Patients with oxygen. n(%) | 0/19 (0.00) | 6/20 (30.00) | <0.001 |
| FEV1 postbronchodilator (%pred). (SD) | 93.92 (18.38) | 38.36 (21.63) | <0.001 |
| FEV1/FVC postbronchodilator (%pred). (SD) | 80.26 (7.36) | 44.58 (13.00) | <0.001 |
| MED % (SD) | 5.92 (8.90) | 50.0 (27.34) | <0.001 |
| LM (mm) (SD) | 220.74 (81.57) | 489.54 (317.92) | 0.0017 |
SD: Standard Deviation; BMI: Body Mass Index; mMRC: Modified Medical Research Council Dyspnoea Scale; ICS: Inhaled Corticosteroids; MED: Macroscopic Emphysema Degree; LM: Mean Airspace Chord length.
In order to determine whether the architecture of elastic fibres in alveolar walls was different among COPD patients and controls, we used Verhoeff's staining, a method that allows highlighting elastic fibres.22
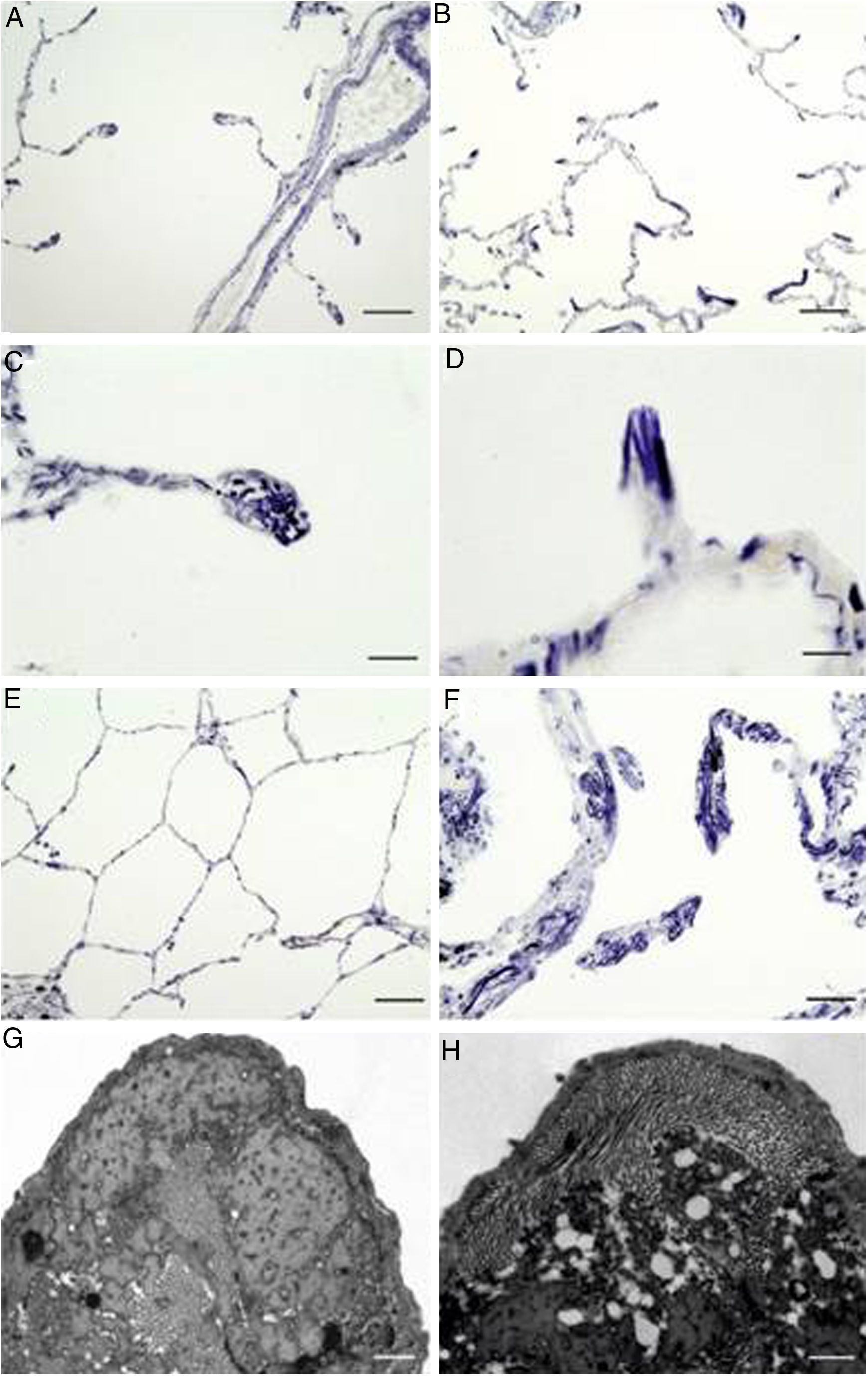
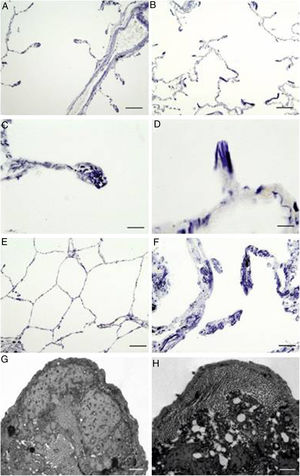
As expected, sections from COPD patients exhibited an altered elastin network when compared to control subjects (Fig. 1). Thus, there were anomalous accumulations of elastic fibres in periemphysematous alveolar walls of COPD patients, suggesting a tissue collapse in periemphysematous areas (Fig. 1B, D and F). By contrast, control samples did not show these histological alterations (Fig. 1A, C and E).
