Introduction
Ventilator-associated pneumonia (VAP) is a common complication in patients requiring mechanical ventilation. Its incidence varies widely (9%-70%) depending on the type of population studied1,2 and the diagnostic methods used. In recent years, the most appropriate method for diagnosing VAP has been the subject of growing controversy. For some time now, invasive techniques, such as the plugged telescoping catheter (PTC) and bronchoalveolar lavage via fiberoptic bronchoscopy have been mentioned in the literature as the most valid techniques for the diagnosis of this condition.3 These methods are, however, invasive, expensive, and not exempt from complications. Moreover, they require the use of an invasive procedure not always available in intensive care units. Some authors have indicated that quantitative cultures of tracheal aspirate (TA) are of equal diagnostic value to the invasive techniques.4,5 The current situation is, therefore, that the scientific community has not yet agreed upon the best routine technique for the diagnosis of VAP.6-11 The objective of this study, proposed by the Infectious Diseases Work Group of the Spanish Society of Intensive and Critical Care Medicine and Coronary Units (GTEI-SEMICYUC), was to investigate the validity of quantitative cultures of TA and compare that method with another conventional method generally considered valid, such as PTC, in patients with suspected VAP.
Materials and Methods
Patient Selection
This prospective, multicenter study enrolled patients undergoing mechanical ventilation for more than 72 hours who were admitted over the period of one year to the intensive care units of the 10 participating hospitals: Hospital Clínic and Hospital del Mar in Barcelona, Hospital de Navarra, Hospital de la Princesa in Madrid, Hospital Río Hortega in Valladolid, Hospital Valme and Hospital Duque del Infantado in Seville, Hospital Son Dureta in Palma de Mallorca, Hospital Parc Taulí in Sabadell, and Hospital Conxo in Santiago de Compostela. The following variables were recorded for each patient: number of days on mechanical ventilation at the time of the study, underlying disease, white blood cell count, positive end-expiratory pressure, simplified acute physiology score (SAPS),12 clinical pulmonary infection score (CPIS)13 for VAP, presence of acute respiratory distress syndrome, antibiotics taken during the week prior to the study (number and type), and total duration of antibiotic therapy. Quantitative TA and PTC were performed in patients suspected of having VAP on the basis of the clinical criteria listed below. The diagnostic procedures to be carried out (TA and PTC) were explained to all patients or to their close family members, and informed consent was requested. The procedures were performed in the same way in all the hospitals: TA first, and then PTC.
Study Protocol
TA specimens were obtained by sterile means using an aspiration catheter and were placed in sterile containers. Then, without interrupting mechanical ventilation, the fiberoptic bronchoscope was passed through the endotracheal tube via a special adaptor using the standard technique described in the literature,3,14 and without aspirating through the internal channel prior to collection of respiratory secretions. Local anesthetics were not used. Then the PTC sample was collected from the area of maximum local inflammation and purulence using the procedure described by Wimberley et al.15 In the case of diffuse infiltrate, the specimen was taken from the right lower lobe. Specimens were dispatched immediately to the laboratory for bacteriological processing. Two blood samples for culture were extracted prior to the above procedure.
Diagnostic Criteria
Suspicion of VAP was based on the presence of a new and persistent pulmonary infiltrate, together with clinical signs indicative of lower respiratory tract infection (fever ≥38°C or hypothermia of ≤35°C, increase in the volume and/or purulence of respiratory secretions), and laboratory findings (leukocytosis ≥12 000/µL or leukopenia ≤4000/µL). The following criteria were used to confirm the diagnosis of pneumonia: clinical response to appropriate antibiotic treatment, absence of an alternative diagnosis during follow up, cavitation of an infiltrate, or autopsy data demonstrating the existence of pneumonia. Pneumonia was considered not to be present in patients for whom an alternative diagnosis emerged during follow up and who did not require subsequent antibiotic treatment, and in patients whose antibiotic treatment was withdrawn when the absence of pneumonia was confirmed. These patients were included in the control group. Patients not suspected of having pneumonia and in whom no radiologic evidence of infiltrate was observed were also included as controls.
Microbiological Processing
All the samples obtained were cultured quantitatively using serial dilutions. The TA samples were mechanically homogenized using glass beads and vortexed for one minute. Serial dilutions of the TA were prepared with normal sterile saline solution in the proportion 1:1. The dilutions were then inoculated in various agar culture media and were incubated at 37ºC in aerobic conditions. The personnel in the microbiology laboratory were not aware of the diagnosis of the patients who produced the specimens, but they were aware of the type of sample delivered in each case.