Localization of elastic fibres in lung samples from COPD patients and control subjects. Verhoeff's staining of elastic fibres in alveolar ducts of a healthy control (A) and a COPD patient (B), with arrows indicating accumulation of elastic fibres in collapsing walls of emphysematous areas (bars=100μm). Detail of a tip of an alveolar wall of a control subject (C), showing the normal distribution of elastic fibres, while the alveolar walls of the emphysematous (D) areas appear collapsed in COPD patients (bars=25μm). Micrography of normal lung parenchyma of a control subject (E, bar=100μm) and emphysematous parenchyma of COPD patient (F, bar=50μm), showing prominent elastic accumulations. (G) Electron microscopy of a tip of a normal alveolar wall (bar=500nm). (H) Electron microscopy of a collapsing alveolar wall, showing vacuoles and a pierced pattern in the amorphous component (bar=500nm).
When the tips of alveolar walls were observed under TEM, COPD patients showed vacuolar changes in the amorphous component of elastic fibres, resulting in a pierced pattern (Fig. 1H). These alterations were absent in control samples (Fig. 1G).
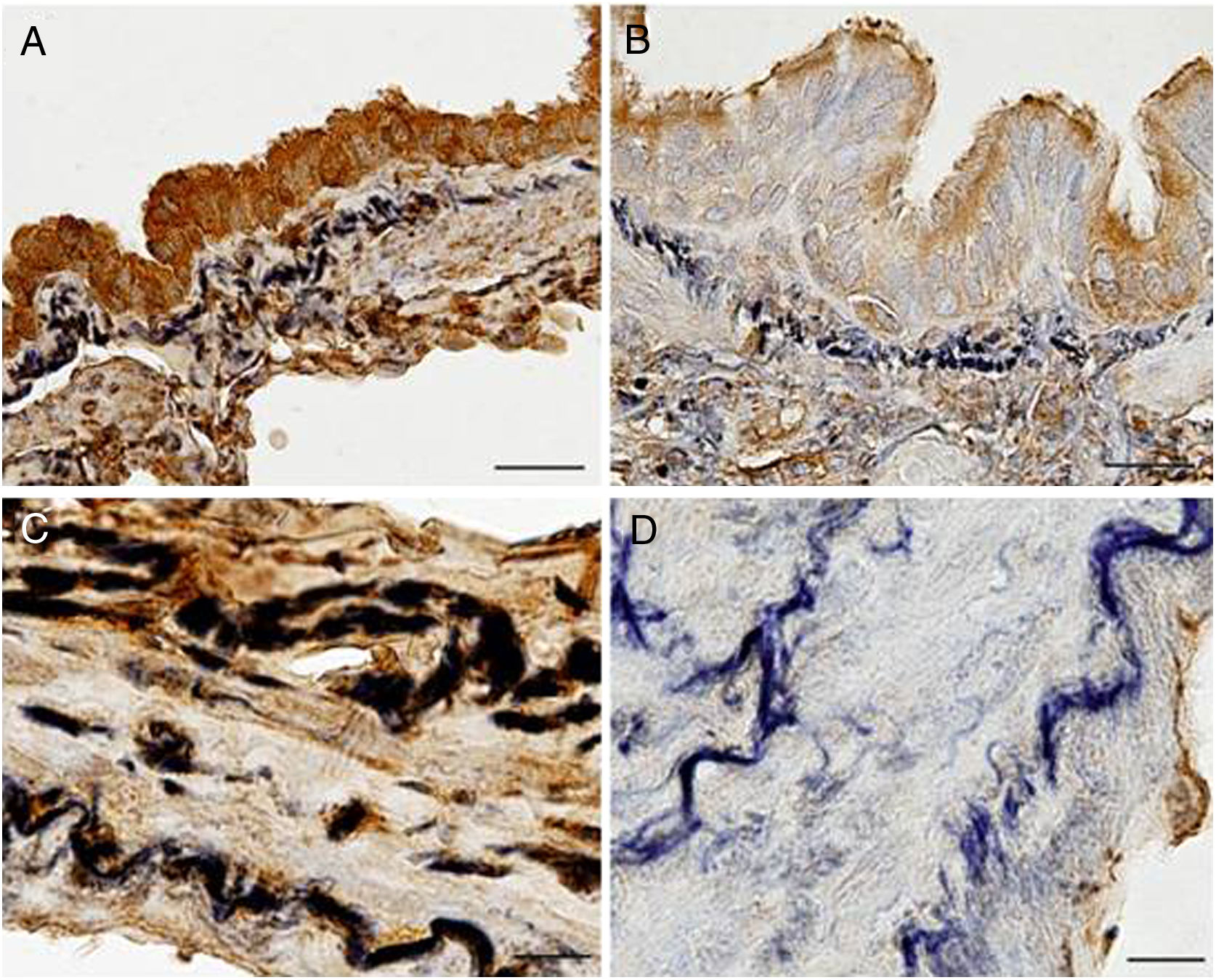
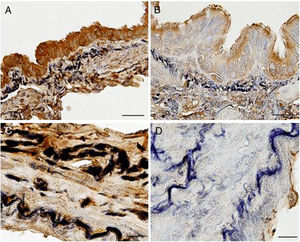
ImmunohistochemistryLight microscopy study showed that EC-SOD was immunolocalised in airway and parenchymal epithelial cells (primarily in the apical area), as well as in alveolar macrophages and endothelial cells. EC-SOD was also present in the perimeter of elastic fibres of pulmonary vessels and alveolar walls, whereas collagen showed a weaker or no staining. Interestingly, EC-SOD immunostaining was higher in airway and vascular walls of control subjects (Fig. 2A and C) when compared to COPD patients (Fig. 2B and D). Regarding fibulin-5, it was also observed mainly in airway and vascular walls, as well as in alveolar interstitium of lung samples (Fig. S1).
Immunolocalization of EC-SOD in airway and vascular walls of COPD patients and control subjects. Presence of EC-SOD was markedly higher in airways (A, bar=25μm) and vascular walls (C, bar=10μm) of controls subjects when compared to those of COPD patients (B, bar=25μm, and D, bar=25μm, respectively).
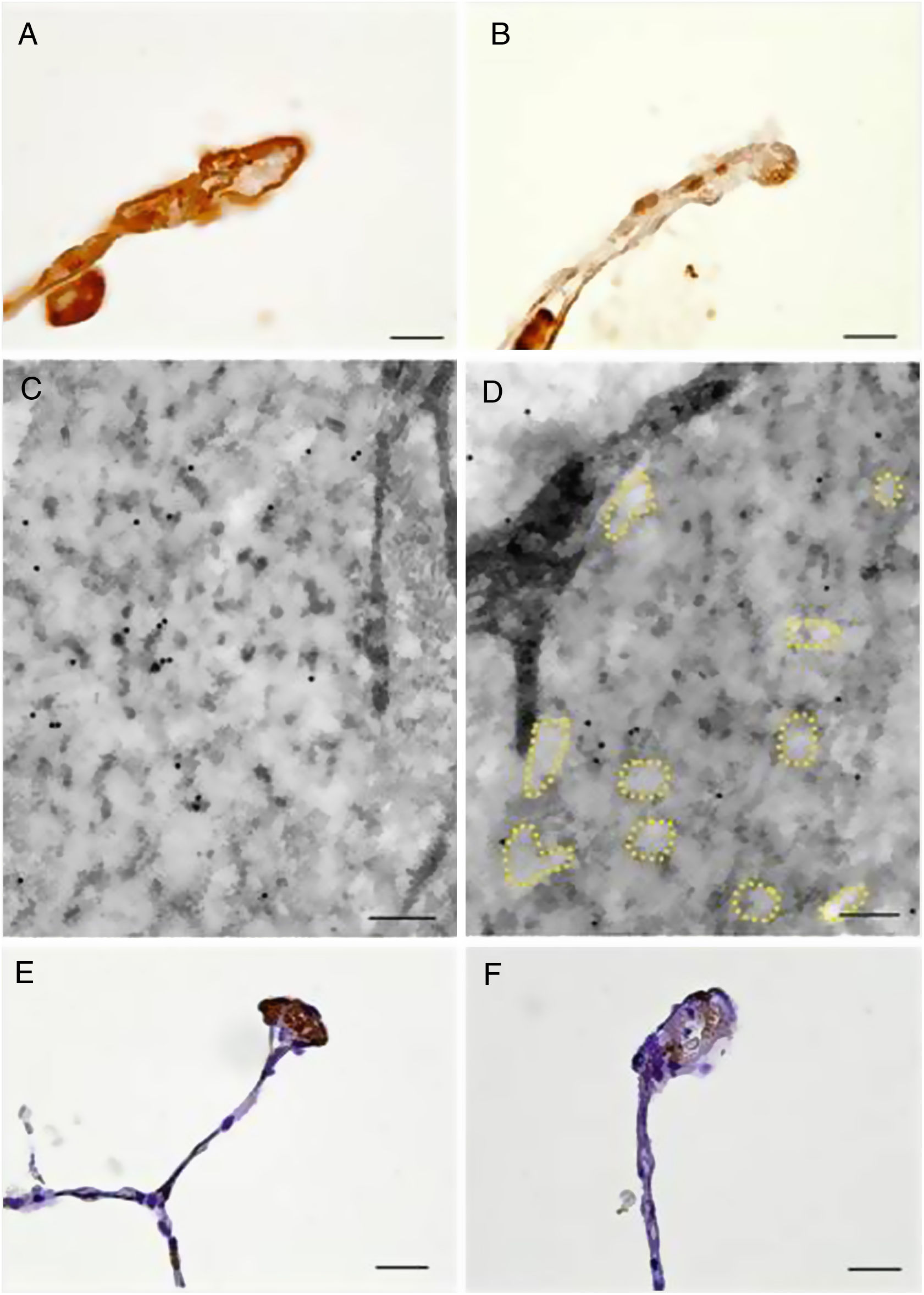
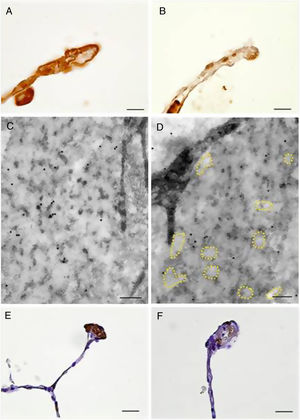
Moreover, immunolocalization of EC-SOD and fibulin-5 allowed us to study the interstitial expression of these proteins. Alveolar, bronchiolar and arteriolar walls of patients with COPD showed a reduced expression of EC-SOD and fibulin-5 when compared to control subjects (Fig. 3 and Fig. S1). The additional immunohistochemical analysis by TEM confirmed the reduced expression of EC-SOD in interstitium of COPD patients, and revealed that EC-SOD was tightly associated with fibrillary components in the unaffected elastic fibres (Fig. 3 and Fig. S1). Moreover, in altered fibres, the decrease in EC-SOD labelling was associated with the loss of the fibrillary component and the partial degradation of elastin, which produced the pierced pattern (Fig. 3 and Fig. S1).
Immunolocalization of EC-SOD and fibulin-5 in alveolar walls of COPD patients and control subjects. Alveolar walls of control subjects showed increased presence of EC-SOD (A, bar=10μm) and fibulin-5 (E, bar=20μm) as compared to those of COPD patients (B, bar=10μm, and F, bar=20μm, respectively). (C) and (D) Electron microscopy of immunolocalised EC-SOD in elastic fibres from alveolar walls of a control subject (C, bar=200nm) and a COPD patient (D, bar=200nm). Colloidal gold labelling indicating EC-SOD immunolocalization was mostly absent in the fibrillary component alveolar elastic fibres of COPD samples. Dashed lines indicate loss of fibrillary component.