All microorganisms isolated were identified by standard laboratory methods. Bacterial pathogens in concentrations of ≥103 cfu per mL in cultures of PTC specimens, and ≥106 cfu/mL in cultures of TA were considered to be causative agents of VAP. The growth of microorganisms that are ordinarily not pathogenic, such as Streptococcus viridans and Staphylococcus epidermidis, were in general considered to be colonization.
The identification of Legionella spp, mycobacteria or Aspergillus spp was considered diagnostic irrespective of the colony count obtained. Isolations of Candida spp were only considered significant when associated with positive blood cultures, or when histological evidence of pneumonia caused by Candida was present.
Statistical Analysis
The data were analyzed with the statistical software package SPSS for Microsoft Windows, version 6 (Chicago, Illinois, USA).
Quantitative variables are expressed as means (SD), and qualitative variables as the frequency of distribution of each one of the categories. Means were compared the Student t test or Mann-Whitney U-test depending on whether or not the distribution of the variable was normal.
Causative variables were compared using the χ² test. When any of the expected values in a 2x2 table were below 5, the Fisher exact test was used.
Sensitivity and specificity were calculated for a specified 95% CI.
The sensitivity and specificity of the different methods were compared (separately for patients with and without pneumonia) using the McNemar test.
The level of statistical significance accepted was 95% (P<.05).
Results
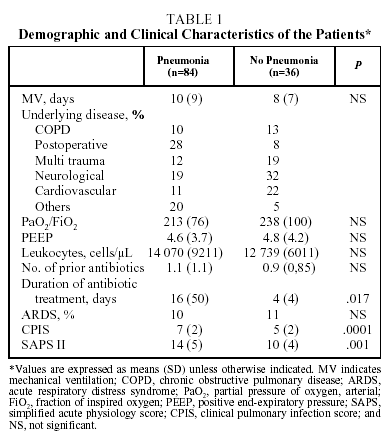
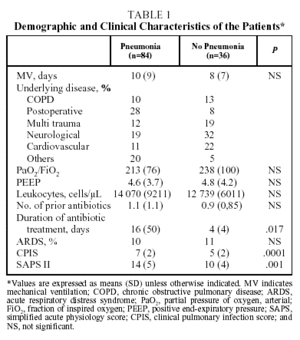
A total of 120 cases were studied, of which 84 were diagnosed with pneumonia and 36 were not (control group). Of the 36 cases without pneumonia, 25 had some kind of radiologic infiltrate. No significant differences were found between the baseline variables of the patients with and without pneumonia except for CPIS and SAPS II scores and the duration of antibiotic treatment, which were all significantly higher in the group with pneumonia (Table 1).
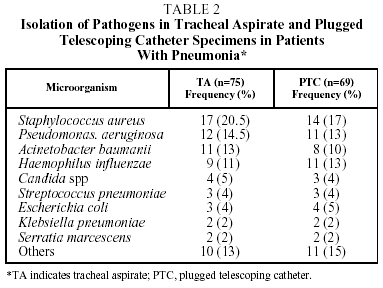
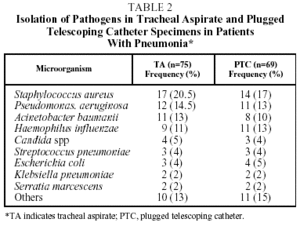
The organism most often isolated both by TA and PTC was Staphylococcus aureus, followed by Pseudomonas aeruginosa and Acinetobacter baumannii by TA, and Haemophilus influenzae and P aeruginosa by PTC. Table 2 shows the different organisms isolated using the 2 diagnostic methods in patients with pneumonia, and their frequency.
On 3 occasions primary pathogens were detected in the TA specimens that were not isolated in those obtained by PTC: Aspergillus spp, Mycobacterium tuberculosis and Legionella pneumophila.
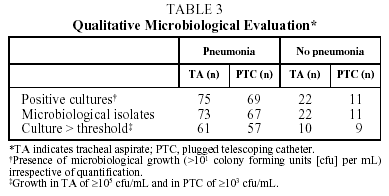
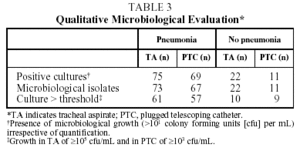
Table 3 shows the number of positive cultures and the total number of microorganisms isolated with each technique in the 2 groups studied. In most cultures, growth of microorganisms was obtained independently of the quantification value and technique used.