Furthermore, when the expression of EC-SOD and fibulin-5 in alveolar, bronchiolar and arteriolar walls of COPD patients and control samples was compared through a semi-quantitative score, we found that COPD patients have a significant reduced expression of both EC-SOD and fibulin-5 (0.59±0.64 and 0.62±0.61, respectively) compared to control subjects (1.39± 0.63 and 1.55±0.52, respectively, p<0.05, Fig. 4 and Fig. S1). Additionally, levels of EC-SOD and fibulin-5 were analyzed in patients without COPD by comparing smokers (n=10) and non-smokers (n=9) and males (n=11) versus females (n=8). Immunoscore values for EC-SOD in alveolar, arteriolar and bronchiolar wall were 1.44±0.23, 1.45±0.21 and 1.67±0.12 in non-smokers and 1.35±0.20, 1.8±0.13 and 1.65±0.11 in smokers without COPD (p: 0.63, 0.12 and 0.89 respectively). The immunoscore for fibulin-5 in these compartments were 1.56±0.18, 1.72±0.11 and 1.73±0.17 in non-smokers and 1.50±0.17, 1.60±0.18 and 1.66±0.13 in smokers without COPD (p: 0.96, 0.81 and 0.51 respectively). Thus, no significant differences were observed in the expression of the antioxidant markers when comparing smokers and non-smokers. Immunoscore values for EC-SOD in alveolar, arteriolar and bronchiolar wall were 1.50±0.20, 1.50±0.20 and 1.65±0.11 in non-COPD females and 1.28±0.20, 1.78±0.15 and 1.67±0.12 in non-COPD males (p=0.37, 0.23 and 0.89, respectively. Values for fibulin-5 in the same compartments were 1.65±0.13, 1.65±0.17 and 1.80±0.11 in non-COPD females and 1.44±0.21, 1.67±0.14 and 1.56±0.18 in non-COPD males (p=0.54, 0.92 and 0.25, respectively).
Regarding oxidative stress markers, malondialdehyde was the most reliable to distinguish COPD patients. Indeed, the adducts of this lipid peroxidation product were mainly immunolocalised in the connectives tissues, such as adventitia layers, alveolar walls, arterioles and the parenchymal interstitium surrounding emphysematous areas of COPD patients, showing significant differences with non COPD samples (Fig. S2). Although 4-hydroxynonenal adducts were detected in all patients, they showed a significant increase in COPD groups (Fig. S3), while the 3-nitrotyrosine biomarker was primarily located in the tissues close to the respiratory tract and alveolar spaces of smokers (Figure S3).
Quantitative real-time PCRQuantitative PCR analyses showed that both mRNA levels of EC-SOD and fibulin-5 were lower in COPD patients than in controls (Table 2), though these differences did not reach statistical significance (p>0.05, Table 2).
DiscussionIt is widely acknowledged that an imbalance between free radicals and antioxidant defences plays a main role in COPD pathogenesis.24 However, the in situ expression of oxidative stress in lungs of COPD patients remains poorly understood. The present study shows for the first time an association between a reduced expression of EC-SOD, the main antioxidant in the lung interstitium, its binding protein fibulin-5 and parenchymal alterations in alveolar walls of COPD patients.
Recent studies assessing the pathological remodelling of elastic fibres in emphysematous lung have shown contradictory conclusions, since while some of them showed a reduction in the volume fraction of elastic fibres,25 others found an increase26 or even no changes.27 Our histochemical analysis by light microscopy revealed matrix alterations coinciding with emphysema in COPD patients. Damage consisted in frayed elastic fibres clumped into dense masses lying within remnants of collapsing alveolar walls, coinciding with pathologic changes described by Robert Wright almost sixty years ago28 When interstitium of collapsing walls were further analyzed through TEM, we observed a loss of fibrillary component, mainly in the core of the elastic fibres, together with the presence of vacuoles affecting the amorphous component. A similar pierced pattern has been described in previous TEM studies in humans and murine models of emphysema,29–31 and underpins the importance of structural abnormalities of elastic fibres in areas of emphysematous destruction.
Our results showed that lungs from COPD patients exhibit lower expression of EC-SOD in alveolar walls likely leading to an impaired antioxidant defence in these patients. Indeed, cigarette smoke and other air pollutants contain high levels of oxidants and other molecules capable of inducing the formation of free radicals in pulmonary cells and a chronic inflammatory response in COPD.32–34 Both oxidative stress and inflammation act in concert to foster the emphysematous destruction of alveolar walls and to affect the repair and maintenance of parenchymal structure.35 Among the rich network of enzymatic antioxidants that lungs possess to protect themselves from oxidative burden, EC-SOD plays a primordial role in the extracellular space, both by scavenging superoxide radicals and by inhibiting the inflammatory response.13–15 Importantly, EC-SOD activity in the lung has been extensively studied in the SOD3 mice model. Gongora and colleagues showed that loss of EC-SOD leads to severe lung damage in mice, while treatment with EC-SOD mimetic MnTBAP reduces lung injury.36 Using the same animal model, Yao and colleagues demonstrated that EC-SOD attenuates fragmentation of elastic fibres.37 Since extracellular matrix components such as elastin, heparin sulphate and hyaluronan are known to trigger lung inflammation, the authors concluded that EC-SOD protects lungs from emphysema by decreasing the inflammatory response.37 Hence, our results support the hypothesis that a reduced antioxidant defence caused by diminished levels of EC-SOD is detrimental for COPD patients by increasing the progression of emphysema.
To the best of our knowledge, only two previous papers studied pulmonary EC-SOD expression in patients with COPD. Thus, whereas Harju and co-workers reported no differences in EC-SOD immunoreactivity among non-smokers, smokers without COPD and COPD patients,20 a more recent study found a significant decrease of EC-SOD immunoreactivity around vessels and bronchioles of COPD patients compared to controls.19 Our results demonstrate a reduced expression of EC-SOD in vascular and airway walls in COPD and TEM analysis confirmed the decreased expression of interstitial EC-SOD observed by light microscopy in COPD patients. The mentioned discrepancy of results among the three studies may be due to differences in the characteristics of COPD patients (mild to moderate in the study by Harju et al. and moderate to severe in our study and the one by Regan et al.) and the different compartments analyzed. Thus the expression of EC-SOD in sputum or bronchoalveolar lavage does not need to mirror its in situ expression in the lung interstitium, where it exerts its protective effect.
A relevant finding in our study is that immunoreactivity of fibulin-5 was significantly lower in alveolar walls, airway and vascular walls of COPD patients than in control subjects. Fibulin-5 and fibulin-4 are the most critical molecules for aiding assembly of elastic fibres,11,12 although fibulin-5 exhibits higher binding affinity to tropoelastin and is expressed at much higher levels than fibulin-4 in lung, aorta and skin.38 In a experimental model, fibulin-5-null mouse developed emphysema and disruption of elastic fibres thus suggesting the critical role of fibulin-5 in the maintenance of the lung architecture.39,40 Furthermore, a decrease of fibulin-5 may likely impair its binding to EC-SOD17 and affect negatively the protection against oxidative stress of the elastin matrix. This lack would favour elastin degradation by extracellular elastase activity thus accelerating the evolution to emphysema in COPD patients.
In spite of our immunohistochemical analyses revealing reduced presence of EC-SOD and fibulin-5 in alveolar walls of COPD patients, the difference between pulmonary mRNA levels of these proteins in COPD patients and controls was not statistically significant. This discrepancy regarding gene expression is not surprising, since mRNA expression is not necessarily related to tissue protein levels.41 Whole tissue samples for PCR analysis contain not only different lung compartments, but also circulating blood and secretions. On the contrary, immunohistochemistry in lung slices allows for detecting the markers in specific tissue localizations, such as alveolar walls. Indeed, in a previous study the levels of EC-SOD were high in sputum of COPD patients, but not in blood or BALF, whereas EC-SOD tissue immunoreactivity was lower in COPD patients.19
Our results suggest that patients with COPD have a reduced antioxidant activity in their lungs which make them more vulnerable to the disease. This finding supports the hypothesis that antioxidant therapy might be useful for these patients. However, further studies must be conducted in the clinical setting to address this probability.
In summary, our results show that EC-SOD and fibulin-5 expression are reduced in alveolar walls of patients with COPD, which also show alterations in elastic fibres. This reduction supports the hypothesis of an impaired pulmonary antioxidant response in COPD patients.
FundingThis study was funded by the Spanish Health Research Fund-Instituto de Salud Carlos III (FIS 04/0635) and the Sociedad Española de Pneumologia (SEPAR 165|2012).
Conflict of interestsThe authors declare that they have no conflict of interest.