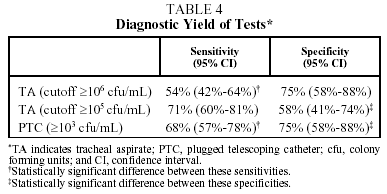
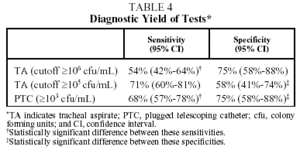
Table 4 shows the diagnostic yield for TA at 2 different cutoff points for bacterial counts (≥105 and ≥106), and for PTC. The sensitivity of TA with a cutoff of ≥105 and that of PTC were significantly higher than that of TA with a cutoff of ≥106 (P=.0001 and .01, respectively), and the difference between TA≥105 and PTC≥103 was not significant (P=.40). The kappa (κ) statistic for the agreement between PTC≥103 and TA≥106 was 0.41, and the same statistic between PTC≥103 and TA≥105 was 0.63.
Comparison of the specificities of the tests gave the following results: the specificity of TA≥106 and PTC≥103 was significantly higher than that of TA≥105 (P=.014), but no significant difference was found between TA≥106 and PTC≥103 (P=1.00). The κ statistic for the agreement between TA≥106 and PTC≥103 was 0.70, and κ between TA≥105 and PTC≥103 was 0.63.
Positive predictive values were 80% (69%-88%), 83% (71%-92%) and 86% (76%-94%) for TA≥105, TA≥106 and PTC≥103 respectively.
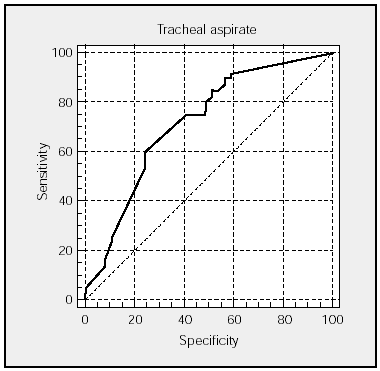
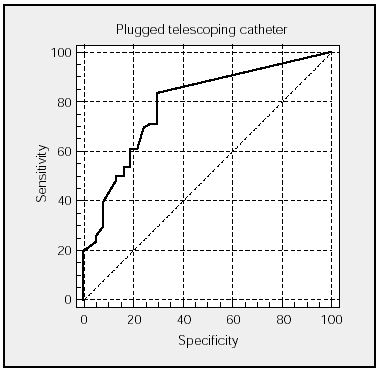
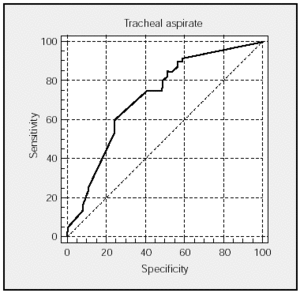
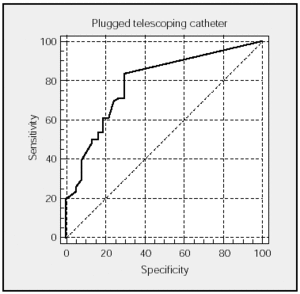
Figures 1 and 2 show the ROC curves for TA and PTC with the corresponding areas under the curve and CI. The sensitivity/specificity relationship is very similar in both techniques.
Fig. 1. ROC curve of tracheal aspirate specimens. Area under the curve: 0.716 (95% confidence interval, 0.625-0.746).
Fig. 2. ROC curve of plugged telescoping catheter specimens. Area under the curve: 0.790 (95% confidence interval, 0.705-0.860).
Of a total of 84 cases of pneumonia, concordant isolations (both tests positive for the same microorganism or both tests negative) were produced with both TA and PTC in 71 cases (83.8%), and the results did not agree in 13 cases (16.2%) (Table 5). The analysis of the association between the quantitative cultures of microorganisms that were detected with both TA≥105 and PTC produced a correlation coefficient (r) of 0.139 (P=.329).
Discussion
The most noteworthy finding in this study was that, when a cutoff point of 106 cfu/mL or higher was used for TA in the diagnosis of VAP, reasonable specificity was obtained (>70%), similar to that obtained with PTC, although the sensitivity of TA at ≥106 was lower than that of both PTC and TA with a cutoff of ≥105 cfu/mL. When the cutoff of TA was changed to ≥105, the sensitivity did not differ significantly from that of PTC. These results show an acceptable yield for the clinical use of quantitative cultures of TA in the diagnosis of VAP, making it an alternative to bronchoscopic methods. Our findings are consistent with those of previous studies, which mention a cutoff point of ≥106 cfu/mL used to differentiate between colonization and infection with TA.5 In 2 earlier studies, sensitivity and specificity values of ≥70% (70% and 72%, respectively) were obtained using a cutoff of 105 cfu/mL, and the results of the present study confirm those findings.4,16
It is known that both PTC and bronchoalveolar lavage are valid methods for the diagnosis of VAP.1,17 On the other hand, the currently most widely held opinion is that the use of qualitative cultures of TA is not a very specific diagnostic method owing to the airway colonization found in this type of patient. Although some groups still continue to use qualitative cultures of TA in the diagnosis of patients with suspected VAP, few doubts remain about the low specificity of the technique. Two earlier studies, carried out by Torres et al3 and Chastre et al,14 which evaluated the methods used to diagnose lower respiratory tract infection in patients on mechanical ventilation, found specificities as low as 14% and 29%, respectively.
In recent years, various investigators have tried to improve bronchoscopic procedures for the diagnosis of VAP. In a relatively surprising manner some of them have achieved acceptable results using methods that do not involve obtaining samples from the area close to the inflamed site of the infection. Blind sampling using bronchoalveolar lavage (protected and unprotected) or using PTC has been shown to produce a high diagnostic yield given the multifocal and dynamic nature of VAP.18-22
On the other hand, quantitative analysis of TA has been recommended as a simple and useful procedure in the diagnosis of VAP,4,7,23 and has the added advantages of being a noninvasive and more inexpensive technique. This study showed low sensitivity and acceptable specificity and a positive predictive value for TA with a cutoff of ≥106 in the diagnosis of VAP. Sensitivity improved with a cutoff of ≥105. PTC, on the other hand, showed better results in sensitivity and in positive predictive value. The specificity of PTC was the same as that of TA at a cutoff of ≥106.
Reasonable agreement was found in this study between the results from TA and PTC samples. The same microorganism was obtained in 51 positive cultures from 84 patients with pneumonia (60%), and 20 negative concordant results were obtained (23.8%). Overall agreement was 83%. In only 2 cases (2.3%) were both tests positive but with different microorganisms (Table 5).
Papazian et al24 compared PTC and TA in 35 mechanically-ventilated patients with bronchopneumonia, and reported a 93% agreement between the microorganisms cultured with the two techniques. However, the quantitative correlation between cultures obtained using the different techniques was not high.
One of the aspects of VAP diagnosis that has given rise to most debate is the role played by invasive and noninvasive methods in the prognosis of these patients and the antibiotic therapy administered. The debate still continues, but some aspects have become clearer. When only randomized clinical trials are analyzed, we find that Sánchez-Nieto et al25 in a pilot study of 51 patients observed that bronchoscopic methods gave rise to more changes in initial antibiotic treatment (42% as opposed to 16%; P<.05), although no significant differences in mortality, either global or attributable, or in morbidity were observed. A limitation of that study was its small sample size and the absence of a standard treatment protocol in the invasive group. A study carried out by Solé Violan et al26 demonstrated that while antibiotic therapy was modified more often based on information from invasive techniques, the changes had no clear effect on mortality, length of stay in the intensive care unit, or duration of mechanical ventilation. Ruiz et al27 compared 76 patients with suspected VAP (39 noninvasive and 37 invasive) and concluded that the diagnostic yield for VAP was similar with both techniques, as was mortality at 30 days, number of days on mechanical ventilation, and length of stay in the intensive care unit. The cost of the invasive investigation was obviously higher.
A study published by Fagon et al28 has reported positive results with respect to the reduction of mortality on day 14, lower sequential organ failure assessment scores on days 3 and 7, a reduction in the use of antibiotics, and more days free of antibiotics with the invasive technique. The limitation of this study was that qualitative cultures of TA were used, thereby limiting comparison with the other studies.
The results obtained using quantitative cultures of TA and a threshold of ≥106 cfu/mL as the differentiator between colonization and infection can be considered relatively acceptable as part of the clinical approach to patients with suspected VAP when the possibility of using bronchoscopic techniques is not available, even though overall they are not as satisfactory as the results obtained with PTC.
*Other members of the Work Group on Ventilator-Associated Pneumonia: M. El-Ebiary (Hospital Clínic, Barcelona); C. León, M. Guerrero and F. Ortega (Hospital Valme, Seville); F. Castillo (Hospital Duque del Infantado, Seville); R. Jordá (Hospital Son Dureta, Palma de Mallorca); J. Barado (Hospital de Navarra); J. Vallés and J. Rello (Hospital Parc Taulí, Sabadell, Barcelona); A. Santos Bouza (Hospital Conxo, Santiago de Compostela, A Coruña).
Correspondence: Dr. A. Torres Martí.
Unidad de Vigilancia Intensiva Respiratoria (UVIR). Instituto Clínico de
Neumología y Cirugía Torácica. Hospital Clínic. Instituto de Investigaciones
Biomédicas August Pi i Sunyer (IDIBAPS). Universidad de Barcelona.
Villarroel, 170. 08036 Barcelona. Spain.
E-mail: atorres@medicina.ub.es
Manuscript received October 31, 2002.
Accepted for publication February 18, 2003.